- LOGIN
- MemberShip
- 2026-06-06 10:09:37
- Policy
- CSO 규제 향방은…복지부, 재위탁·수수료율 손질 가능성
- by Lee, Jeong-Hwan May 26, 2026 03:21pm
- [데일리팜=이정환 기자] 보건복지부가 의약품 판촉영업 대행사(CSO) 추가 규제를 통한 의약품 유통 구조 투명성 강화 필요성을에 공감하면서 이어질 정책 방향에 시선이 모인다.제약업계에서 회자되고 있는 규제는 CSO가 다른 CSO에게 의약품 영업판촉 업무를 재위탁하는 행위를 금지하거나 관련 규제 수위를 지금보다 높이는 방식이다.이와 함께 CSO가 제약사에게 요구하는 수수료율의 상한선을 규정하는 등 CSO 수수료에 대한 행정적·법적 제한을 신설하는 규제도 거론되고 있다.25일 제약업계는 복지부가 한국제약바이오협회와 함께 CSO 실태조사에 착수한 만큼 연내 구체적인 규제 방향성을 수립할 것으로 전망중이다.현재 CSO 업계는 제약바이오산업 육성을 위해 CSO도 산업으로 육성할 필요성을 강조하며 복지부를 향해 한국CSO협회의 사단법인 인가를 촉구하는 실정이다.반면 복지부와 국내외 제약사들은 CSO의 사단법인 인가는 아직 시기상조라는 입장이다. CSO 업계가 CSO협회를 조직해 자체적으로 산업 선진화 등 노력을 기울이고 있지만, 여전히 CSO를 활용한 불법이 정화되지 않는 등 사단법인으로서 지위를 인정해주기엔 역부족이라는 평가다.이에 복지부와 제약협회 실태조사 이후 추가 규제가 이뤄져야 논의 가능성이 향상할 것으로 보인다.일단 CSO를 악용한 불법 리베이트의 가장 실질적인 원인으로 꼽히는 '무제한 재위탁'이 규제 사정권으로 분류된다.제약사가 CSO에게 의약품 판촉영업 대행 위수탁 계약을 체결한 이후, 해당 CSO가 다른 CSO와 또 위수탁 계약을 체결하면서 불법 리베이트 진원지를 찾을 수 없는 문제를 해결할 필요가 있다는 게 복지부와 제약업계, 국회의 공감대다. CSO 위탁 계약 원천 금지 규제 가능성이 거론되는 이유다.다음으로는 제약사가 CSO에 지급하는 의약품 판촉영업 수수료 비중을 제한하는 수수료 상한제다. 높은 CSO 수수료는 결국 불법 의약품 리베이트 재원으로 쓰이게 된다는 비판을 법으로 규제하는 차원이다.다만 CSO 수수료 규제는 헌법에 위배될 소지가 있어 복지부와 정치권은 입법에 고심하는 표정이다. 그럼에도 CSO 수수료 제한이 실질적인 리베이트 근절과 투명한 의약품 유통질서 강화 효과가 기대될 경우 간접적으로 CSO 수수료 구조를 투명화하는 행정 규제와 입법이 추진될 공산이 크다.복지부는 일단 이번 실태조사에서 제약사와 CSO, CSO와 CSO 간 위수탁 계약, 재위탁 계약 현황 분석으로 규제 방향성을 수립한다는 방침이다.특히 복지부는 CSO 산업을 육성하는 게 복지부의 주된 업무가 아니라는 입장으로, 공정하고 투명한 의약품 유통질서 확립 즉, CSO 리베이트 근절을 위한 규제 신설을 고민할 것으로 보인다.제약업계 관계자는 "복지부는 CSO 수수료율 규제를 정부가 나서서 추진하는 것에 일부 부담감을 느끼는 동시에 위헌 가능성에 집중하는 분위기"라며 "법으로 CSO 수수료율을 옭아매는 게 무조건 좋은 방법인지 여부에 대한 판단도 안 선 것 같다. 다만 재위탁 문제가 심각해 불법 리베이트 규제 공백을 키우고 있는데 대한 문제의식엔 공감하는 것으로 안다"고 귀띔했다.그러면서 "CSO 수수료 상한제는 제약사들로서는 가장 직접적이고 효율적인 규제 방향일 수 있다. 하지만 법으로 당장 제한하기 어려울 수 있어 간접적으로 CSO가 과도한 수수료를 제약사에 요구하고 정당한 판촉이 아닌 리베이트 영업으로 품목 처방 매출을 유지하는 문제를 막는 방법을 고민할 필요가 있다"며 "CSO 업계 자체도 일단 자정 필요성에 공감은 하고 있는 것으로 안다. 문제는 너무 점 조직으로 구성돼 CSO 업계에 대한 일괄적 규제나 의견 수렴, 통합이 어렵다는 게 문제"라고 말했다.이어 "일단 불법 리베이트 근절에 앞장서고 있는 다수 제약사는 CSO 신고제 시행 2년차를 맞은 지금, 신고제를 넘어선 추가 규제가 필요하다는 의견"이라며 "복지부의 제네릭 약가인하 정책이 예고된 시점과 맞물려 추가 행정이 시급하다"고 덧붙였다.
- Opinion
- "The expected probability of a clinical cure for TNBC"…Keytruda
- by Son, Hyung Min May 22, 2026 10:25am
- Although triple-negative breast cancer (TNBC) accounts for approximately 15 to 20% of all breast cancer cases, it is recognized as a highly aggressive subtype with a significant risk of recurrence.Due to the lack of hormone receptors (HR) or HER2 expression, targeted therapies cannot be used, leaving patients with relatively limited treatment options. It has been classified as a high-risk breast cancer, particularly due to the high risk of recurrence and mortality within five years of diagnosis.TNBC carries a high risk of recurrence even in relatively early stages (Stages I and II), and recurrence is highly likely to manifest as distant metastasis. Notably, the T2N0 patient population, characterized by a tumor size of 2 to 5 cm and node-negative status, has recently regarded as a high-risk subgroup requiring aggressive therapeutic intervention.Amid this therapeutic landscape, analysis suggests that a perioperative treatment strategy based on the cancer immunotherapy 'Keytruda (pembrolizumab)' is leading a paradigm shift in the management of early-stage TNBC.Professor Hee Kyung Ahn, Department of Hematology-Oncology, Samsung Medical CenterKeytruda received regulatory approval from the Ministry of Food and Drug Safety (MFDS) in July 2022 for the perioperative treatment of high-risk early-stage TNBC.Based on the KEYNOTE-522 clinical study, Keytruda demonstrated significant improvements not only in pathological complete response (pCR) but also in event-free survival (EFS) and overall survival (OS). Consequently, it is drawing attention as a therapeutic strategy aimed at increasing the probability of a clinical cure rather than merely delaying disease recurrence.As a result, an integrated therapeutic approach encompassing neoadjuvant chemotherapy, definitive surgery, and subsequent adjuvant therapy is increasingly well established in real-world clinical practice.DailyPharm met with Professor Hee Kyung Ahn of the Department of Hematology-Oncology at Samsung Medical Center to discuss the shifting treatment paradigms in early-stage TNBC and the clinical implications of Keytruda-based perioperative therapy.Q. What are the changes in clinical practices since Keytruda was approved for early-stage TNBC?The therapeutic landscape has transformed significantly over the past few years, and Keytruda is now widely used in clinical settings.Today, we no longer proceed to surgery first, even for node-negative Stage IIA patients. Instead, neoadjuvant therapy with Keytruda is used, followed by a risk-based adjuvant regimen regardless of whether the patient achieves pCR post-surgery. Because robust clinical trial data have demonstrated survival benefits with this strategy, proactive clinical interest and adoption have risen substantially.Based on the KEYNOTE-522 clinical data, the pCR rate was 64.8% in the Keytruda arm compared to 51.2% in the control arm, representing an absolute improvement of approximately 13.6 percentage points. Furthermore, EFS improved by roughly 8 percentage points, and the overall survival rate improved by about 5 percentage points. Demonstrating these absolute benefit margins underscores a major breakthrough in therapeutic outcomes, making it a highly clinically meaningful finding.Q. Why is the T2N0 patient cohort frequently highlighted in discussions regarding TNBC?T2N0 defines a case where the tumor size spans 2 to 5 cm with node-negative status. Patients often feel reassured because it is classified as Stage IIA, a relatively early stage. However, even within the same clinical stage, TNBC carries a profoundly higher risk of recurrence compared to other subtypes, such as hormone receptor-positive breast cancer.While the stage of the cancer is important, TNBC carries the risk of a poor prognosis and aggressive biological phenotype that even Stage IIA patients are classified into a high-risk group with an elevated risk of recurrence. Consequently, formulating an aggressive treatment strategy is essentialQ. How do you evaluate the clinical significance of Keytruda demonstrating an OS benefit?Demonstrating an OS benefit in early-stage curative settings is incredibly difficult. It requires proving that a therapy extends actual patient survival, rather than merely delaying disease recurrence.Certain therapies may successfully prolong the time to recurrence, but once the disease recurs, the biological characteristics of the tumor can shift aggressively, preventing extended patient survival. In those situations, an OS benefit is absent.In contrast, the validation of an OS benefit implies that early-intervention treatment successfully translates into a potential cure for a substantial proportion of patients, or at the very least, prolongs overall life expectancy even if the disease eventually recurs.Because cancer directly threatens life, the absolute benchmark and ultimate objective of all oncology treatments is to extend survival as much as possible. Thus, improving overall survival holds significance, demonstrating that the therapy has successfully achieved the gold standard goal of oncological intervention.Q. What do you consider to be the most noteworthy findings from the subgroup analysis?The fact that consistent outcomes were demonstrated across nearly all subgroups is highly meaningful. An especially intriguing aspect is the divergence between metastatic TNBC and early-stage TNBC data.In the metastatic setting, Keytruda's efficacy was confirmed exclusively in PD-L1-positive patients. However, in early-stage TNBC, pCR and EFS improvements were achieved regardless of PD-L1 expression status.This means that early-stage TNBC may possess distinct immunological profiles compared to the metastatic stage, suggesting that Keytruda-based regimens function more effectively when deployed early in the disease stage.Q. Why is the integrated therapeutic approach of continuing perioperative adjuvant therapy so crucial in early-stage TNBC?Previously, if a patient achieved pCR following neoadjuvant chemotherapy, further post-operative systemic treatment was frequently omitted. However, the KEYNOTE-522 study design mandated that even patients achieving pCR continue receiving adjuvant Keytruda post-surgery. Driven by these data, current guidelines recommend maintaining therapy unless precluded by significant adverse events.Notably, highly meaningful outcomes were confirmed in the non-pCR cohort. While this subpopulation historically carries a dismal prognosis, the inclusion of Keytruda demonstrated a trend toward improved survival outcomes. This strongly suggests that maintaining adjuvant therapy plays a critical role in managing residual disease burdens and mitigating the risk of recurrence.Keytruda's ability to fundamentally transform treatment outcomes across the entire TNBC continuum (from early-stage to metastatic disease) is significant. The observable decline in recurring patients compared to the past directly translates to lives saved, which is why its clinical value is so deeply felt on the ground.Q. How do you view the current reimbursement landscape and the challenges ahead?Currently, Keytruda is not reimbursed for this indication; partial reimbursement is restricted only to the backbone cytotoxic chemotherapies used in the combination regimen. While this alleviates a fraction of the cost, the out-of-pocket financial burden on patients remains substantial.The crucial factor we must not overlook is the societal and economic value of a clinical cure. Achieving a cure goes beyond the mere eradication of cancer cells; it represents returning a patient fully to society. When a patient is cured, it eliminates the massive future medical expenditures and drug costs associated with recurrence, while preserving the economic and social productivity that would otherwise be lost.In South Korea, a stringent threshold demand high standard for OS data for early-stage indications due to fiscal constraints. However, capturing definitive OS data requires an extended timeline, and while waiting for those results, a significant number of patients miss their critical window for a cure.While managing metastatic disease is vital, I personally believe that curative-intent treatment in the early-stage setting should take precedence. This is particularly justifiable because therapies deployed in the neoadjuvant setting have a fixed number of treatment cycles, allowing total healthcare expenditures to be capped and predicted in advance.Therefore, as a realistic alternative, if budget impact is the primary hurdle delaying full reimbursement, health authorities should actively explore mechanisms to enhance patient access, such as implementing partial reimbursement structure or adjusting patient co-payment tiers to a realistically affordable level.Allowing regulatory and institutional flexibility by taking into account the manageable financial thresholds for individuals confronting critical illnesses will serve as a practical solution to improve market access and patient drug availability for cancer patients.
- Company
- 10th year of Boksan-Suzuken partnership…"Establishing a platform for mutual growth"
- by Kim, Jin-Gu May 22, 2026 10:25am
- Commemorating the 10th anniversary of their strategic partnership, Boksan Nice and Japan's Suzuken presented the 'redefinition of roles between pharmaceutical companies-wholesalers' and the 'transition to digital distribution platforms' as key strategies to overcome the crisis facing the pharmaceutical industry, which is currently pressured by drug price cuts and market restructuring.On the 21st, Boksan Nice and Suzuken hosted the 'Boksan Nice-Suzuken Partnership 10th Anniversary Seminar' at the Orchid Room of the Westin Josun Seoul. The event brought together executives from both companies and around 80 stakeholders from the pharmaceutical distribution industry.The event was organized to reflect on the decade-long partnership between the two companies and to outline specific future collaboration pathways for pharmaceutical companies and the wholesale sector to achieve mutual growth amid ongoing large-scale drug price reductions and structural inefficiencies in distribution in South Korea. The two companies established their relationship in 2016 when Suzuken made a KRW 52 billion investment in Boksan Nice.In his opening remarks, Boksan Nice Chairman Eom Tae-ung emphasized, "This seminar goes beyond a simple 10th-anniversary celebration; it is a venue to collectively address the rapid changes and future direction facing the pharmaceutical industry and distribution sector today." and added, "As the global weight of marketing and distribution continues to grow, Boksan Nice’s clear direction in this era of selection and concentration is to become a healthy and competitive partner for domestic pharmaceutical companies."Suzuken Chairman Hiromi Miyata introduced the background behind the long-standing trust between the two companies over the past decade. Chairman Miyata stated, "When we entered the Korean market 10 years ago, we conducted interviews and surveys targeting pharmaceutical companies, healthcare institutions, and pharmacies, and Boksan Nice's reputation and credibility were overwhelmingly high," and added, "This deep trust became the decisive factor for our collaboration. Over the last 10 years, we have built a foundation for growth through continuous joint projects and close talent exchange. We will continue to support the growth of Boksan Nice."Boksan Nice Chairman Eom Tae-ung (left) and Suzuken Chairman Hiromi Miyata.Boksan Nice and Suzuken jointly assessed that the pharma-biotech markets and pharmaceutical distribution environments in both Korea and Japan are undergoing profound shifts. In Korea's case, a heightened sense of crisis is escalating due to large-scale drug pricing restructuring.In response to this crisis, Japan's Suzuken proposed a "digital platform" strategy as an alternative solution. Shigeru Asano, Director and Chairman of Suzuken, shared collaboration case studies from the Japanese market, noting, "Amid changing market environments, wholesalers as well as pharmaceutical companies must move beyond simple logistics fulfillment to deliver new value-added functionalities."Chairman Asano identified "Visualization, Connection, and Change" as the core strategies of the Suzuken Group, explaining, "We are currently building a digital-based 'healthcare data platform' that organically links data from medical institutions, healthcare professionals, and the supply chain," and added, "Through this, we will perform an advanced role by supporting the digital marketing efforts of pharmaceutical companies while, in turn, providing a stable infrastructure for pharmaceutical supply."Chairman Eom Seung-wook of Boksan Nice identified fundamental inefficiencies in the domestic pharmaceutical distribution structure and proposed streamlining distribution channels as a solution.Eom diagnosed, "Currently, there are around 4,000 wholesalers in Korea, and the collective market share of the top 10 companies does not exceed 40%," and added, "Consequently, Korean wholesalers have remained in the role of simple delivery vendors until now, and under the pressure of large-scale drug price cuts, the legacy approach is unsustainable for both pharmaceutical companies and wholesalers."Eom proposed a structural division of business as a concrete model for collaboration. He emphasized, "Simplifying distribution channels is no longer an option but an absolute necessity. Roles must be redefined so that pharmaceutical companies can focus entirely on their core operations, such as R&D and manufacturing. At the same time, wholesalers solely handle distribution and operational efficiency," and concluded, "Boksan Nice will become a core partner that substantially contributes to improving the profitability of pharmaceutical companies hit by drug price cuts by eliminating unnecessary costs, expanding market accessibility, and simplifying workflows."
- Company
- HPV prevention expanded to male adolescents
- by Son, Hyung Min May 22, 2026 10:25am
- With the National Immunization Program (NIP) expanding to include male adolescents, Korea’s HPV (human papillomavirus) prevention strategy is shifting from a male-centered approach to a system that prevents HPV in both male and female individuals.Analysts emphasize the significance of this move, noting that while major countries abroad have been implementing simultaneous vaccination for both genders for several years as part of their strategies to reduce HPV-related cancers, Korea has finally established a foundation for vaccinating male adolescents, albeit belatedly.On the 20th, MSD Korea held a media session at Seongam Art Hall in Seoul under the theme “A New Standard for HPV National Immunization: Vaccination for Both Genders.”Starting this month, the Korea Disease Control and Prevention Agency (KDCA) has included 12-year-old male adolescents (born in 2014) in the National HPV Immunization Program. Previously, the program only covered female adolescents aged 12 to 17 and low-income women.This expansion is considered significant because it extends free HPV vaccination, which had been limited to females for the past decade, to include male adolescents. According to the presented materials, the first-dose HPV vaccination rate among male adolescents in Korea stood at just 0.2%. In contrast, rates were 77.7% in Australia, 71.2% in the UK, and 59.0% in the US.HPV is known to be a virus that can infect both men and women. According to the International Papillomavirus Society (IPVS), approximately 5% of all cancer cases worldwide are reported to be related to HPV. While most infections clear up naturally, persistent infection can lead to cervical cancer, vaginal cancer, anal cancer, vulvar cancer, and genital warts.Dong-hyun Kim, Professor of Pediatrics at Inha University HospitalDong-hyun Kim, a professor of pediatrics at Inha University Hospital, stated, “HPV is not just a cervical cancer issue. It is a virus that can also cause anal cancer and genital warts in men. Since it is an infectious disease that affects and spreads among both men and women, it is difficult to approach it with prevention strategies targeting only a specific gender.”According to data from the Korea Disease Control and Prevention Agency, reported HPV cases in Korea increased from 10,945 in 2020 to 14,534 in 2024. In particular, reported cases among men rose 82.9% during the same period, from 117 to 214 cases. A domestic study of men found that approximately 59% tested positive for HPV DNA.Professor Kim cited high reinfection risk and low natural clearance rates as characteristics of HPV infection in men.“HPV is a virus that can infect both men and women, and it is not an issue limited to a specific gender. We need to approach prevention strategies from the perspective of infectious disease, rather than gender.”He continued, “Men often carry the virus asymptomatically, and the rate of natural clearance is known to be slower than in women. This means transmission may continue repeatedly without the person even being aware of it.“Experts recommend 11–12 years of age as the optimal age for HPV vaccination. This is because vaccination before sexual debut ensures high immunogenicity, and the 9–14 age group can expect a sufficient immune response with fewer doses than adults.Professor Kim emphasized that HPV vaccination is not a gender-specific issue, but an infectious disease prevention strategy for both men and women.The quadrivalent HPV vaccine available through the HPV national immunization program is administered in two doses for those aged 9 to 13 (6–12 months apart)In particular, study results showed that two doses administered at ages 9 to 13, when immune responses are active, demonstrated non-inferior immunogenicity (geometric mean antibody titers, GMT) against HPV types 16 and 18 compared with three doses in women aged 16 to 26. This highlights the need to secure preventive effects through early vaccination.Professor Kim said, “When vaccinated before age 15, two doses alone show high immunogenicity compared with three doses in older age groups. It is also meaningful in terms of vaccination convenience because it can be administered at the same time with other vaccines included in the national immunization schedule, such as Tdap and Japanese encephalitis vaccines.”He continued, “Overseas, countries have already set the eradication of cervical cancer as a national goal based on simultaneous vaccination of both men and women. Korea also needs to begin serious discussions on shifting its HPV prevention strategy, starting with this expansion of the NIP to male adolescents.”
- InterView
- [Reporter’s View] Beyond Korea's drug pricing reform
- by Lee, Jeong-Hwan May 22, 2026 10:25am
- The government has finalized implementation of a drug pricing system reform plan centered on applying a 45% pricing rate for generics and granting pricing incentives to innovative pharmaceutical companies, quasi-innovative pharmaceutical companies, and companies contributing to the stable supply of essential medicines. However, the tasks needed to maximize the effects of the reform and minimize side effects remain unfinished.Fortunately, the ruling party and the government appear to have immediately begun work on preparing the “next-level” administrative and legislative specifics needed to create synergy with the drug pricing reform.The direction is clear: Korea must break away from and reform the abnormal multi-product generic structure in which hundreds of products are approved for a single ingredient, leading the market to be mired in distorted promotional competition—specifically, illegal rebate competition—rather than competition based on product quality.The specific policy goal of the ruling party and the government is to broadly gather opinions from the pharmaceutical industry, professional organizations, and academia to reduce or abolish the currently permitted 1+3 contract-based joint bioequivalence system for generics, while also overhauling the pharmaceutical industry structure that abuses contract sales organizations (CSOs) to distribute illegal rebates.This is a necessary policy shift for Korea’s pharmaceutical industry to move beyond domestic generic drug sales competition and evolve into a nation capable of developing blockbuster new drugs that dominate the global market.Jae-hyun Lee, head of the Korea Regulatory Affairs Professional Society and an expert in drug approval regulations and health policy, criticized the 1+3 joint bioequivalence system as “a deformed system that cannot be found anywhere else in the world and the main culprit behind the proliferation of generics in Korea,” calling on the government to seriously reflect and change course.Lee said, “The domestic regulatory system itself, which grants product approval through joint bioequivalence, makes no sense. Even a 1+1 system, which would allow one contract manufacturer to grant approval rights to only one consignment company, is unreasonable. Korea needs to shift to a single-generic per product policy by abolishing consignment bioequivalence testing and eliminating identical generics.”Won-jun Cho, Senior Policy Advisor for Health and Medical Policy for the Democratic Party of Korea and the Director of the Party’s Policy Committee, also assessed that the Ministry of Health and Welfare’s planned drug pricing reform can land smoothly without distortion only if if the government actively considers abolishing consignment bioequivalence testing and simultaneously imposes strict regulations on CSOs, which are used as a means for illegal rebates.Cho said, “To maximize the effects of the drug pricing reform for real pharmaceutical companies leading innovation, quasi-innovation, and stable supply, we must eliminate ‘free-riding’ pharmaceutical companies and paper companies that contribute little to the pharmaceutical industry or national development and are solely focused on rebate-driven promotion and sales from generics approved via consignment bioequivalence studies. Together with the government, we will also pursue regulations against pharmaceutical companies and problematic CSOs that damage the sound distribution order of medicines by colluding with certain medical institutions to provide indirect rebates through CSOs, as well as substandard CSOs.”In other words, academia, the government, and the ruling party share the view that unresolved issues long left unattended must now be actively addressed following the implementation of the drug pricing reform.Accordingly, the government (Ministry of Health and Welfare and the Ministry of Food and Drug Safety), academia, and the National Assembly led by the ruling party are expected to take immediate legislative and administrative measures regarding the drug approval system and CSO regulations in the near future.The reason Korea needs to review the reduction or abolition of the 1+3 joint bioequivalence system from multiple angles is that it directly conflicts with the philosophy of the newly revised drug pricing system. It is difficult to establish a logical basis for guaranteeing the same drug price to consignment generic companies that have no production infrastructure or research personnel and simply resell drugs made by others under a different brand name, using only sales networks. After multiple rounds of gathering opinions from the pharmaceutical industry, the government needs to assess the utility and limitations of the consignment bioequivalence system and make a reasonable administrative decision.Furthermore, the success of the drug pricing reform plan also requires eliminating the illegal CSO rebate structure, which serves as a distorted business practice for some generic drug companies. In a situation where the products themselves have virtually no competitive edge, some generic drug companies currently rely on distributing rebates to doctors to induce prescriptions, and this must be stopped. Regulatory standards must be tightened to ensure that CSOs are no longer used as scapegoats.It is time to consider introducing a dual-penalty system that clearly links illegal rebate liability between pharmaceutical companies and CSOs, while also strengthening penalties for doctors who receive rebates to a level sufficient to restore market order. This is because drug price cuts alone cannot completely block the flow of illicit funds into illegal business practices.The Ministry of Health and Welfare’s drug pricing reform is the first step toward innovation in the pharmaceutical industry. Unless the National Assembly enacts legislation to eliminate unreasonable systems and health authorities follow up with aggressive measures, there is a risk that this reform plan will remain just another unfinished policy. To ensure that the principles of “removing free riders” and “rewarding innovation” become firmly rooted in the market, the 22nd National Assembly and the government must act without delay.
- Company
- Reimbursement listing discussions for Vyloy in slow progress
- by Eo, Yun-Ho May 22, 2026 10:25am
- Reimbursement discussions for the gastric cancer-targeted anticancer therapy Vyloy remain sluggish. Therefore, attention is now on whether progress can be made within the first half of the year.According to Dailypharm coverage, Astellas Pharma Korea’s Claudin 18.2-positive gastric cancer targeted therapy Vyloy (zolbetuximab) is expected to be presented to the Pharmacoeconomic Evaluation Subcommittee of the Health Insurance Review and Assessment Service today, on the 21st.Vyloy passed the Cancer Drug Review Committee last October. This means more than 6 months have passed before its submission to the Pharmacoeconomic Evaluation Subcommittee.Vyloy, which was approved in Korea in September 2024, failed to pass the Cancer Drug Review Committee on its first attempt in February last year. However, the company immediately resubmitted the application and succeeded in obtaining a positive result. However, as subsequent procedures are proceeding slowly, it is expected to take even longer to reach a final listing decision.Vyloy is the world’s first approved Claudin 18.2–targeted therapy. It is a monoclonal immunoglobulin antibody that binds to Claudin 18.2, a protein expressed and exposed on gastric cells.According to the Phase III SPOTLIGHT study, which formed the basis for Vyloy's approval, the median progression-free survival (mPFS) for Vyloy combined with mFOLFOX6 (oxaliplatin, leucovorin, fluorouracil) was 10.61 months, exceeding the placebo group’s 8.67 months. Median overall survival (mOS) was also longer at 18.23 months versus 15.54 months.In the GLOW study, the Vyloy plus CAPOX (capecitabine and oxaliplatin) combination group achieved a median progression-free survival of 8.21 months, reducing the risk of disease progression or death by approximately 31%.Professor Sun Young Rha of Yonsei Cancer Center stated, “About 90% of patients with metastatic gastric cancer are HER2-negative, underscoring the urgent need for therapies targeting new biomarkers. Given that about 40% of HER2-negative patients are reported to be Claudin 18.2-positive, the emergence of Vyloy, which selectively binds to Claudin 18.2, presents a new therapeutic option for these patients.”Meanwhile, the Korean Gastric Cancer Association revised its treatment guidelines on January 6, 2025, in the Journal of Gastric Cancer (JGC), granting Vyloy the ‘highest-level’ recommendation as first-line therapy for patients who are HER2-negative and Claudin 18.2-positive.Vyloy has also been listed as a standard treatment option in Japan’s gastric cancer guidelines and the European Society for Medical Oncology (ESMO) clinical practice guidelines. It is included as a preferred regimen in the U.S. NCCN guidelines, rapidly establishing itself as a global standard of care for gastric cancer.
- Company
- Yuhan sets record-breaking deals and exports
- by Chon, Seung-Hyun May 21, 2026 10:16am
- Yuhan Corp’s active pharmaceutical ingredient (API) export business is continuing its strong momentum. The company secured its largest-ever single contract worth over KRW 200 billion. Through supply agreements signed since last year, Yuhan has already secured API export sales approaching KRW 300 billion for this year. As multinational pharmaceutical companies increasingly recognize the quality of Yuhan’s APIs, the company has continued landing major contracts, more than doubling overseas sales compared to five years ago. Yuhan Chemical, which manufactures and supplies the APIs, is also accelerating factory expansion efforts after repeatedly posting record-breaking performances.According to the Financial Supervisory Service on the 20th, Yuhan signed a KRW 210.2 billion API supply agreement with Gilead Sciences. The contract amount corresponds to 9.91% of the company’s recent revenue.The contract period runs from May 19, 2026 through December 31, 2027. Yuhan purchases APIs manufactured by its subsidiary, Yuhan Chemical, and exports them to multinational pharmaceutical companies. Under the structure, Yuhan secures orders from global pharmaceutical firms, while Yuhan Chemical handles the actual production.Yuhan Corp’s main API supply agreements (Source: Financial Supervisory Service) *AI-generated imageThis marks the largest single API export contract in Yuhan’s history. The previous record was a KRW 140 billion antibiotic API supply agreement signed with Wyeth in 2007. Yuhan has now broken its own record for the largest single contract after 19 years.If the supply volume is exhausted within the contract period of one year and seven months, this implies average monthly sales of over KRW 10 billion. Since 2024, Yuhan Corp has secured a series of large-scale API supply agreements. Most of these contracts are with Gilead.In September 2024, Yuhan signed a KRW 107.7 billion HIV treatment API supply agreement with Gilead.Last year alone, Yuhan signed three separate API supply agreements with Gilead totaling KRW 258.1 billion. In May this year, the company also finalized a KRW 56 billion API supply agreement with BridgeBio Pharma. Industry analysts believe repeated large-scale contracts reflect multinational pharmaceutical companies’ confidence in Yuhan’s API quality and stable supply capabilities.All API supply agreements signed since last year are expected to generate revenue this year. A contract signed with Gilead in May last year scheduled KRW 88.8 billion in supplies from May 2025 through December 2026.Another KRW 84.3 billion contract signed in August last year covers supply from March 2026 through February 2027. In addition, a KRW 85 billion HCV treatment API supply agreement signed in August last year will generate sales over a two-year period beginning this May. Assuming supplies proceed as scheduled, Yuhan is expected to secure more than KRW 280 billion in overseas sales this year alone.Yuhan’s overseas business revenue has expanded significantly amid strong API export growth.The company’s overseas business revenue reached KRW 386.5 billion last year, up 26.1% year-on-year. After surpassing KRW 300 billion for the first time in 2024 and setting a new record for the first time in seven years since 2017, the company achieved a second consecutive annual record high. Compared to export sales of KRW 182 billion in 2022, Yuhan’s export revenue more than doubled within three years, reflecting a steep growth trajectory over the past three years.AI-generated imageYuhan recorded export sales in the KRW 200 billion range for four consecutive years from 2016 to 2019, but this dropped to KRW 144.9 billion in 2020. Exports rebounded to KRW 156.2 billion in 2021, up 7.9% year-on-year, and have continued rising for five consecutive years through last year. In the first quarter of this year, overseas business revenue reached KRW 106 billion, up 21.4% from the same period last year, maintaining the upward trend.While overseas operations accounted for only 8.9% of Yuhan’s total revenue in 2020, this share has risen for five consecutive years, reaching 17.7% last year.Yuhan Chemical, which supplies APIs to Yuhan, is also posting strong results.Yuhan Chemical recorded KRW 289.7 billion in sales last year, up 36.5% from KRW 212.3 billion the previous year, while operating profit rose 89.9% year-on-year to KRW 22.9 billion. Operating margin improved by 2.2 percentage points to 7.9%. Compared to sales of KRW 149.5 billion in 2022, last year’s revenue nearly doubled within three years. Operating profit during the same period more than tripled from KRW 6.6 billion.Yuhan Chemical is also accelerating its expansion plans to increase production capacity. In April last year, the company completed the expansion of the HB building at its Hwaseong plant, securing production capacity of 995,000 liters. The facility is equipped with continuous manufacturing systems and data-integrity-based infrastructure, earning recognition for achieving global-level quality competitiveness. This has enabled the company to establish a one-stop response system covering everything from small-scale clinical manufacturing to large-scale commercial production.Additional expansion is also underway. Yuhan Chemical is building an HC facility at the Hwaseong plant with a capacity of 292,000 liters. Construction is scheduled to begin this year, with operations targeted for the first half of 2028. Upon completion, Yuhan Chemical’s total production capacity is expected to expand to 11,287,000 liters. Through this, the company aims to secure manufacturing infrastructure capable of responding to growing global CDMO demand while strengthening its mid- to long-term growth momentum.
- Company
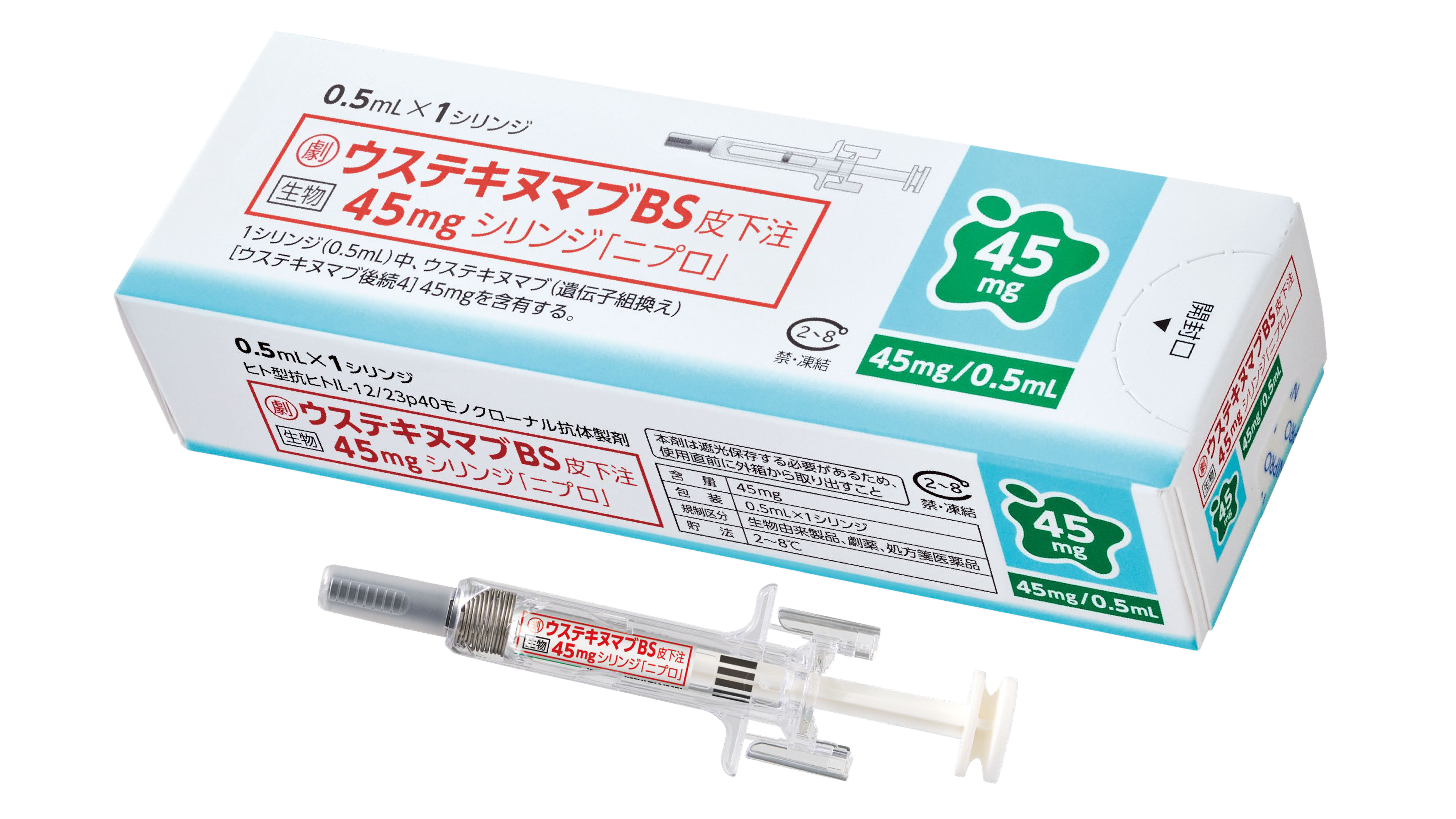
- Samsung Bioepis enters Japanese market with Nipro
- by Hwang, byoung woo May 21, 2026 10:16am
- Samsung Bioepis product image_SB17 JapanSamsung Bioepis has officially entered the Japanese market with the launch of its first biosimilar product in the country.Samsung Bioepis announced on the 20th that it has launched the autoimmune disease treatment ‘SB17’ (active ingredient: ustekinumab) in Japan through its local marketing partner, Nipro Corporation.The product is approved in Japan as ‘Ustekinumab BS 45 mg Syringe for S.C. Injection「NIPRO」.’SB17 referencs Johnson & Johnson’s autoimmune disease treatment Stelara. The therapy works by inhibiting the activity of interleukin-12 and interleukin-23, signaling molecules involved in immune responses. In Japan, the product has been approved for the treatment of plaque psoriasis and psoriatic arthritis.Samsung Bioepis received marketing authorization for SB17 from Japan’s Ministry of Health, Labour and Welfare in December last year. The product was officially launched follosing its listing on Japan’s National Health Insurance reimbursement list on the 19th.The launch carries significance as it marks Samsung Bioepis’ first commercialized biosimilar product in Japan. Last June, the company signed a strategic partnership agreement with Nipro to commercialize multiple biosimilar products in Japan, including its Stelara biosimilar.Samsung Bioepis plans to expand the launch of subsequent biosimilar products in Japan, starting with this SB17 launch.Jin-han Chung, Vice President and Head of Commercial Strategy for International Markets, at Samsung Bioepis. said, “By providing high-quality biosimilar treatment options to patients with autoimmune diseases in Japan, we hope to improve treatment accessibility and will continue to strive to address unmet needs across various therapeutic areas.”Meanwhile, Samsung Bioepis is marketing its Stelara biosimilar under the brand name ‘Pyzchiva’ in Europe and the United States, and ‘Epyztek’ in Korea.
- Company
- BeOne Medicines strengthens clinical-centered role
- by Son, Hyung Min May 21, 2026 10:16am
- BeOne Medicines is accelerating its research through an early-stage clinical-focused anticancer development strategy. As the paradigm of anticancer drug development shifts toward biomarker-based precision medicine, integrated Phase 1/2 development and simultaneous verification of multiple treatment strategies are becoming increasingly common.In particular, the company is strengthening combination therapy strategies and patient-selection-based development from the early stages while also focusing on expanding its domestic clinical network. According to the company, rather than simply competing on development speed, the focus is on rapidly delivering effective therapies while maintaining patient safety and data integrity.On the 19th, BeOne Medicines held a media forum in Gwanghwamun, Seoul under the theme “Latest Trends in Oncology Clinical Research and the Current Status and Outlook of Domestic Clinical Trials.” The event was organized ahead of Clinical Trials Day on the 20th.Presentations highlighted recent changes in oncology clinical research trends, the strengths and challenges of Korea’s clinical trial environment, and the need to expand early-stage clinical research.Eui-kyung Lee, Regional Communication and Patient Engagement Lead at BeOne Medicines, said, “From the company’s founding stage, BeOne Medicines aimed to create a structure that more rapidly and comprehensively connects drug discovery, clinical development, regulatory approval, and commercialization. In fact, since our in-house clinical team directly manages most of our trials, the time required until database lock is approximately 30% faster than the industry averageShe added, “We invest more in clinical research than we generate in sales. Guided by the philosophy that only innovation that reaches patients is true innovation, we are conducting oncology research focused on improving patient accessibility.”According to BeOne Medicines, the company currently has more than 180 ongoing global clinical trials, with over 40 studies already in Phase III or regulatory review stage. Approximately 30,000 patients are participating in these global trials.“Boundary between Phase 1 and 2 Is blurring”…Combination and frontline treatment validation begins earlierHye-sun Kim, Associate Director, Scientific Site Engagement Lead, Clinical operations at BeOne Medicines KoreaHye-sun Kim, Associate Director of Clinical Operations at BeOne Medicines Korea, explained that oncology clinical trials are increasingly moving away from traditional phased development structures.Kim said, “In the past, the process was sequential: determining dose in Phase I, confirming efficacy in Phase II, and then proceeding to confirmatory Phase III trials. Recently, however, integrated Phase I/II designs that simultaneously evaluate multiple patient groups and combination strategies have become increasingly common.”She continued, “While it was previously common to first obtain approval for monotherapy in later-line treatments before moving it to earlier-line settings, the trend is now shifting toward evaluating c combinations involving chemotherapy, immuno-oncology agents, and targeted therapies from the early stages.”She also emphasized the growing importance of biomarkers.Kim stated, “Biomarkers are evolving beyond mere tools for identifying targets to serve as a language that explains outcomes. Today’s clinical trials must simultaneously consider patient selection, risk management, treatment timing, and combination strategies, making their design and operation far more complex than in the past.”She also noted that the rapid expansion of antibody-drug conjugates (ADCs), bispecific antibodies, and multi-target therapies is increasing the burden of clinical trial design.“In the past, therapies targeted a single mechanism, but now development involves simultaneously controlling two or more targets or utilizing immune cells. This is an era in which drug-drug interactions, toxicity management, and optimal dose adjustment all need to be considered.”She added, “Simply moving quickly does not in itself constitute innovation. The key is to rapidly deliver effective treatments to patients who truly need them while maintaining patient safety and data integrity.”Korea’s strength in early-stage clinical trials highlightedJihye Cha, Senior Manager and Scientific Site Engagement Lead at BeOne Medicines KoreaJihye Cha, Senior Manager and Scientific Site Engagement Lead at BeOne Medicines Korea, said Korea still maintains strong competitiveness in the global oncology clinical trial landscape.According to Cha, Korea ranked fourth globally in 2023 and sixth in 2024 in terms of company-sponsored clinical trial registration data, particularly maintaining strength in early-stage clinical trials.Cha said, “Korea’s strengths include rapid patient enrollment, high data reliability, and strong global communication capabilities among investigators. The university hospital-centered healthcare system also enables highly efficient clinical trial execution.”She added, “The direction of global clinical development is shifting beyond simple trial execution toward involvement from the strategic development stage itself. Investigators and institutions, therefore, need to expand their roles in early-stage clinical research.”BeOne Medicines is also accelerating the expansion of early-stage clinical trials in Korea, leveraging the domestic clinical environment.According to the company, it has conducted or is conducting 55 clinical trials domestically, including 20 Phase I, 15 Phase II, and 20 Phase III trials. Currently, 27 studies are ongoing, involving more than 1,300 registered patients in Korea. Solid tumors account for approximately 75% of the company’s domestic clinical portfolio.In particular, various target- and modality-based pipelines beginning with immuno-oncology therapies are entering Korean clinical trials, and key candidates targeting breast cancer, lung cancer, liver cancer, and gynecologic cancers are preparing to advance into subsequent clinical phases.Cha explained, “Over the past three years, we have been one of the companies conducting the largest number of Phase 1 clinical trials in Korea. We are expanding clinical opportunities so that as many investigators and institutions as possible can gain experience with promising novel therapies.”She added, “People sometimes ask why more anticancer drugs are needed when so many already exist, but from the patient perspective, that is absolutely not the case. Even within the same cancer type, biomarkers and treatment responses differ greatly between patients, so more treatment options are still needed.”BeOne Medicines also stated that it is expanding discussions with Korean biotech companies regarding combination therapies and collaborative research.A company representative said, “We have held approximately 30 meetings with Korean biotech companies. We are focusing on systematically expanding domestic R&D synergies primarily through licensing-in opportunities.”
- Policy
- Korea approves Keytruda SC, developed with Alteogen's technology
- by Lee, Tak-Sun May 21, 2026 10:15am
- Product photo of KeytrudaThe subcutaneous (SC) formulation of 'Keytruda (pembrolizumab),' a global blockbuster immunotherapy for cancer treatment, is set to enter the Korean market after securing marketing authorization from the Ministry of Food and Drug Safety (MFDS). This approval is evaluated as a major achievement for the domestic biopharma industry, as the product integrates Alteogen's platform technology, and Samsung Biologics has been confirmed as part of the global contract manufacturing organization (CMO) lineup.On the 19th, the MFDS granted official marketing authorization for MSD Korea's 'Keytruda SC Inj (recombinant).'The newly approved Keytruda SC was developed using ALT-B4, a modified human hyaluronidase platform technology from the domestic biotech company Alteogen. It features a formulation combining the active ingredient, pembrolizumab, with 'berahyaluronidase alfa,' an enzyme that temporarily degrades the extracellular matrix of subcutaneous tissue. Using this technology, the existing intravenous (IV) formulation was converted into an SC formulation, enabling rapid dispersion and permeation of the drug into subcutaneous tissue.The contribution of South Korean companies is also significant in the manufacturing sector. According to the MFDS approval details, Keytruda SC's global contract manufacturing network includes Samsung Biologics, Korea's largest CMO, alongside BSP Pilot, Boehringer Ingelheim, and MSD headquarters. This system has enabled an innovative new drug developed with Korean platform technology to be supplied globally through Korea's state-of-the-art manufacturing facilities.Patient convenience is expected to improve dramatically. With the conventional Keytruda IV formulation, patients had to spend 30 minutes to an hour receiving an infusion in a hospital infusion center bed. In contrast, Keytruda SC can be administered by healthcare professionals into the thigh or abdomen in just one to two minutes. The dosing schedule can be selected based on the patient's condition, offering options for either a 3-week cycle (395 mg) or a 6-week cycle (790 mg). Patients currently receiving the IV formulation can also transition to the SC formulation starting with their next scheduled dose, which is expected to yield strong preference in clinical settings.The approved indications include the extensive scope of the existing IV formulation. Keytruda SC secured approvals across a total of 18 cancer types, including ▲melanoma ▲non-small cell lung cancer (NSCLC) ▲malignant pleural mesothelioma ▲head and neck cancer, classical Hodgkin lymphoma ▲urothelial carcinoma, gastric cancer ▲esophageal cancer ▲renal cell carcinoma, endometrial carcinoma ▲triple-negative breast cancer (TNBC) ▲cervical cancer ▲biliary tract cancer ▲hepatocellular carcinoma ▲microsatellite instability-high (MSI-H) solid tumors. This approval is based on data from a large-scale global bioequivalence clinical trial (MK-3475A-D77), which demonstrated comparable pharmacokinetic (PK) properties, efficacy, and safety profiles to the IV formulation.A pharmaceutical industry employee stated, "Keytruda is a mega-blockbuster competing for the top spot in global sales," and added, "The fact that Alteogen's technological advancement and Samsung Biologics' manufacturing capabilities have been integrated into global pharma giant MSD's core pipeline is a major milestone, demonstrating that the standing of the Korean pharma and biotech industry has taken a significant leap forward." Keytruda is a mega blockbuster that recorded global sales of KRW 46 trillion last year. In South Korea, Keytruda's sales have surpassed KRW 500 billion, ranking as the highest-performing pharmaceutical in the country.