- LOGIN
- MemberShip
- 2026-06-06 10:09:36
- Policy
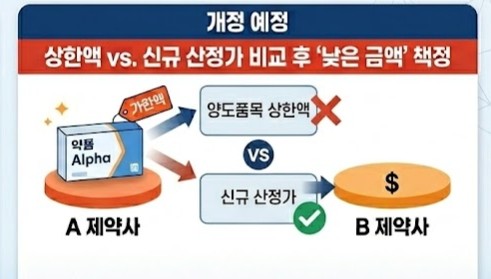
- Inheritance of drug price ceiling to be blocked in transfers
- by Jung, Heung-Jun May 08, 2026 01:15pm
- Changes are expected to the current rules that allow drug price ceilings to be inherited during product transfers. The system is likely to revert to past regulations that apply the lower of the new calculated price and the price at the time of transfer.As a result, acquiring high-priced generics to avoid the impact of drug pricing reform will no longer be viable.According to industry sources on the 6th, a working-level consultative body comprising the Ministry of Health and Welfare and industry representatives recently discussed changes to the transfer regulations that were not brought before the Health Insurance Policy Deliberation Committee in March.The rules governing price ceilings in transfers have changed multiple times. Until 2020, prices were set based on the lower of the final ceiling price of the transferred product and the newly calculated price.However, following the stepwise pricing reform, industry backlash arose as transfers were treated as new listings, leading to significant price drops.In response, the government allowed inheritance of the existing ceiling price starting during transfers in 2021.Now, the government plans to revert to the previous rule, applying the lower of the existing ceiling and the new calculated price.Even if a high-priced generic is acquired, the newly calculated price at the moment of transfer is applied, effectively enforcing a lowest price rule.Pharmaceutical companies that had considered acquiring high-priced generics to avoid pricing reform will lose this option.With the reform, the generic pricing rate has been reduced to 45%, and the reduction rate for products failing to meet the baseline requirements has been strengthened from 85% to 85%. Combined with multi-listing management rules, prices could fall to as low as 30.6% within one year if bioequivalence studies are not conducted.Previously, companies might have chosen to acquire products with higher maintained prices.However, if prices are reset based on the calculation price at the time of transfer, transfers that take existing drug prices into account become meaningless.
- Company
- Hunter syndrome drug Hunterase ICV approved in Peru
- by Lee, Seok-Jun May 08, 2026 01:15pm
- GC Biopharma announced on the 6th that its intracerebroventricular (ICV) Hunter syndrome treatment, ‘Hunterase ICV’, has received marketing approval from Peru’s regulatory authority, General Directorate of Medicines, Supplies and Drugs (DIGEMID).This marks the third overseas approval following Japan and Russia. The company plans to expand into additional countries using its Latin America entry as a foothold.Hunter syndrome is a rare inherited disease caused by a deficiency of the IDS (Iduronate-2-sulfatase) enzyme, leading to the accumulation of glycosaminoglycans (GAGs). It presents with skeletal abnormalities, joint deformities, respiratory and cardiac dysfunction, and cognitive impairment. It is known to occur in approximately 1 in every 100,000 to 150,000 male infants.About two-thirds of patients develop severe forms involving central nervous system damage. As the disease progresses, cognitive decline and behavioral abnormalities emerge, affecting patients’ quality of life and prognosis.Hunterase ICV is administered directly into the brain ventricles once a month. In Japanese clinical trials, it significantly reduced heparan sulfate, a key factor in CNS damage.The company also reported stabilization or improvement in cognitive and developmental functions, with sustained efficacy confirmed in long-term follow-up.Jae-woo Lee, head of R&D at GC Biopharma, said, “Based on long-term clinical data, we will focus on addressing the unmet medical needs of patients with severe Hunter syndrome.”
- Company
- SK bioscience's successful M&A
- by Chon, Seung-Hyun May 08, 2026 01:15pm
- The sales share of SK bioscience’s German contract development and manufacturing organization (CDMO) subsidiary has reached nearly 80%. By deploying funds secured during the COVID-19 pandemic into mergers and acquisitions (M&A), the company has strengthened its financial position and improved its defense against market pressures.According to SK bioscience, on the 7th, IDT Biologika’s first-quarter sales reached KRW 128.3 billion, an 8.5% increase from the same period last year.IDT Biologika is a German biotech company acquired by SK bioscience in 2024. SK bioscience purchased a 60% stake in IDT Biologika, previously held by the German biopharmaceutical firm Klocke Gruppe, through a wholly owned German subsidiary. The total acquisition price for IDT Biologika amounted to KRW 370.0 billion. SK bioscience decided to allocate paid-in capital to a third party, issuing 1,519,543 new shares worth KRW 75.7 billion to the Klocke Gruppe. Consequently, the total cash invested by SK bioscience in the acquisition of IDT Biologika is KRW 294.3 billion.Sales of SK bioscience and IDT Biologika (unit: KRW 100 million, source: SK bioscience) BLUE: SK bioscience, GREEN: IDT BiologikaFounded in 1921, IDT Biologika is a major biotech company operating CDMO businesses in Germany and the United States. It has a track record recognized by more than 10 core drug regulatory agencies, including those in the U.S. and Europe. The company develops processes and analytical methods while producing drug substances (DS) and drug products (DP) across the entire vaccine and biotech spectrum from clinical to commercial stages, employing approximately 1,800 people.IDT Biologika’s performance began to be reflected in SK bioscience’s consolidated financial statements starting from the fourth quarter of 2024. Revenue from IDT Biologika of KRW 111.2 billion was recognized in Q4 2024, and last year it recorded revenue of KRW 465.7 billion.With the inclusion of IDT Biologika’s results, SK bioscience’s revenue surged. In the first quarter of this year, SK bioscience’s revenue expanded more than sevenfold compared to the first quarter of 2024.SK bioscience faced significant fluctuations in performance throughout the COVID-19 pandemic.In 2020, SK bioscience signed a contract manufacturing agreement with AstraZeneca to produce and supply COVID-19 vaccine drug substances and finished products. In the same year, SK bioscience signed a tripartite agreement with the Ministry of Health and Welfare and Novavax to supply the COVID-19 vaccine 'NVX-CoV2373,' beginning full-scale contract manufacturing and supply in 2021.SK bioscience’s sales in the first quarter of 2020 was only KRW 22.7 billion. Then, it jumped nearly fivefold over the year to KRW 112.7 billion in the first quarter of 2021, and then rose to KRW 450.9 billion in the fourth quarter of 2021.However, as the contract manufacturing performance for COVID-19 vaccines dissipated, sales dropped. In the first quarter of 2023, SK bioscience’s sales amounted to KRW 20.6 billion, a 95.4% reduction compared to the fourth quarter of 2021.While sales fell sharply after the end of the pandemic, the M&A strategy successfully minimized the sales gap. In the first quarter of this year, IDT Biologika accounted for 76.1% of SK bioscience’s total sales. IDT Biologika’s sales were more than three times higher than SK bioscience’s separate sales of KRW 40.3 billion.From the fourth quarter of 2024 to the first quarter of this year, IDT Biologika’s cumulative sales reached KRW 705.2 billion, accounting for 72.2% of the parent company's sales.
- Policy
- Innovative pharmas need permanent pricing incentives
- by Lee, Jeong-Hwan May 08, 2026 01:15pm
- Rep. In-soon Nam of the Democratic Party of Korea has drawn attention by stating that permanent pricing incentives for innovative pharmaceutical companies should be institutionalized in order to advance Korea into a pharmaceutical and biotech powerhouse and create blockbuster new drugs.Rep. Nam criticized the Ministry of Health and Welfare, noting that while the ministry recently formalized a differential drug pricing system between innovative and non-innovative pharmaceutical companies through a resolution by the Health Insurance Policy Deliberation Committee, the inclusion of preferential pricing for innovative pharmaceutical companies only as a temporary provision left some gaps.Regarding legislation to introduce limited international nonproprietary name (INN) prescribing, Rep. Nam also expressed the view that it would be reasonable to either remove penalty provisions for doctors who fail to comply with mandatory INN prescribing regulations or to downgrade and relax these penalties to administrative fines.Her point is that policy and legislation should be pursued with a focus on ensuring that the public can access frequently out-of-stock and essential medicines without barriers, setting aside conflicts between specific professions such as doctors and pharmacists.Having officially announced her candidacy for Deputy Speaker of the second half of the 22nd National Assembly, Rep. Nam In-soon promised that, if elected, she would strive to create a national environment that advances the pharmaceutical and biotech industry and enhances public access to essential medicines.On the 7th, Rep. Nam held a meeting with the press corps and stated, “As the Jae-myung Lee administration has adopted a national agenda to transform Korea into a leader in digital health and the pharma-biotech industry, I will consider various ways to support this goal as Deputy Speaker.”“Pharma-biotech lost priority under Yoon administration… will restore momentum”Rep Nam assessed that while fostering the pharma-biotech industry has always been a priority across administrations, interest declined under the former Yoon, weakening growth momentum.In contrast, she noted that the Lee administration has set a goal of becoming one of the world’s top 5 pharma powerhouses and developing domestic blockbuster drugs, raising expectations for policy reforms across related ministries, including the Ministry of Health and Welfare.She therefore argued for the need to permanently extend preferential drug pricing for innovative pharmaceutical companies with the goal of fostering blockbuster new drugs.Rep Nam stated, “I have consistently advocated for differential pricing between innovative and general pharmaceutical companies, and I welcome its inclusion in the recent reform. However, making the incentive temporary limits its impact on new drug R&D investment.”“The government’s goal is to improve the domestic pharmaceutical industry’s structure using the funds secured through drug price cuts. If we want innovative new drugs and blockbusters to emerge in Korea, preferential treatment for innovative pharmaceutical companies cannot be temporary. Instead of offering temporary preferential treatment to innovative companies, we must remove the ‘temporary’ qualifier to allow for active support.”Rep Nam also noted that the Health and Welfare Minister Eun-Kyeong Jeong has shown a strong commitment to fostering domestic new drug development.She said, “The Lee administration has declared its intention to nurture blockbuster drug companies and has merged the Bio Committee and Bio-Health Innovation Committee. Previously, the Ministry of Health and Welfare did not appear to take a leading role, but after speaking with Minister Jeong Eun-kyung following the integration of the innovation committees, it appears the Ministry intends to take the initiative and carry out its duties. The atmosphere will change going forward.”He noted, “Currently, South Korea lags behind advanced countries in terms of pharmaceutical technology, new drug development, and medical AI, so we must catch up quickly. Since I enacted the Special Act on the Promotion of the Pharmaceutical Industry, preferential measures have been stalled for several years. If elected Deputy Speaker, I will prioritize this area in my legislative work.”“Let’s carry out limited INN prescribing focused on patients, without provisions punishing doctors”Regarding the limited INN prescribing bill currently pending in the National Assembly’s Health and Welfare Committee, Rep Nam expressed the view that even if provisions punishing doctors who violate the mandatory generic prescribing requirement are removed, a legislative environment must be created that ensures patients can access essential medicines without inconvenience.She emphasized that, since limited INN prescribing is a key policy agenda item of the current administration, efforts should focus on addressing resistance from the medical community and ensuring smooth institutional adoption.The current bill mandates INN prescribing for selected drugs designated by the Ministry of Health and Welfare following a government committee review, and stipulates penalties of up to one year in prison or a fine of up to KRW 10 million for violations.Rep Nam clearly stated that these penalty provisions should either be removed entirely or downgraded to administrative fines.She also noted the need to carefully consider concerns raised by the Korean Medical Association regarding the scope of bioequivalence testing and trust in substitution and INN prescribing.Rep Nam said, “The KMA is opposing the measure, raising concerns about the broad scope of bioequivalence testing, and their arguments should be taken seriously. We need further discussion on how to incorporate these concerns into policy. For now, it seems there is a lack of objective data or research on generic substitution and INN prescribing, so we need to accumulate more data.”She added, “Gradual implementation of INN prescribing for out-of-stock and essential medicines could serve as a middle ground between physicians and pharmacists. “Public awareness of generic substitution and INN prescribing has significantly increased during the COVID-19 pandemic, as more people have experienced receiving alternative drugs with the same active ingredient.”Nam emphasized, “We should activate systems such as post-notification of substitution and build more research data to support future legislative discussions. But imposing criminal penalties for INN prescribing violations is inappropriate. A more reasonable approach would be to remove penalties or reduce them to administrative fines, prioritize patient-centered legislation, and consider enforcement measures in stages.”“Warehouse pharmacy regulations need to prevent misuse”The recent amendment to the Pharmaceutical Affairs Act, which strengthens regulations on warehouse-style pharmacy advertising, is also one of Nam’s achievements.She explained that the ultimate goal of the regulation is to prevent excessive consumption and misuse of medicines that could lead to adverse effects.The provision in the Pharmaceutical Affairs Act prohibits expressions that may encourage misuse and delegates authority to the Ministry of Health and Welfare to specify concrete and unique expressions that cannot be used in pharmacy names or promotional slogans through subordinate regulations, prioritizing public safety above all else.Rep Nam said, “We also brought forward the implementation date from six months to three months after the government’s promulgation, and added a supplementary provision allowing public health centers to provide administrative guidance to pharmacists opening new pharmacies before the law takes effect. I was the one who codified the provision for pharmacists to provide medication guidance within the Pharmaceutical Affairs Act. At first, pharmacists weren’t very fond of it, but today, medication guidance has become a crucial system and phrase for ensuring public medication safety.”She continued, “When I look at warehouse-style pharmacies, I'm sorry whether pharmacists can actually provide proper medication guidance to patients. We must prevent the public from perceiving medications as mere commodities. Even after my election as Deputy Speaker, I will engage in policy monitoring to curb the misuse of medications by the public by regulating warehouse-style pharmacies.”
- Company
- 'Super antibiotic' Fetroja enters the prescription lists of gen hospitals
- by Eo, Yun-Ho May 08, 2026 01:15pm
- The 'super antibiotic' Fetroja is entering the prescription lists of major general hospitals.According to industry sources, Fetroja (cefiderocol tosylate sulfate), Jeil Pharmaceutical’s treatment for multidrug-resistant Gram-negative bacterial infections, has passed the drug committees (DC) of tertiary general hospitals, including Samsung Medical Center and Seoul National University Hospital.Following its inclusion on the insurance reimbursement list last February, the drug is gradually expanding its prescribing area.Fetroja was approved in South Korea in February 2025 for the treatment of ▲complicated urinary tract infections, including pyelonephritis ▲hospital-acquired pneumonia, including ventilator-associated pneumonia.Developed by Shionogi, Fetroja is the world's first siderophore cephalosporin antibiotic. To overcome reduced efficacy caused by bacterial resistance mechanisms, it uses a unique mechanism of action: it binds to ferric iron. It is actively transported into the bacterial cell through the pathogen's own iron porin channels.Jeil Pharmaceutical secured the rights to develop and commercialize Fetroja in South Korea after signing an exclusive domestic supply agreement with Ping An-Shionogi in July 2022.Fetroja has demonstrated in vitro activity against various antimicrobial-resistant (AMR) pathogens, including Carbapenem-resistant Enterobacterales (CRE), Carbapenem-resistant Acinetobacter baumannii (CRAB), and Metallo-beta-lactamase (MBL)-producing Carbapenem-resistant Pseudomonas aeruginosa (CRPA).Meanwhile, Shionogi has a long history and expertise in developing treatments for infectious diseases. The company continues to conduct research and development, particularly in antibiotics, antivirals, and central nervous system (CNS) treatments, and maintains subsidiaries in Japan, the U.S., Europe, and China.Ping An-Shionogi was established as a joint venture between Japan's Shionogi and China's Ping An in 2020. Ping An-Shionogi obtained Asian rights to Fetroja. In December 2024, Shionogi acquired all of Ping An's shares, and Ping An was subsequently incorporated as a wholly owned subsidiary of Shionogi.
- Company
- Cochrane review on ineffectiveness of Alzheimer’s drug sparks backlash
- by Eo, Yun-Ho May 07, 2026 10:29am
- “Anti-amyloid Alzheimer’s drugs show no clinically meaningful effect.”The backlash is as fierce as the message itself. Last April, the Cochrane Database published the results of a systematic review and meta-analysis covering anti-amyloid targeted therapies.The study concluded that these drugs, as a whole, failed to demonstrate clinically meaningful benefits in inhibiting cognitive and functional decline and raised significant safety concerns, such as amyloid-related imaging abnormalities (ARIA).Cochrane is a nonprofit health research organization with over 11,000 experts from more than 190 countries worldwide, providing evidence to inform healthcare decision-making. Such an announcement in a prestigious academic journal naturally had a significant impact.However, before assessing the value and validity of the study, the academic community has come forward to directly refute the results of the Cochrane meta-analysis.It is true that the conclusions of a meta-analysis can vary completely depending on “how the data is pooled.”Medical professionals argue that the core of the controversy surrounding this review lies in the fatal “methodological limitations” inherent in the study design.Because the study produced results that fundamentally overturn the current landscape of clinical practice, it is also true that any misinterpretation could cause significant turmoil for patients with early-stage Alzheimer’s disease, whether they are about to begin treatment or are already undergoing it, as well as their families.Integration of drugs with different development stages and regulatory statusThe main criticism is this: the review analyzed in a single pool four drugs that failed to meet clinical endpoints (e.g., ‘bapineuzumab’), ‘aducanumab (withdrawn due to safety concerns),’ and currently approved ‘lecanemab’ and ‘donanemab.’The argument is that grouping drugs with fundamentally different levels of development maturity, regulatory status, and mechanisms of action with equal weighting is scientifically inappropriate.Furthermore, the review is criticized for indiscriminately combining exploratory early-stage clinical trials (Phase 2) with large-scale confirmatory trials (Phase 3). Small-scale Phase 2 data with low statistical power were treated on par with Phase 3 results involving thousands of participants, leading to a severe distortion of the overall effect estimate. As a result, positive signals from drugs like lecanemab or donanemab, whose efficacy was clearly demonstrated in individual clinical trials, were diluted by the vast amount of noise from drugs that had failed in the past.Ji-won Seo, planning secretary of the Korean Dementia Association (Dongguk University Ilsan Hospital), stated, “If drugs at different development stages and regulatory statuses are combined, results will inevitably be diluted by the majority of data from failed drugs. It is not scientific to group successful drugs, failed candidates, and even those whose approvals have been revoked into the same basket and conclude that ‘the efficacy of this class is unclear.’”Regulatory approvals are based on independent dataGlobal regulatory authorities worldwide evaluate new drugs based on the specific clinical data unique to each drug, rather than on a generalized impression of the entire class.For example, lecanemab has been approved in over 50 countries worldwide, including the US FDA (July 2023), Japan PMDA (September 2023), China NMPA (January 2024), Korea MFDS (May 2024), UK MHRA (August 2024), and the European Commission (April 2025).Donanemab has also received sequential marketing authorization in over 40 major countries worldwide, including the US (July 2024), Japan (September 2024), China (December 2024), and Europe (September 2025).This global wave of approvals was made possible by thorough and independent reviews by regulatory agencies in each country, which evaluated the clinical evidence supporting these drugs’ ability to inhibit cognitive and functional decline, as well as the manageability of side effects such as ARIA. This means that, unlike the results of the Cochrane Review’s overly broad meta-analysis, their actual clinical value has already been validated worldwide.The Cochrane review cited ARIA as a major safety concern. ARIA is divided into cerebral edema (ARIA-E) and microbleeds (ARIA-H). While these are side effects that require careful management in the ATT class, the majority are asymptomatic or mild and can be adequately managed through regular MRI monitoring.These safety profiles have already been thoroughly reviewed during regulatory approval processes and are reflected in labeling and prescribing information.Alzheimer’s disease is ultimately a fatal, progressive neurodegenerative disorder. Now that evidence has been established that intervention during the early symptomatic stage can slow disease progression, unfounded misconceptions can lead to negative outcomes, causing patients to miss the “golden window” for treatment.Seo added, “With new treatments emerging in a field that previously had limited options, expectations for early diagnosis and treatment are increasing. However, following recent reports of research findings that were misinterpreted, many patients are expressing anxiety in the clinic or experiencing confusion when deciding on a treatment plan.”Seo emphasized, “We must remember that each drug has been approved by regulatory authorities based on clear scientific evidence. If the optimal window for treatment is missed due to misunderstandings, it could later result in far greater burdens for patients and their families.”
- Company
- "Data analysis of 500,000 patients…clinical utility of choline alfoserate reconfirmed"
- by Kim, Jin-Gu May 07, 2026 10:29am
- 'Choline alfoserate expert forum' recently hosted by DailyPharmA large-scale real-world data (RWD) analysis of 500,000 Korean patients with mild cognitive impairment (MCI) has reconfirmed the clinical utility of choline alfoserate in reducing the risk of progression to dementia.Experts evaluate that this study significantly strengthens the evidence base for prescribing choline alfoserate. It is considered academically meaningful because it revalidated the efficacy evidence from existing randomized controlled trials (RCTs) using massive datasets from actual clinical settings.Analysis of 500,000 Korean MCI patients...“Reduction in risks for both Alzheimer’s disease‧vascular dementia”At the 'Choline alfoserate expert forum' recently hosted by DailyPharm, Professors Han-kyeol Kim (Wonju Severance Christian Hospital), Choi Hojin (Hanyang University Guri Hospital), Lee Chan Nyung (Korea University Anam Hospital), and Kim Geon Ha (Ewha Womans University Mokdong Hospital), all neurologists, focused on the results of the study titled 'Effect of Choline Alfoserate: NHIS Cohort Study,' published last year.Professor Han-kyeol Kim (Wonju Severance Christian Hospital)Professor Han-kyeol Kim, who delivered the keynote presentation, introduced the research findings based on the National Health Insurance Service (NHIS) big data, which followed 508,107 patients newly diagnosed with MCI between 2013 and 2016.The study results showed that the choline alfoserate user group had a significantly lower risk of progression to Alzheimer’s dementia by 10.1% (HR 0.899) and vascular dementia by 16.8% (HR 0.832) compared to the non-user group. Furthermore, the risk of ischemic stroke decreased by 16.7% (HR 0.833), and the risk of hemorrhagic stroke decreased by 15.3% (HR 0.847).Professor Han-kyeol Kim said, "This was not a simple statistics of prescriptions. We linked health examination data to precisely adjust for variables such as smoking, alcohol consumption, income, and chronic diseases (hypertension, diabetes, dyslipidemia), as well as the duration of drug exposure to enhance objectivity," and added, "The study confirms that the use of choline alfoserate can be effective in reducing the risk of dementia progression and stroke in MCI patients, serving as a useful option for early intervention."Combining the scientific achievement of RCTs with the confirmed RWD… “Completing complementary evidence for dementia suppression”A discussion continued, chaired by Professor Choi Hojin, with Professors Lee Chan Nyung and Kim Geon Ha in attendance. They noted that this RWD study addresses the structural limitations of previous RCTs.The previous 'ASCOMALVA' study was designed as a multicenter, randomized, double-blind, controlled trial (RCT). The study targeted 210 patients with Alzheimer’s disease dementia accompanied by ischemic stroke, comparing a donepezil monotherapy group with a choline alfoserate + donepezil combination therapy group. The results confirmed significant improvements in the combination group across ▲the Mini-Mental State Examination (MMSE) ▲ Alzheimer's Disease Assessment Scale-Cognitive Subscale (ADAS-cog) ▲Instrumental Activities of Daily Living (IADL). Long-term follow-up over four years also showed that the rate of cognitive decline was inhibited in the combination therapy group.Experts agreed that this RWD study supplements the limitations of the RCT. Professor Choi Hojin evaluated, "The evidence for the efficacy of choline alfoserate has become even more robust by adding the analysis results of a large-scale Korean patient population to the ASCOMALVA findings," and added, "Results tracking the effects of drugs prescribed in the real world over a long period are highly meaningful as evidence from actual clinical practice, distinct from RCTs conducted in controlled environments."Professors Choi Hojin (Hanyang University Guri Hospital) ·Lee Chan Nyung (Korea University Anam Hospital) ·Kim Geon Ha (Ewha Womans University Mokdong Hospital)Professor Lee Chan Nyung stated, "While RCTs are suitable for proving drug efficacy in standardized environments, it is not easy to confirm actual conversion rates in slowly progressing diseases like dementia due to limitations in sample size and follow-up duration," and "This RWD study serves as powerful complementary evidence to existing RCTs by reaffirming the outcome of 'inhibiting dementia progression' through data accumulated over several years in actual clinical settings."“MFDS policy to enhance RWD reliability…increasing potential for utilization in clinical practice”Experts also emphasized that this study aligns with the recent government stance on the importance of RWD. In this regard, the Ministry of Food and Drug Safety (MFDS) announced the 'Guidelines for the Utilization of RWD/RWE in the Approval and Licensing of Medicines, etc.' in June 2024. Through this, the ministry presented specific criteria for actively recognizing RWD as clinical evidence for indication expansion or efficacy validation. In contrast, it had previously been used only as a supplementary tool for post-marketing surveillance (PMS) of side effects.The view among experts is that RWD research, which precisely analyzes NHIS big data from over 500,000 citizens, can yield new clinical evidence amid these changing regulatory environments. Professor Lee Chan Nyung emphasized, "As the vast RWD figures of 500,000 show, the dementia suppression effect confirmed in a large-scale patient group will serve as crucial evidence that gives clinical specialists the confidence to prescribe."Professor Kim Geon Ha said, "The 16.8% lower risk of progression to vascular dementia shown in this study holds great significance for the Korean elderly population with high vascular risk factors such as hypertension," and Kim concluded, "It raises the level of evidence for preemptive treatment of MCI patients in actual clinical settings."Professor Choi Hojin highlighted the public health significance of early intervention at the MCI stage. Choi added, "Delaying the onset of dementia by even just a few years can drastically reduce national medical costs and the caregiving burden on patients' families. Preemptive response through proven options will be a practical solution to alleviate the dementia burden in an aging society."
- Policy
- MFDS to revise labeling for 951 metformin products
- by Lee, Tak-Sun May 07, 2026 10:29am
- AI-generated imageThe government is pushing to revise the labeling for 951 metformin-containing products, the most widely used diabetes treatment in the market. This measure aims to explicitly state the potentially fatal side effects that may occur when metformin-containing products are administered to patients with hereditary mitochondrial disorders.According to industry sources on the 5th, the Ministry of Food and Drug Safety (Division of Drug Safety Evaluation) recently prepared a proposal to amend the approval conditions based on the European Medicines Agency (EMA)’s safety review regarding metformin-containing products and has begun collecting industry feedback.The key change is to restrict use and require immediate discontinuation in patients suspected or diagnosed with mitochondrial disorders.Under the proposed revision, metformin is not recommended for patients with ▲ MELAS syndrome (mitochondrial encephalopathy with lactic acidosis and stroke-like episodes) and ▲ MIDD (maternally inherited diabetes and deafness), due to increased risks of lactic acidosis and neurological complications.Additionally, if symptoms suggestive of MELAS or MIDD occur after administration, treatment should be immediately discontinued, and prompt diagnostic evaluation should be performed. The analysis indicates that metformin can affect intracellular mitochondrial metabolism, potentially causing severe energy metabolism disorders in patients with underlying genetic diseasesThe revision applies to a total of 951 products, including both monotherapy and combination drugs. As metformin is a core component of the domestic diabetes treatment market, most major pharmaceutical companies, including Chong Kun Dang, Daewoong, Hanmi, and Yuhan, are expected to be affected.The MFDS will accept feedback from relevant associations and companies until May 15. It will then comprehensively review the submitted comments and issue a final order to amend the approved product information.An MFDS official stated, “This measure aims to enhance the safe use of medicines for patients in Korea based on the latest safety information from the EMA. We ask that relevant associations and their member companies submit their review comments within the deadline.”
- Company
- Growth hormone deficiency drug Sogroya gains reimb in KOR
- by Son, Hyung Min May 07, 2026 10:29am
- Novo Nordisk Korea (General Manager: Kasper Roseeuw Poulsen) announced that starting on the 1st of this month, its long-acting, once-weekly growth hormone deficiency treatment ‘Sogroya Prefilled Pen (somapacitan)’ has been granted reimbursement for patients with growth hormone deficiency.Under the new reimbursement criteria, pediatric patients with growth hormone deficiency are eligible for Sogroya if they meet the following conditions: ▲ height below the 3rd percentile for chronological age, ▲ confirmed diagnosis through at least two growth hormone stimulation tests, ▲ and delayed bone age relative to chronological age.The recommended dosage is 0.16 mg/kg per week. Treatment is administered from a chronological age of 3 years until epiphyseal closure. However, reimbursement is limited to patients whose bone age falls within the range of 14–15 years for females and 15–16 years for males. However, patients within this category whose current height exceeds 153 cm (female) or 165 cm (male) must bear the full cost. In addition to children, Sogroya is covered for adult patients with growth hormone deficiency, provided certain criteria are met.Growth hormone deficiency is a condition characterized by delayed growth and may be accompanied by deficiencies in other pituitary hormones. Since consistent treatment is required until the end of the growth period, treatment adherence plays a critical role in treatment outcomes.The clinical efficacy and safety of Sogroya were confirmed in the global Phase III REAL4 trial.The REAL4 study was a randomized, parallel-group, open-label, active-controlled Phase III trial involving 200 treatment-naïve prepubertal pediatric patients with growth hormone deficiency. It evaluated the efficacy and safety of once-weekly Sogroya compared to once-daily growth hormone therapy.The primary endpoint was annualized height velocity (HV; cm/year) at Week 52. Secondary endpoints included changes in height velocity SDS, height SDS, the ratio of bone age (BA) to chronological age (CA), and IGF-1 SDS from baseline to Week 52.In the REAL4 study, Sogroya demonstrated non-inferiority in annual height velocity compared to daily growth hormone. At Week 52, the annual height velocity was 11.2 cm/year in the Sogroya group and 11.7 cm/year in the daily growth hormone group, showing comparable results.In terms of safety, the two treatment groups showed generally similar profiles. Most adverse reactions were mild or moderate in severity, and injection site reactions were reported in 5.3% and 5.9% of the Sogroya and daily growth hormone groups, respectively.Kasper Roseeuw Poulsen, General Manager of Novo Nordisk Korea, stated, “The reimbursement of Sogroya marks an important turning point in improving treatment access for patients with growth hormone deficiency. We expect that the once-weekly dosing option will improve treatment adherence, reduce the burden on patients and caregivers, and contribute to treatment continuity and improved quality of life.”
- Company
- Thyroid eye disease medicine 'Tepezza' lands in Korea
- by Son, Hyung Min May 07, 2026 10:28am
- Targeted therapy for thyroid eye disease (TED) 'Tepezza'Changes in treatment strategies are expected as the first targeted therapy for thyroid eye disease (TED) enters the Korean market.With follow-up candidates still in development or facing clinical hurdles with certain mechanisms, the possibility has been suggested that the market will remain centered on Amgen's already-commercialized Tepezza (teprotumumab).According to industry sources on the 7th, Amgen Korea recently obtained marketing authorization from the Ministry of Food and Drug Safety (MFDS) for Tepezza. The approved indication is for the treatment of adult patients with moderate-to-severe TED.TED is a rare disease in which autoimmune reactions cause inflammation and swelling of the orbital tissues. It causes proptosis, double vision, eye pain, and vision loss. If the disease progresses to a severe stage, it can lead to permanent disfigurement or even the risk of blindness.Until now, treatment in Korea has centered on symptomatic therapies such as steroids, radiation therapy, and orbital decompression surgery. However, critics have consistently pointed out that these have limitations in restoring structural changes such as proptosis or double vision.Tepezza is a monoclonal antibody that targets the Insulin-like Growth Factor-1 Receptor (IGF-1R), a key pathogenic mechanism in TED. It differentiates itself from existing treatments by targeting the disease to suppress the inflammatory response and tissue expansion.In the global Phase 3 OPTIC study, the proptosis response rate (a reduction of 2 mm or more) at 24 weeks was 83% for the Tepezza group and 10% for the placebo group. The diplopia (double vision) improvement rate also recorded 68% and 29%, respectively.In the OPTIC-J study of Japanese patients, the proptosis response rate was 89% in the Tepezza group and 11% in the placebo group.Tepezza already obtained regulatory agency authorization in the U.S. in 2020 and is currently the only approved targeted therapy for TED.FcRn inhibitors face hurdles… Delayed follow-up competitionSubcutaneous (SC) formulation 'Vyvgart (efgartigimod)'The competitive landscape for TED-targeted therapies remains limited, even in the global market.In particular, new drug candidates in the FcRn inhibitor class, which targeted to enter the TED market by reducing autoantibodies, are facing successive clinical barriers.Inhibition of the neonatal Fc receptor (FcRn), the protection receptor for Immunoglobulin G (IgG), is an approach that blocks the recycling pathway of IgG antibodies, thereby lowering the levels of pathogenic antibodies. It is evaluated as a treatment strategy applicable to autoimmune diseases in general. However, it appears to be struggling in the field of TED, beyond myasthenia gravis.HanAll Biopharma’s global partner, Immunovant, recently announced that its FcRn inhibitor batoclimab (IMVT-1401) failed to meet its primary endpoint in a global Phase 3 trial for TED. It failed to secure statistical significance in improving the Proptosis Responder Rate.Belgium-based Argenx also discontinued its clinical research on the FcRn inhibitor 'Vyvgart (efgartigimod)' for TED. Development was halted as the Independent Data Monitoring Committee (IDMC) judged the possibility of meeting the primary endpoints to be low.Accordingly, expert analysis suggests that a simple approach of reducing autoantibodies alone may be difficult to achieve sufficient clinical effects in TED.Chase by Viridian and Roche…the competition continuesNew drug development has not stagnated entirely. Among the followers, the most advanced is U.S.-based Viridian Therapeutics. Viridian is developing 'elegrobart,' a candidate based on the same IGF-1R mechanism as Tepezza.Elegrobart recently confirmed improvements in proptosis and diplopia in a global Phase 3 trial for patients with chronic TED, demonstrating the fastest rate of improvement among follow-up candidates.Roche’s 'Enspryng' However, Amgen has also moved to defend its market, recently reporting positive results from a Phase 3 trial of a subcutaneous (SC) formulation of Tepezza using an on-body injector (OBI). Consequently, competition in the TED market appears to be shifting toward competing for both efficacy and convenience, rather than simply SC conversion.Competition based on other mechanisms is also continuing. Roche’s 'Enspryng (satralizumab)' is a treatment that blocks the Interleukin-6 (IL-6) receptor, which is involved in inflammatory responses.Enspryng differs from the IGF-1R class, which directly targets ocular tissue expansion, by targeting the disease by suppressing orbital tissue inflammation and immune responses in TED.Roche recently announced positive results from the global Phase 3 SatraGO program for TED and disclosed plans to apply for authorization within the year.Enspryng failed to achieve statistical significance for the primary endpoint in the global Phase 3 SatraGO-1 study in patients with active TED. However, a pooled analysis with the follow-up Phase 3 study, SatraGO-2, reportedly confirmed a proptosis improvement effect.Until now, the treatment with the clearest clinical efficacy in TED has been the IGF-1R-targeted therapy. As FcRn inhibitors face successive clinical struggles, follow-up competition is trending toward a reorganization around IL-6R inhibitors and next-generation IGF-1R agents.