- LOGIN
- MemberShip
- 2026-06-06 10:09:37
- Company
- The 10th anniversary of 'Tagrisso' approval in Korea
- by Son, Hyung Min May 20, 2026 02:28pm
- As the therapeutic landscape for EGFR-mutated non-small cell lung cancer (NSCLC) shifts toward all-cycle management from early to metastatic stages, AstraZeneca’s 'Tagrisso (osimertinib)' is recognized as a key driver of treatment strategy changes, based on clinical evidence accumulated over the past decade.Industry analysis suggests that by securing clinical data demonstrating not only progression-free survival (PFS) but also overall survival (OS) improvements and reduced post-operative recurrence, the treatment paradigm for EGFR-mutated lung cancer is transitioning from a life-extension focus to expanding curability.Se Hoon Lee, Professor of Hematology-Oncology at Samsung Medical CenterOn May 19, AstraZeneca Korea hosted a press conference at the Plaza Hotel in Jung-gu, Seoul, to commemorate the 10th anniversary of Tagrisso’s approval in South Korea, sharing major global clinical trial outcomes and the future direction of its treatment strategies.Tagrisso was first approved in South Korea in May 2016 as a second-line therapy for patients with locally advanced or metastatic EGFR T790M mutation-positive NSCLC. This drug received an expanded indication for first-line monotherapy in 2018. In 2021, Tagrisso became the only EGFR tyrosine kinase inhibitor (EGFR-TKI) to secure an indication for adjuvant therapy following complete surgical resection.Furthermore, its indication for combination use with platinum-based chemotherapy, broadening its therapeutic scope in 2024 byCurrently, Tagrisso is the only third-generation EGFR-TKI recommended as a "Preferred" Category 1 option by the National Comprehensive Cancer Network (NCCN) guidelines for both first-line monotherapy and combination chemotherapy regimens. Its application as a post-operative adjuvant therapy also holds a Category 1 recommendation.Se Hoon Lee, Professor of Hematology-Oncology at Samsung Medical Center, said, "The lung cancer targeted therapy paradigm has evolved most rapidly for the EGFR mutation," and added, "With a high prevalence of these mutations among Korean patients and subsequent improvements in survival rates driven by targeted therapies, the outpatient landscape for long-term patient management has fundamentally transformed."Professor Lee added, "The advancement of EGFR-TKIs, including those for Tagrisso, established the benchmark for developing subsequent mutation-targeted strategies in ALK, ROS1, and KRAS." Professor Lee said, "Progression beyond treating metastatic disease to a stage where we aim for a cure in Stage I and II patients is highly meaningful."Ji Yoon Lee, AstraZeneca Korea's Medical Affairs Unit DirectorIndeed, Tagrisso has built clinical evidence across various stages of the disease through major global clinical trials, including FLAURA, FLAURA2, ADAURA, LAURA, and AURA3.In the FLAURA study, Tagrisso became the standard of care by demonstrating both overall survival (OS) and progression-free survival (PFS) benefits in the first-line treatment of advanced NSCLC positive for EGFR Exon 19 deletions or Exon 21 (L858R) mutations.After that, in the FLAURA2 study, the final analysis of Tagrisso combined with platinum-based chemotherapy demonstrated a median OS of 47.5 months. Experts highlighted that the combination also showed a trend toward a reduced risk of death in patients with central nervous system (CNS) metastases compared to the control group.In the ADAURA study, which evaluated post-operative adjuvant therapy, the analysis of the overall patient population with Stages IB to IIIA disease demonstrated a 73% reduction in the risk of disease recurrence or death compared with the control group, highlighting its potential to expand into early-stage treatment.Additionally, the LAURA study reported a statistically significant prolongation of progression-free survival in patients with unresectable Stage III disease, extending the drug's targeted strategy into the locally advanced setting.Ji Yoon Lee, AstraZeneca Korea's Medical Affairs Unit Director, said, "Over the past 10 years, Tagrisso has continuously accumulated core clinical evidence within the global and domestic EGFR-mutated NSCLC treatment landscape," and added, "We will continue our research and development efforts to advance tailored treatment strategies optimized for specific patient characteristics and disease stages."Hyun Ju Lee, AstraZeneca Korea's Oncology Business Unit director, stated, "The 10th anniversary of Tagrisso’s approval in South Korea represents the shifting paradigm of EGFR-mutated lung cancer care. Based on our oncology R&D capabilities and leadership, we will continue to contribute to improving a patient-centric treatment environment."
- Policy
- Generic market for anti-cancer drug Xtandi heats up
- by Lee, Tak-Sun May 20, 2026 02:28pm
- Original Xtandi TabThe generic market for Xtandi (enzalutamide, Astellas), a blockbuster prostate cancer treatment, is in turmoil.With the expiration of the substance patent approaching this June and approvals for soft capsule generics coming one after another, latecomers are now setting their sights squarely on the ‘tablet’ market—the original manufacturer’s latest strategic weapon— rapidly expanding the competitive battlefield.According to industry sources, on April 24, applications for approval of three film-coated tablet strengths of enzalutamide (40mg, 80mg, and 160mg) were simultaneously submitted to the Ministry of Food and Drug Safety. Industry attention is focused on these applications because they aim to secure ‘first generic exclusivity,’ considered the key to early market dominance.From capsules to tablets…follow-up approvals accelerate after breaking the 2033 barrierOriginally, the Xtandi generic market had been developing primarily around soft capsule formulations timed with the expiration of the substance patent on June 27. Major pharmaceutical companies, including Alvogen Korea, Daewon Pharmaceutical, Hanmi Pharmaceutical, GL Pharma, and Dongkook Pharmaceutical, have already obtained approvals for soft capsule generics and are preparing to launch.In response, original manufacturer Astellas Pharma Korea introduced ‘Xtandi Tab’ (40mg and 80mg) with improved dosing convenience and completed reimbursement listing last April. This was part of a defensive strategy to shift the market’s center of gravity toward tablets and fend off the generic competition. This was because the tablet formulation was protected by a separate composition patent valid until September 2033.However, the defensive line collapsed as domestic pharmaceutical companies successively succeeded in circumventing this 2033 formulation patent through a series of negative scope confirmation trials. The latest film-coated tablet generic applications are therefore interpreted as the first step toward realizing a ‘dual competition’ structure between dosage forms after neutralizing the patent barrier.Addition of a 160mg high-dose option… Aiming for exclusivity through first generic approval rightsA notable feature of the film-coated tablet lineup applied for this time is the inclusion of a 160mg high-dose product, which is not available in the original Xtandi Tab lineup (40mg, 80mg).Currently, patients face the inconvenience of having to take four 40mg soft capsules at once each day. In response to the original manufacturer introducing an 80mg tablet form to reduce the number of pills taken to two, the generic manufacturer has launched a counterattack by developing a 160mg ultra-high-dose formulation that requires taking “just one pill a day.” This is a strategy to surpass the original in terms of convenience.Furthermore, if these companies secure first generic exclusivity based on the latest applications, they will gain the privilege of blocking other tablet generics from entering the market for a certain period while competing exclusively against the original product.A pharmaceutical industry official predicted, “If the ‘first war’ takes place in June when soft capsule generics flood the market immediately after the substance patent expires, then approvals and reimbursement listings for tablet generics pursuing first generic exclusivity will ignite the ‘second war’ in the second half of the year.”As a direct confrontation approaches between the original company’s market-defense strategy through tablet conversion and domestic pharmaceutical firms seeking early dominance of the tablet market through patent circumvention and independent high-dose lineups, the industry’s attention is focused on who will take the lead in the enzalutamide market. Xtandi recorded domestic sales of KRW 38 billion last year, according to UBIST data.
- Policy
- MFDS to maintain small-packaging drug regulations
- by Lee, Tak-Sun May 20, 2026 02:28pm
- The Ministry of Food and Drug Safety has recommended that pharmaceutical companies voluntarily adjust their own packaging practices rather than granting the industry’s request to relax small-packaging regulations due to stable drug supply concerns arising from the Middle East conflict. The move is interpreted as maintaining the small-packaging regulations while expanding recognition of exceptional cases. Industry requests for a grace period on administrative penalties this year appear to have been rejected following opposition from pharmacy organizations and related groups.This is interpreted as the government’s intention to expand the recognition of exceptions while maintaining compliance with the small-packaging regulations. It is analyzed that the industry’s initial request for a suspension of administrative penalties this year was rejected due to opposition from groups such as the Korean Pharmaceutical Association.According to industry sources on the 18th, the MFDS recently sent an official notice to pharmaceutical associations requesting that each pharmaceutical company voluntarily adjust small-package supply ratios.Under the current “Regulations on Supply of Pharmaceuticals in Small Packaging Units,” pharmaceutical manufacturers and importers are required to supply at least 10% of their annual production or import volume to pharmacies in small packaging units.The standards are set at 30 tablets or fewer for bottle packaging, 100 tablets or fewer for blister packs, and 500 mL or less for syrups. Violations result in administrative penalties such as suspension of sales operations.The mandatory small-packaging rule was originally introduced to reduce excessive inventory at pharmacies and the resulting social costs from the disposal of unused stock.Recently, the pharmaceutical industry requested relaxation of the small-packaging regulations due to shortages of pharmaceutical packaging materials caused by instability in naphtha supply stemming from the Middle East conflict. In particular, the industry requested suspension of administrative penalties this year, even if mandatory small-package production ratios were not met.On the 2nd of last month, MFDS Minister Yu-Kyoung Oh visited JW Pharmaceutical to inspect IV solution packaging and announced that the ministry would swiftly pursue proactive administrative measures, including the relaxation of the small-packaging obligations.Following this, the MFDS working-level department reviewed the potential easing of the small-packaging requirement. However, pharmacy organizations, including the Korean Pharmaceutical Association, opposed such measures, citing concerns such as inventory disposal, making policy decisions difficult.Ultimately, it appears the MFDS has decided to adhere to the regulations while actively utilizing exceptions.In an official notice, the MFDS stated, “The MFDS has recommended that pharmaceutical companies voluntarily reduce the use of drug packaging materials, such as by adjusting the ratio of small-volume packaging.”The ministry added, “While complying with the minimum standards for small-package supply in consideration of the purpose and intent of the ‘Regulations on Supply of Pharmaceuticals in Small Packaging Units,’ if compliance becomes difficult due to shortages in packaging material raw supplies caused by wartime conditions, companies may submit statements on product-specific explanations and supporting materials to the MFDS under Article 5 of the regulation to receive exemption from the small-package requirements.”The measure is ultimately interpreted as maintaining the small-package supply obligation while broadly recognizing exception products.A pharmaceutical industry official stated, “In response to opposition from the Korean Pharmaceutical Association and others, it appears that the request for a suspension of administrative penalties requested by the industry has not been accepted. However, the MFDS’s request for voluntary adjustment by pharmaceutical companies can be interpreted as an intention to actively recognize exceptions.”
- Opinion
- "New triple-combination therapy Levosartan Plus for hypertension"
- by Kim, Jin-Gu May 20, 2026 02:28pm
- Ahngook Pharmaceutical recently launched ‘Levosartan Plus Tab’, a triple combination therapy for hypertension. The product contains valsartan (an ARB) + S-amlodipine (a CCB) + indapamide (a diuretic). It is the first time this active pharmaceutical ingredient (API) combination has been approved in South Korea.Professor Seok-Min Kang of the Division of Cardiology at Severance Hospital, who led the clinical trial for regulatory approval, stated, “As the comorbidities of patients with hypertension become increasingly diverse, the importance of combining drugs with different mechanisms of action (MOAs) is growing. It will be highly useful, particularly for elderly patients or high-risk hypertensive patients with multiple underlying conditions.”Korea's first triple combination hypertension therapy, additional blood pressure-lowering efficacy compared to dual therapyProfessor Seok-Min Kang of the Division of Cardiology at Severance HospitalThe approval basis of trial for Levosartan Plus Tab was conducted over 3 years, starting in April 2022, and enrolled 306 hypertensive patients across 30 hospitals in South Korea. The study evaluated the incremental blood pressure-lowering efficacy of the triple combination therapy compared with conventional ARB·CCB dual fixed-dose combinations.The trial results demonstrated that Levosartan Plus Tab achieved an additional reduction of 6.3 mmHg in systolic blood pressure (SBP) and 3.69 mmHg in diastolic blood pressure (DBP) compared to the control group (dual combination). After 10 weeks, the blood pressure normalization rate in the investigational group was statistically significantly higher than that of the control group.Professor Kang said, “Tertiary hospitals see many high-risk hypertensive patients with various comorbidities such as diabetes, chronic kidney disease (CKD), stroke, and myocardial infarction (MI), making blood pressure management challenging with single-mechanism therapies alone," and added, "This clinical trial confirmed that the combination of three components with distinct MOAs offers incremental efficacy in controlling blood pressure in actual Korean hypertensive patients.”“Indapamide lowers risk of electrolyte imbalance aberrations...Synergy with valsartan and S-amlodipine”The most differentiating component in this combination is the diuretic, indapamide. Previously, the South Korean market for hypertension combination drugs predominantly utilized diuretics from the chlorthalidone or hydrochlorothiazide classes.Professor Kang cited the HYVET (Hypertension in the Very Elderly Trial) study as evidence for indapamide. Published in 2008, the study demonstrated that indapamide-based treatment reduced the ▲risk of mortality ▲fatal stroke ▲heart failure incidence ▲cardiovascular events compared to placebo. “In elderly hypertensive patients, indapamide-based therapy showed positive outcomes not only in blood pressure control but also in terms of cardiovascular prognosis,” Professor Kang explained.Professor Kang highlighted safety as a key advantage of indapamide. He explained that while conventional diuretics often pose a burden to elderly patients by lowering serum sodium and potassium levels, indapamide carries a relatively lower risk of such adverse effects.“While chlorthalidone offers potent antihypertensive efficacy, it can induce hyponatremia and hypokalemia in some elderly patients,” Professor Kang said. “In contrast, indapamide tends to be used stably in clinical practice.” Kang further emphasized, “The risk of metabolic side effects, such as elevated cholesterol, is also relatively low, making it highly applicable for hypertensive patients with concurrent hyperlipidemia.”The other two components, valsartan and S-amlodipine, are also evaluated as having distinct therapeutic roles. Professor Kang anticipated synergistic effects arising from the unique strengths of each component.“Amlodipine is a long-established CCB-class antihypertensive drug, but it can cause side effects like edema in elderly patients,” Professor Kang said. “In contrast, because S-amlodipine isolates only the active S-enantiomer responsible for lowering blood pressure, it carries a lower risk of adverse events such as edema or facial flushing compared to conventional amlodipine.”Regarding valsartan, Kang noted, “It is a drug with significant clinical evidence. In addition to lowering blood pressure, valsartan acts as an RAS (renin-angiotensin system) blocker, providing renal and cerebral protection. It can be utilized with a relatively stable safety profile even in patients with early-stage diabetes or those with a high metabolic risk profile.”“Complementary actions of three components... Expected to benefit elderly and high-risk hypertensive patients”Professor Kang projected that the new combination therapy containing valsartan + S-amlodipine + indapamide would benefit hypertensive patients who are elderly or present with underlying comorbidities. “It could be considered a preferred option for hypertensive patients with early-stage diabetes, obesity, or chronic kidney disease,” Professor Kang said. “It is also suitable for patients who previously suffered from ankle edema when using standard CCBs.”Professor Kang noted that the competitive edge of this therapy among the numerous existing hypertension combinations lies in its drug synergy. “It is designed for stable blood pressure control with S-amlodipine, the enhanced antihypertensive efficacy of indapamide, and the long-term clinical evidence and organoprotective effects of valsartan to complement one another.,” Professor Kang assessed. “It is a combination designed not just for efficacy in lowering blood pressure, but also taking into account real-world clinical experience.”Professor Kang also anticipated improvements in patient medication adherence. “Most hypertensive patients are elderly and take a load of medicines,” Professor Kang noted. “Taking a single pill is far more convenient than taking three separate medications," and concluded that "This will improve patient adherence, leading to a reduction in cardiovascular events.”
- Policy
- SK plasma gains approval for Revolade generic
- by Lee, Tak-Sun May 20, 2026 02:28pm
- Original drug, Novartis’ ‘Revolade’SK Plasma has officially entered the generic market for Novartis’ rare disease treatment Revolade (eltrombopag olamine). Following Pharmbio Korea, which first entered the market, SK Plasma is joining as a latecomer, turning the competition among domestic generic manufacturers into a two-way race.On the 18th, the Ministry of Food and Drug Safety approved two dosage strengths (25mg and 50mg) of SK Plasma’s thrombocytopenia and severe aplastic anemia treatment, “Revolpag Tab.”The original drug for Revolpag is Novartis Korea’s ‘Revolade.’ The indications SK Plasma received approval for this time include all key efficacy and indication areas held by the original drug.The three approved indications include: ▲Treatment of thrombocytopenia in chronic immune (idiopathic) thrombocytopenia patients who showed insufficient response to corticosteroids or immunoglobulins; ▲ Treatment of thrombocytopenia to initiate and maintain interferon-based therapy in chronic hepatitis C patients, and as ▲First-line treatment of severe aplastic anemia in pediatric patients aged 2 years and older and adults in combination with immunosuppressive therapy, as well as treatment of severe aplastic anemia unresponsive to prior therapies.However, for chronic immune thrombocytopenia and chronic hepatitis C treatment, the drug is approved only for use in clinical conditions associated with increased bleeding risk and cannot be used for the purpose of normalizing platelet counts.Previously, SK Plasma successfully circumvented three “new pharmaceutical composition” patents that had served as the core barrier protecting Revolade, winning a negative scope confirmation trial after a dispute that went all the way to the Supreme Court. This approval was achieved after fully resolving patent risks.As a result, the domestic eltrombopag olamine market is expected to enter an intense competitive phase between Pharmbio Korea’s first-mover product “Elpag Tab” and SK Plasma’s late-entry “Revolpag Tab.” Elpag launched in October 2024.The Revolade market has continued to grow after reimbursement criteria were expanded in 2024, reaching import sales of approximately USD 5.23 million (around KRW 7.8 billion).A pharmaceutical industry official commented, “With Pharmbio Korea already in the market and expanding its market share, SK Plasma has secured approval and is leveraging its full indication coverage. Once reimbursement listing and pricing procedures are completed and the product launches in earnest, treatment options for physicians and patients will expand significantly.”
- Policy
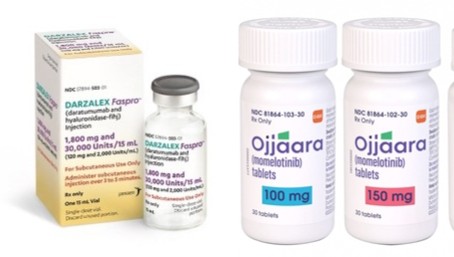
- Darzalex SC and Omjjara complete drug price negotiations
- by Jung, Heung-Jun May 19, 2026 11:08am
- Janssen Korea’s multiple myeloma treatment, Darzalex SC (daratumumab), is expected to be listed for reimbursement after completing drug price negotiations with the National Health Insurance Service.In addition, Korea GSK’s new myelofibrosis treatment Omjjara Tab (momelotinib) 100mg, 150mg, and 200mg has also reached a pricing agreement and will enter reimbursement listing.According to industry sources on the 18th, Darzalex SC and Omjjara Tab, which entered negotiations with the NHIS in March, recently finalized pricing agreements.Both drugs are new therapies that passed the Drug Reimbursement Evaluation Committee in January. Darzalex SC was recognized as appropriate for reimbursement as “combination therapy with bortezomib, cyclophosphamide, and dexamethasone in newly diagnosed light-chain amyloidosis patients.”Unless unexpected issues arise, both drugs are expected to proceed with reimbursement listing next month.Janssen Korea is continuing to expand the approved indications for Darzalex SC. Last April, the drug additionally received approval for 3 new indications, including combination therapy with bortezomib, lenalidomide, and dexamethasone (DVRd) for newly diagnosed transplant-eligible multiple myeloma patients. Accordingly, further applications for reimbursement expansion are expected.The reimbursement appropriateness for GSK’s myelofibrosis treatment Omjjara Tab was recognized at this year’s first DREC meeting for “treatment of intermediate- or high-risk myelofibrosis with anemia in adults,” provided that the price is set below the evaluation threshold.Omjjara passed the Cancer Drug Deliberation Committee in March last year, but submission to the reimbursement evaluation committee was delayed due to issues such as the selection of comparator drugs. After approximately 10 months, it was resubmitted and cleared the first hurdle at this year’s DREC meeting.Like Darzalex SC, it entered NHIS drug price negotiations in March and ultimately reached a final agreement. Reimbursement listing is scheduled for next month.Meanwhile, negotiations for Mounjaro, which entered price negotiations in January this year, broke down. Although the company applied for the flexible pricing contract system, it is reported that the parties reportedly failed to narrow differences regarding the separately negotiated amount.
- Policy
- Multi-listing rule hits non-innovative companies harder
- by Jung, Heung-Jun May 19, 2026 11:08am
- The ‘multi-product listing management’ rule, introduced to prevent excessive proliferation of generics, will operate far more harshly against non-innovative companies.While innovative and quasi-innovative companies will receive a three-year grace period, general companies will face immediate price cuts to 30.6%-38.5%, depending on whether they pass bioequivalence testing, leading to a significant widening of the price gap.According to industry sources on the 18th, the impact of the multi-product listing penalty system, newly introduced starting this August, will differ dramatically depending on a company’s innovation classification.Impact of the multi-product listing penalty systemThe Ministry of Health and Welfare recently issued an administrative notice revising the “Standards for Drug Price Determination and Adjustment,” newly introducing a provision applying 85% of the calculated price once the total number of identical formulations reaches 14 or more. Drugs eligible for additional premiums will also be subject to the 85% rule once the premium period ends.For innovative and quasi-innovative companies, the drug price premium period is 1+3 years if domestic manufacturing conditions are met. In other words, a 3-year grace period applies to the multi-product listing penalty as well.In contrast, general companies face an immediate reduction from the standard calculation rate of 45% to 38.25% once the number of listed products exceeds 14. If they failed to conduct their own bioequivalence testing, the price is reduced by an additional 20%, falling to 30.6%.For example, if a generic drug with an original product price of KRW 1,000 reaches 14 or more listed generics, an innovative company would maintain a price of KRW 600 won, reflecting a 60% premium, for four years before eventually dropping to KRW 382.5. A quasi-innovative company would maintain KRW 500 under a 50% premium for four years before dropping to KRW 382.5.However, the generic drug of a general company would sell at KRW 450 for only one year under the standard calculation rate before falling to KRW 382.5.This means that while innovative and quasi-innovative companies maintain prices of KRW 500–600 for three years by meeting domestic manufacturing requirements, general companies immediately face deteriorating profitability at KRW 382.5.If an ordinary company additionally failed to meet the bioequivalence testing requirement, the price would fall to KRw 306, creating nearly a twofold price gap compared with innovative companies.Ultimately, this structure ensures that the penalty for listing multiple products is applied more strictly to general companies, and a sales gap with innovative and quasi-innovative companies is inevitable during the grace period for the price adjustment.In effect, general companies effectively lose the advantage of product listing after the 14th generic entrant, subject to the multi-product pricing rule, enters the list.
- Opinion
- "Eliminating generic pharma's shared bio-equivalence·illegal CSO"
- by Lee, Jeong-Hwan May 19, 2026 11:08am
- Won-jun Jo, Chief Representative for Health and Welfare of the Democratic Party's Policy Committee"The goal of the Democratic Party’s drug pricing system reform is not simply to reduce the National Health Insurance budget by cutting generic prices. The goal is to eliminate the space for paper-company pharmaceutical companies and free-riders that parasitize the pharmaceutical ecosystem, thereby establishing an industry order in which companies that properly manufacture and invest in novel drugs and supply unstable medicines are dramatically rewarded. This is also a principle we have firmly held in our general and presidential election pledges. Now that the reform plan has been finalized, the ruling party will work with the government to complete the remaining puzzle pieces of the drug pricing system through the abolition of the generic '1+3' shared bioequivalence system and follow-up measures to regulate unsound CSOs (pharmaceutical sales marketing agencies)."As the government finalized the broad framework and key details of the drug pricing system reform plan, the Democratic Party of Korea drew attention by expressing a strong determination to accelerate policies aimed at eliminating illegal rebates through the abolition of the generic 1+3 shared bioequivalence test system and the regulation of poor CSOs to complete the mission of "pharmaceutical and biotech industry innovation."By lowering the generic drug price calculation rate from 53.55% to 45% and strengthening selective and differential incentives compared to the past for innovative pharmaceutical companies, semi-innovative pharmaceutical companies, and companies contributing to supply-unstable medicines, the party intends to thoroughly exclude name-only pharmaceutical companies from the market that do not align with the values of pharmaceutical industry development, healthy employment and job creation, and the establishment of a sound medicine distribution structure.On the 17th, Won-jun Jo, Chief Representative for Health and Welfare of the Democratic Party's Policy Committee, met with DailyPharm and expressed, "Starting with the Ministry of Health and Welfare's drug pricing system reform plan, we are discussing follow-up policies that can send a clear and unambiguous message to the pharmaceutical and biotech industry, encompassing both novel drugs and generics, as well as the CSO industry."Jo evaluated that the Ministry of Health and Welfare's drug pricing system reform plan, which passed the Health Insurance Policy Review Committee and is set for implementation this year, deleted remaining inefficiencies and irrationalities while presenting a future vision for South Korea's pharmaceutical industry to pursue.The aim is to smartly restructure the reward system for pharmaceutical companies that contribute to novel drug research and development (R&D), the stable supply of essential medicines, and the growth of the national pharmaceutical industry, so that the nation and its citizens achieve a tangible level of practicality they can directly perceive."Novel Drug National Health Insurance Reimbursement, from an 'Admissions Quota System' to a 'Graduation Quota System'Jo explained that the discussions and designs for the drug pricing system reform with the Ministry of Health and Welfare (MOHW) focused primarily on significantly narrowing the gap between the public and novel drugs under limited health insurance budget conditions, and satisfying the social demand to resolve the issue of essential medicines that frequently go out of stock due to supply instability.Because the expansion of public access to novel drugs was incorporated into the reform plan, some civil and patient advocacy groups raised concerns that it might grant excessive privileges to global pharmaceutical companies focused on novel drugs, a perspective Jo believes should also be adequately acknowledged.In particular, Jo emphasized that the speed of implementing health insurance reimbursement to novel drugs is not the only factor that is increasing. He stated that, following the rapid reimbursement of a novel drug, a follow-up policy based on a mechanism to remove it from reimbursement immediately will be implemented if its drug efficacy cannot be proven through RWD.Jo said, "Until now, the barrier to entering novel drug reimbursement was high, and once a drug received reimbursement, it was a structure where reimbursement was continuously recognized thereafter. There were criticisms that this system was actually more irrational," and explained. "A judgment was made that it is more efficient to change the reimbursement barriers and criteria to be relatively flexible, but transition to a system where reimbursement is deleted if real-world prescription efficacy data is not clear during post-evaluations."He added, "We will proceed with follow-up work to prepare policies capable of determining reimbursement ejection based on real-world data. This is a package that the drug pricing system reform plan must accompany. To use a simple analogy to the college admission system, we are transitioning novel drug reimbursement from an admissions quota system to a graduation quota system. It changes to a graduation quota system where reimbursement cannot be maintained unless clear evidence of drug efficacy is proven after entering reimbursement.""Free-riding pharmaceutical companies must be sorted out for good generics to distribute"Regarding the significance of the generic drug pricing system reform, Jo summarized it as meaning "there are no drug prices to give to free-riding pharmaceutical companies.""Can a company that does not perform its own bioequivalence testing, its own clinical trials, or even its own direct manufacturing be called a pharmaceutical company?" Jo asked and assessed that "If you give the same drug price to a consignment pharmaceutical company simply because it has the same ingredient, that company has no reason to invest in personnel or spend money on infrastructure. Consequently, they become entirely consumed by maximizing generic sales competitiveness, which deteriorates into a structure that inevitably links to illegal rebates."Jo noted, "The major meaning of the generic reform plan is not about cutting drug prices, but rather about rewarding only those pharmaceutical companies that manufacture proper generics with a proper drug price. We believed a policy response was needed to address whether it is right to continue embracing consignment generics within the health insurance system. Therefore, we adjusted and newly established criteria for innovative and semi-innovative pharmaceutical companies, and embedded regulations in the reform plan that can yield benefits for companies contributing to supply-unstable medicines."Regarding the reduction of the generic calculation rate to 45%, Jo evaluated, "Although it may not be completely satisfactory to either the government or the pharmaceutical industry, an agreement was reached at a level that cannot be viewed as fatal to either side at the same time.""The pharmaceutical industry demanded 48% as a baseline margin, while the MOHW discussed the low 40% range. Looking only at the surface, it is the product of a social consensus, and political circles adjust aspects," he said. "It was also determined at a median level between the calculation values of Japan and France, which were referenced during the system design. This is why the pharmaceutical industry, which had major anxieties, was able to say after the system was finalized that it was difficult. They must endure a portion of it."Jo stated that the "graduation quota system" for novel drug reimbursement, the abolition of the 1+3 consignment generic system, and the eradication of illegal CSO rebates are the path the domestic pharmaceutical industry must take."Abolition of the '1+3' shared bioequivalence system is the goal"Jo asserted with confidence that the generic consignment bioequivalence system must be completely abolished so that paper-company pharmaceutical companies that free-ride on pharmaceutical industry development and the national health insurance budget disappear, establishing a pharmaceutical industry environment where only genuine pharmaceutical companies receive justified rewards.Jo's opinion that the current method, which allows three shared consignment pharmaceutical companies for every one pharmaceutical company performing a generic bioequivalence test, is a contradictory policy that stands at the opposite pole of the reformed drug pricing system.Jo announced that he will take the necessary legislative and administrative steps, alongside the MOHW and the Ministry of Food and Drug Safety, to implement a system that allows only a single generic per original medicine. This policy is expected to exert a significant impact on the overall structure of the domestic pharmaceutical industry, increasing the need to focus on future National Assembly legislation and government administrative movements."Currently, the 1+3 consignment bioequivalence generic system is permitted, but the rationale for why identical health insurance reimbursement drug prices should be given to three consignment generic items cannot be explained by any logic. It completely runs counter to the philosophy of the reformed drug pricing system," Jo emphasized. "In the past, it carried the meaning of partially cleaning up the market as a transitional phase, shifting from permitting unlimited bioequivalence tests to restricting it to 1+3, but now, the biggest characteristic of the reformed drug pricing system is that it will not give drug prices to pharmaceutical companies that free-ride by permitting generic consignment bioequivalence and manufacturing."Jo pointed out, "The consignment bioequivalence system has reached the point where it must be abolished. Previously, the MFDS also stated its position when announcing the 1+3 system that it was a temporary and provisional allowance. Some argue that the abolition of 1+3 will lead to job reductions. It is questionable what kind of industrial or national production consignment generic companies, which have a large proportion of paper companies, induce, or what employment creation effects they demonstrate.""Rebates exploiting rogue CSOs are evolving...We will eliminate these cases"Jo cited the elimination of CSO rebates as one of the critical follow-up measures that must be implemented after the drug pricing reform.Jo stated that, alongside the ejection of free-riding consignment pharmaceutical companies, regulations must be placed on malpractice in which some pharmaceutical companies exploit CSOs to continue illegal rebate sales operations to secure unfair profits, thereby amplifying the effects of the reformed drug pricing system."For good generics made by real pharmaceutical companies to be properly distributed in the market and for citizens to take their medications, the next targets for restructuring are unsound CSOs and the pharmaceutical companies that exploit them," Jo pointed out. "Like an open secret, some pharmaceutical companies select and operate with CSOs as indirect actors to diversify the risks and responsibilities of illegal rebates. These CSOs operate under a subcontracting and further re-subcontracting structure, ultimately making it ambiguous as to who bears the final liability for the rebate activity."Accordingly, he introduced, "Recently, some hospitals and directors have been disrupting the order of the sound medicine market by operating family CSOs as a method of evasion for family business succession to secure illegal profits. We are contemplating measures to strengthen CSO compliance alongside the Ministry of Health and Welfare. One of the measures under consideration is making it fundamentally impossible for a pharmaceutical company to cut off its relationship with a CSO to evade responsibility when a rebate is detected."In conclusion, Jo said, "We will strengthen regulations on malpractice CSOs through a joint penalty system for pharmaceutical companies and CSOs to eliminate problems where CSOs make it impossible to trace the origin of a rebate by subcontracting and re-subcontracting, and pharmaceutical companies cut relationships and deflect responsibility by blaming the CSO. Legislation that clarifies the mutual chain of responsibility between a pharmaceutical company and its contracted CSO will follow to support the success of the drug pricing reform plan."
- InterView
- [Desk's View] The 16-year shackle around K-toxin must be broken
- by Lee, Seok-Jun May 19, 2026 11:08am
- The declassification of botulinum toxin as a National Core Technology is under discussion. This time, the atmosphere is noticeably different. It has been reported that the Biotechnology Expert Committee under the Ministry of Trade, Industry and Energy (MOTIE) has reached internal consensus in favor of declassification. All that remains now is a conclusion.According to MOTIE and industry sources, the Biotechnology Committee, operating under the Industrial Technology Protection Committee, is currently reviewing whether to remove botulinum toxin product manufacturing technology and its strains from the National Core Technologies designation list. MOTIE states that this is part of a routine, periodic review process to reassess 79 national core technologies across sectors such as semiconductors, displays, and biotechnology.However, the market does not view this as a mere routine review. This is because a regulatory controversy that has persisted for 16 years is shifting direction for the first time.In fact, sources inside and outside the industry observe that the Biotechnology Committee has reached consensus primarily on the declassification view. Assessments also indicate that the atmosphere shifted noticeably after MOTIE recently replaced several long-tenured, consecutively reappointed committee members.This change is significant. Previously, the controversy surrounding the national core technology designation for botulinum toxin focused on the issue of specific committee members serving long, consecutive terms. During last year's National Assembly audit, critics pointed out the expert committee's closed nature and its structural reliance on specific individuals. MOTIE's personnel overhaul aligns directly with this trend, implying that the newly appointed expert committee members for this year have converged on the opinion of lifting the designation. The core issue is simple. The current discussion is not about whether to unlock a single technology. It is about whether the outdated regulatory framework can be normalized.Meanwhile, the domestic botulinum toxin industry has long argued that the National Core Technology designation constitutes excessive, duplicate regulation. Their view is that because management systems are already functioning under the Pharmaceutical Affairs Act, the Infectious Disease Control and Prevention Act, and the Foreign Trade Act, layering National Core Technology regulations on top has only inflated administrative burdens.Critics have pointed out that South Korea is the only country to designate botulinum toxin as a National Core Technology, even though it is recognized globally as a generic manufacturing technology. Furthermore, the strains are publicly available on global genetic information networks, and a significant number of domestic companies utilize overseas strains. Despite this, additional reviews were repeatedly required in South Korea for every export, technical cooperation, and licensing process. Ultimately, it was a structure where administration outpaced industrial development.The problem lies in the outcome. The global botulinum toxin market has reorganized around American and European companies. Even though domestic companies possess manufacturing competitiveness, they have found themselves tied down by various administrative procedures. Uncertainty has also repeatedly plagued overseas market expansion and technology transfer negotiations.The National Core Technology system itself cannot be discredited. However, regulations must keep pace with reality. If the restrictive effects on industry outweigh the actual benefits of protection, the policy must be amended.Above all, these discussions must not end in another round of dragging time. In the past, whenever the possibility of declassification was raised, postponing a conclusion was a recurring pattern. In the meantime, market uncertainty only intensified.Now, the situation is different. A shift in the committee's internal current has been detected, and MOTIE has signaled a personnel overhaul. Now, only the final judgment remains.If the shackles that have bound K-toxin for 16 years cannot be broken this time either, the market will inevitably ask once again. For whose benefit is the South Korean biotech industry being regulated?The ball is now in the Industrial Technology Protection Committee's court. Conclusion awaits.
- Company
- Blincyto enters pricing negotiations as consolidation therapy
- by Eo, Yun-Ho May 19, 2026 11:08am
- The blood cancer treatment ‘Blincyto’ has entered the final stage of the process to expand its health insurance coverage in Korea.According to industry sources, Amgen Korea recently began drug price negotiations with the National Health Insurance Service regarding reimbursement for Blincyto (blinatumomab) as consolidation therapy for precursor B-cell acute lymphoblastic leukemia (ALL).The indication, which received expanded approval in Korea in February 2025, previously passed the Health Insurance Review and Assessment Service’s Drug Reimbursement Evaluation Committee in April.Patients with Philadelphia chromosome-negative (Ph-) precursor B-cell ALL frequently experience relapse even after achieving minimal residual disease (MRD)-negative status through conventional chemotherapy-based induction therapy, and continue to face challenges with long-term survival even after hematopoietic stem cell transplantation, indicating a significant unmet medical need.Blincyto’s consolidation therapy indication demonstrated efficacy through the E1910, AALL1731, AALL1331, and 20120215 studies.In the E1910 study, which compared chemotherapy alone versus alternating Blincyto and chemotherapy as post-induction consolidation therapy in adult precursor B-cell ALL patients, the 3-year overall survival (OS) rate among MRD-negative patients was 85% in the Blincyto-plus-chemotherapy alternating group, compared to 68% in the chemotherapy-alone group.Compared with chemotherapy alone, the Blincyto-plus-chemotherapy alternating group showed a 59% reduction in risk of death over a median follow-up period of 43 months.In addition, the 3-year recurrence-free survival (RFS) rate was 80% in the Blincyto-plus-chemotherapy alternating group versus 64% in the chemotherapy-alone group, representing a 47% reduction in the risk of recurrence or death over a median follow-up of 43 months.Furthermore, results from the AALL1731 study involving MRD-negative pediatric precursor B-cell ALL patients in the National Cancer Institute (NCI) standard-risk (SR) category at average or high risk of relapse showed that the estimated 3-year disease-free survival rate at a median follow-up of 2.5 years was 96.0% in the Blincyto-plus-chemotherapy alternating group, s a significant improvement compared to 87.9% in the chemotherapy-alone group.Meanwhile, the ‘2024 National Comprehensive Cancer Network (NCCN) Guidelines’ recommend a regimen that includes Blincyto as first-line consolidation therapy.