- LOGIN
- MemberShip
- 2026-06-06 12:33:12
- Company
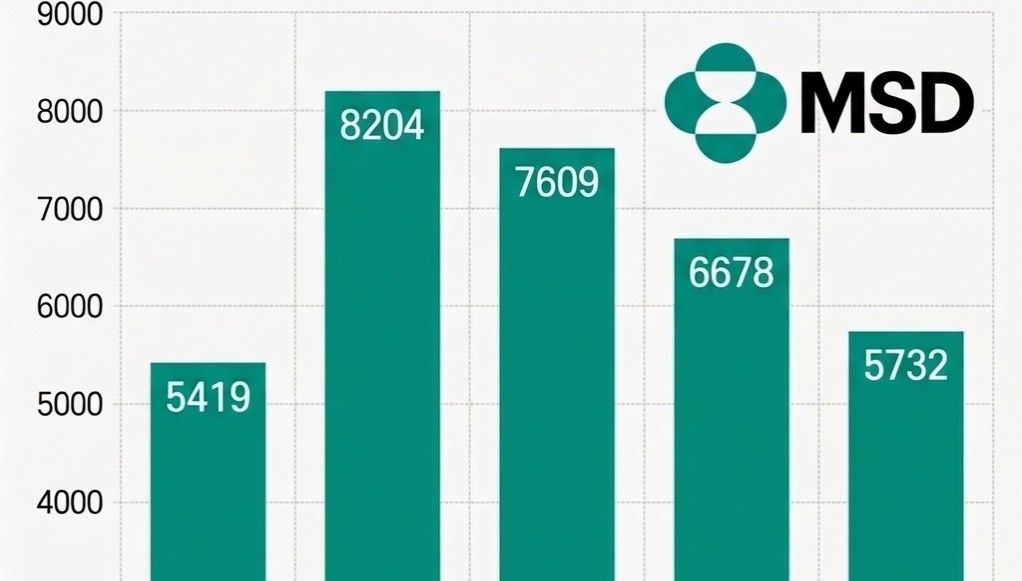
- MSD Korea sales 30%↓ in three years
- by Son, Hyung Min Apr 13, 2026 09:11am
- MSD Korea's performance continued to decline due to lower demand for COVID-19 treatments. As sales from treatments that drove performance during the pandemic have rapidly shrunk, existing core products have failed to offset the loss.According to the Financial Supervisory Service's electronic disclosure system on the 13th, MSD Korea's sales decreased by 14.2%, from KRW 667.8 billion in 2024 to KRW 573.2 billion last year. During the same period, operating profit dropped 13.0%, from KRW 24.9 billion to KRW 21.6 billion.MSD Korea Sales Trend by Year (unit: KRW 100 million). MSD Korea's sales decreased by 14.2%, from KRW 667.8 billion in 2024 to KRW 573.2 billion in 2025.The primary reason for the sales decline is the lack of supply of the COVID-19 treatment 'Lagevrio (molnupiravir).' MSD Korea explained that the absence of a supply contract with the Korea Disease Control and Prevention Agency (KDCA) last year affected the sales decrease.In fact, MSD Korea's sales have shown significant volatility, reflecting demand for COVID-19 treatments. Revenue peaked at KRW 820.4 billion in 2022 when demand was the highest, but subsequently decreased to KRW 760.9 billion in 2023 and KRW 667.8 billion in 2024 following the transition to the endemic phase. Compared with last year's sales of KRW 573.2 billion, revenue has shrunk by 30.1% over the past three years.Despite having a strong lineup of major products, including the immunotherapy 'Keytruda (pembrolizumab),' the cervical cancer vaccine 'Gardasil,' and the pneumococcal vaccines 'Vaxneuvance' and 'Capvaxive,' it was not enough to fill the void left by the end of the COVID-19 sales.However, the company continued investing in its Research and Development (R&D).According to data released by MSD Korea, the company invested 78 billion KRW in R&D last year, accounting for approximately 14% of its revenue, and has consistently invested over KRW 70 billion annually for the past five years. Despite the short-term performance decline, the company appears to be continuing its strategic investments to secure a foundation for medium- to long-term growth.New Indications·Pipeline Additions…Seeking a Rebound in PerformanceWhile performance has declined due to the sales void from COVID-19 treatments, MSD Korea is seeking a rebound opportunity by reorganizing its portfolio around oncology, vaccines, and rare diseases.The scope of Keytruda was rapidly expanded this year, with 11 additional indications, including triple-negative breast cancer and endometrial cancer, added to the reimbursement list. Furthermore, reimbursement for combination therapy with 'Padcev (enfortumab vedotin)' in urothelial carcinoma is also imminent.RSV preventive antibody injection 'Enfloncia (clesrovimab)' Keytruda has become a pillar of treatment with expanded reimbursement scope as a standard of care (SOC) across major solid tumors. This treatment has the most indications among drugs authorized in Korea.At the same time, efforts to develop new growth engines for infectious diseases are underway. MSD Korea has applied for the authorization of 'Enfloncia (clesrovimab),' an RSV preventive antibody injection for neonates and infants, and there is talk of possible approval in the second half of this year.Enfloncia is a long-acting monoclonal antibody that, in Phase 2b/3 clinical studies, demonstrated reductions of 60.5% in the occurrence of RSV-related lower respiratory tract infections and 84.3% in the risk of hospitalization.In addition, the reimbursement process for 'Winrevair (sotatercept),' a treatment for pulmonary arterial hypertension (PAH), is accelerating following its inclusion in the pilot project for concurrent authorization, evaluation, and negotiation.Winrevair is the first approved activin signaling inhibitor (ASI) in pulmonary arterial hypertension and offers a new mechanism of action after 20 years. This treatment works by blocking excessive activin signaling. This protein complex promotes cell proliferation in pulmonary arterial vessels, and restores the balance with anti-proliferation signals to induce reverse remodeling, normalizing altered vascular structures.As the impact from the termination of the COVID-19 special demand is being reflected, the expanded reimbursement for major products and the introduction of new drugs are expected to be key drivers of a future performance rebound.
- Policy
- Label updated for hypertension drug nebivolol
- by Lee, Tak-Sun Apr 10, 2026 08:27am
- Nebivolol original ‘Nebilet’The label for the antihypertensive drug nebivolol will now include the statement, “Beta-blockers may further increase the risk of severe hypoglycemia when co-administered with sulfonylureas (SU).”This follows safety measures taken by the European Medicines Agency (EMA), with Korea’s Ministry of Food and Drug Safety (MFDS) moving to revise product labeling.While concerns about hypoglycemia due to symptom masking when beta-blockers are used in combination with sulfonylureas (SU) are well known, the latest change is intended to further emphasize the severity of the risk and strengthen monitoring.On the 6th, MFDS announced draft labeling revisions for nebivolol-containing products and will collect feedback through the 21st.This draft is based on safety information from the European Medicines Agency (EMA). Nebivolol is a third-generation beta-blocker.According to the proposed amendment, the following statement will be added to the General Precautions section: “Beta-blockers may further increase the risk of severe hypoglycemia when co-administered with sulfonylureas. Patients with diabetes should be advised to carefully monitor their blood glucose levels.”Additionally, the following statement will be added to the “Drug Interactions” section. “Concomitant use of beta-blockers and sulfonylureas may increase the risk of severe hypoglycemia.”Current labeling already advises caution when administering the drug to patients with diabetes. This is because beta-blockers, including nebivolol, can mask specific symptoms of hypoglycemia, such as tachycardia and palpitations,As a result, patients may fail to recognize hypoglycemia and progress to severe hypoglycemia.Consequently, this may increase the likelihood that patients will progress to severe hypoglycemia without recognizing the symptoms.The Interactions section currently also includes a statement that co-administration with insulin or oral antidiabetic agents may mask specific symptoms of hypoglycemia (palpitations, tachycardia).However, the current labeling does not specifically warn against co-administration with sulfonylureas (SU) among antidiabetic agents.Last year, the EMA reviewed evidence suggesting that the risk of severe hypoglycemia may increase when nebivolol is used in combination with sulfonylureas and required that this information be added to the product label.MFDS appears to have prepared the Korean revision as a follow-up measure to the EMA’s actions.Currently, there are 25 nebirol-containing products approved in Korea. These include the original “Nebilet Tab” (Menarini Korea), the low-dose “Nebilet M Tab” (Kwangdong Pharmaceutical), and the nebirol-rosuvastatin combination drugs “Nebirosta Tab” (Elyson Pharm) and “Crebista Tab” (Arlico Pharmaceutical).According to UBIST, Nebilet recorded KRW 8.6 billion in outpatient prescriptions last year.
- Policy
- Opposition to labeling obesity drugs as 'misuse drug'
- by Lee, Tak-Sun Apr 10, 2026 08:27am
- AI-generated imageVoices are growing against the plan to designate GLP-1 obesity treatments, such as Wegovy and Mounjaro, as "high potential misuse and abuse." Critics point out that such a designation could instead drive patients toward illegal distribution channels.Analysis suggests that other countries are already putting effort into establishing an environment for safe use by monitoring distribution and prescription, rather than regulating the drug itself.On the 8th, the Ministry of Food and Drug Safety (MFDS) will discuss whether to designate these treatments through the Central Pharmaceutical Affairs Advisory Committee (CPAC).Currently, this category includes erectile dysfunction treatments, premature ejaculation treatments, and anabolic steroids. Once designated, the phrase "high potential misuse and abuse" must be displayed on the product packaging, and sales without a prescription are prohibited even in areas exempt from the separation of prescribing and dispensing.The MFDS's push for this designation is due to issues such as non-face-to-face prescribing, online black-market transactions, and indiscriminate off-label use, following the immense popularity of Wegovy and Mounjaro. Following discussions in last year's parliamentary audit, health authorities have been reviewing this designation.However, the industry is voicing concern that the designation could lead to illegal distribution, resulting in a balloon effect.However, the industry warns that labeling these drugs as potentially misused could shrink the patient base and prevent those who genuinely need treatment from receiving it.They argue that global trends focus on regulating the distribution and prescription stages rather than the drugs themselves.In Japan, as the prescribing of Ozempic (semaglutide) for obesity purposes increased, guidelines were established for prescribing institutions. According to an analysis conducted by the University of Tokyo research team of Japanese medical institution websites, institutions that advertised non-reimbursed prescriptions for GLP-1 receptor agonists exhibited significantly lower information quality. Furthermore, a large majority of these institutions were found to violate pharmaceutical advertising guidelines.Japan's Ministry of Health, Labor and Welfare (MHLW), when approving health insurance coverage for Wegovy in February 2024, set strict standards for prescribing institutions and patient lifestyle habits.Australia is also focusing on blocking unofficial distribution channels. From October 2024, Australia's Therapeutic Goods Administration (TGA) excluded GLP-1 drugs from the pharmacy compounding exemption. The system is designed to allow pharmacies to prepare similar drugs in response to drug shortages or to provide customized preparation for a specific patient.They also intensified advertising regulations, imposing fines of approximately 198,000 AUD (about 200 million KRW) for illegal advertisements on telehealth platforms.Japan and Australia have clear medical evidences and are focusing on monitoring illegal distribution and prescriptions, rather than designating obesity drugs that are approved by the regulatory authority as having the potential to be misused and abused.The WHO also emphasized 'regulated distribution networks, prescriptions by qualified medical professionals, and strong supervision' in its December 2023 global guidelines to counter the spread of counterfeit and substandard GLP-1 products.An industry official stated, "Major countries are setting standards for eligible prescribing institutions and restrictions on non-face-to-face prescriptions," adding, "They are putting effort into monitoring whether prescriptions align with BMI and comorbidities." The official added, "The Korean government and medical community should develop systematic management plans that block cosmetic use while protecting patient access for those who truly need treatment."There is also concern that the negative stigma of being a "high potential to misuse and abuse" could affect severely obese and type 2 diabetic patients. Another industry official stated, "Designation of 'high potential to misuse and abuse' mandates labeling, and the label points at all obesity patients. Not only those with severe obesity but also type 2 diabetic patients would receive medication stamped with 'potential to misuse'."Furthermore, there is a risk that patients who find it harder to obtain legitimate prescriptions may turn to unverified online purchases or illegal distribution channels. With cases of fraud and harm from illegal transactions already present in Korean online communities, critics fear that the official designation could expand this illegal market.
- Policy
- Samsung Bioepis-Hanmi joint sales 'Obodence' wins nod for IIT
- by Lee, Tak-Sun Apr 10, 2026 08:27am
- Product photo of ObodenceA large-scale investigator-initiated trial (IIT) for 'Obodence,' Samsung Bioepis' denosumab biosimilar, will begin.Led by Yeouido St. Mary's Hospital, this clinical trial is drawing significant attention as a large-scale study involving 13 medical institutions across South Korea. It is expected that this trial will provide clearer evidence of Obodence's efficacy, specifically in Korean patients.On the 8th, the Ministry of Food and Drug Safety (MFDS) approved the clinical trial protocol for 'Obodence Prefilled Syringe' requested by Catholic University of Korea Yeouido St. Mary's Hospital.The clinical trial aims to evaluate the efficacy of the denosumab biosimilar in postmenopausal women diagnosed with advanced osteopenia. It is designed as a multicenter, randomized, open-label study, corresponding to a Phase 4 post-marketing clinical trial.Obodence was launched in July of last year as a biosimilar to Prolia, an osteoporosis treatment. Prolia has dominated the osteoporosis market in South Korea, recording annual sales of approximately KRW 180 billion, given its strong efficacy and the convenience of once-every-six-month administration.The biosimilar market took off last year. In March, the first biosimilar, Celltrion's 'Stoboclo,' began joint sales with Daewoong Pharmaceutical. In July, Samsung Bioepis launched Obodence in the market through a partnership with Hanmi Pharmaceutical.The market share for biosimilars is on the rise, with Stoboclo recording sales of KRW 11.8 billion within its first 10 months.Samsung Bioepis, with Obodence launched slightly later than Stoboclo, is now focusing on marketing strategies to secure trust through clinical evidence.Notably, Samsung Bioepis is emphasizing to medical professionals that Obodence demonstrated results highly similar to the original in Phase 3 trials that included Korean participants. The Phase 3 study involved 457 postmenopausal patients with osteoporosis across five countries, including South Korea.In this regard, the current IIT is expected to serve as an opportunity to accumulate further evidence of its efficacy in Koreans. Given that the trial is being conducted across 13 domestic hospitals, it is anticipated to include a larger number of Korean subjects than the Phase 3 trial did.However, the company clarified that, as this is an investigator-initiated trial, it does not necessarily reflect the firm's specific corporate objectives.Stoboclo also received approval for an IIT last February. The study, conducted by Ajou University Hospital, is designed to evaluate the efficacy of denosumab in obese subjects treated with GLP-1 receptor agonists who exhibit weight loss and bone metabolism risk factors.
- Company
- AZ launches Tezspire in Korea with expanded indication
- by Son, Hyung Min Apr 10, 2026 08:27am
- AstraZeneca Korea (CEO Eldana Sauran) announced on the 8th the domestic launch of Tezspire (tezepelumab) as an add-on maintenance treatment for severe asthma and chronic rhinosinusitis with nasal polyps (CRSwNP).With this domestic launch, Tezspire has simultaneously expanded its indication to include its use as an add-on maintenance treatment for adults with inadequately controlled CRSwNP, broadening its use as an anti-TSLP (Thymic Stromal Lymphopoietin) treatment option for severe asthma to CRSwNP.TSLP is a driver of multiple inflammatory responses and is expressed at higher levels in CRSwNP patients than in patients without polyps. Tezspire is an anti-TSLP monoclonal antibody that blocks TSLP activity at the upstream level of inflammatory pathways. Tezspire’s clinical efficacy and safety profile were confirmed in the global Phase III WAYPOINT trial.The WAYPOINT study was a multicenter, randomized, double-blind, placebo-controlled Phase III clinical trial conducted in 10 countries involving 408 patients aged 18 years and older with CRSwNP who had severe, uncontrolled symptoms.Results showed that at Week 52, the Tezspire treatment group demonstrated statistically significant improvements compared to the placebo group, with a decrease of -2.07 in the Nasal Polyps Score (NPS), which assesses the size and extent of nasal polyps, and a decrease of -1.03 in the Nasal Congestion Score (NCS), which assesses the degree of nasal congestion. Furthermore, these improvements were observed as early as week 4 and week 2 of treatment, respectively, and were sustained through week 52.Ji-young Kim, Executive Director of AstraZeneca Korea’s Respiratory Division, said, “We are pleased that we were able to expand Tezspire’s indication in Korea following FDA approval in October last year for CRSwNP. Clinical trials confirm Tezspire can be an effective treatment option not only for asthma but also for patients with CRSwNP, and we expect it to help patients manage their respiratory conditions.”Rhinosinusitis is characterized by two or more symptoms, including nasal congestion, nasal obstruction, or a runny nose, and becomes chronic when these symptoms persist for 12 weeks or longer. Additionally, when accompanied by nasal polyps, it is classified as CRSwNP.
- Company
- Boston Scientific Korea’s sales surpass ₩200 billion
- by Hwang, byoung woo Apr 10, 2026 08:26am
- Boston Scientific Korea has surpassed KRW 200 billion in sales following portfolio restructuring.Analysts attribute this revenue expansion to the robust growth of the Pulse Field Ablation (PFA) system, coupled with concurrent growth across all major therapeutic areas.According to a recent audit report, Boston Scientific Korea posted KRW 218.5 billion in sales in 2025, up 18.4% from KRW 184.6 billion the previous year.This marks the first time the company has surpassed KRW 200 billion in revenue since entering the Korean market, continuing its four-year growth streak from KRW 151.6 billion in 2022, KRW 175.3 billion in 2023, and KRW 184.6 billion in 2024.Operating profit also grew in tandem with topline growth, from KRW 9 billion in 2022, KRW 10.5 billion in 2023, KRW 11 billion in 2024, to KRW 13 billion in 2025.Increased PFA procedures for arrhythmia drive growthThe key driver behind the company’s growth lies in changes to the business portfolio. The product lines currently supplied by the company to domestic medical institutions span the cardiovascular, oncology, and urology fields. After previously attempting to enter the structural heart disease segment with TAVI before discontinuing the business, the company restructured its portfolio.Representative products include the AVIGO Plus coronary ultrasound imaging device, the TheraSphere liver tumor embolization device, and the Rezum system for benign prostatic hyperplasia.Among these various products, the PFA system had the greatest impact on growth last year.Unlike conventional radiofrequency ablation or cryoballoon ablation, PFA selectively destroys myocardial cells only, reducing procedure time by more than half and lowering complication risks, thereby expanding its influence in major domestic general hospitals.The Korean PFA market is currently contested by Boston Scientific, Medtronic, and Johnson & Johnson (J&J).Boston Scientific was the fastest to enter the Korean market, securing catheter certification for its FARAPULSE platform in April 2024 and generator approval in September.Analysts attribute this growth to the expanding market for electrophysiological procedures, driven by an increase in atrial fibrillation patients amid an aging population, with a recurring revenue model centered on related catheters and systems serving as the foundation for growth.In a Korea Health Industry Development Institute report on PFA, Professor Bo-young Joung of the Department of Cardiology at Severance Hospital stated, “PFA cuts procedure time by half compared with conventional methods and lowers complication risk, leading to high satisfaction among both physicians and patients. Currently, 35% of atrial fibrillation ablation procedures at Severance are performed using PFA, and this trend is expected to continue.”Given this, Boston Scientific Korea’s growth momentum is expected to strengthen further.Rezūm expansion and global M&A broaden business scopeAnother factor contributing to growth is the continued expansion of the Rezūm System, a medical device introduced in 2023 for benign prostatic hyperplasia, which has now surpassed 6,000 cumulative procedures in Korea.The Rezūm System received approval from the U.S. Food and Drug Administration (FDA) in 2015, obtained authorization from the Ministry of Food and Drug Safety in 2022, and was designated as a new health technology by the Ministry of Health and Welfare in 2023. Currently, Rezūm procedures are expanding their scope of application in clinical settings as a treatment option that can be considered even for patients unsuitable for medication or surgery.Regarding this, Boston Scientific Korea Country Manager Ae Ri Jung said, “Achieving 6,000 Rezūm procedures is a meaningful milestone demonstrating its establishment as a treatment option for BPH in Korea. We will continue contributing to expanding treatment options that improve patients’ quality of life.”As the portfolio expands, the company appears to be strengthening its field sales capabilities. Looking at the sales and administrative expenses, promotional expenses rose from KRW 6.3 billion in 2024 to KRW 7.1 billion in 2025.In particular, as Boston Scientific is pursuing mergers and acquisitions (M&A) on a global scale involving tens of trillions of won, its influence in the domestic medical market is expected to continue to grow in the future.The company acquired Axonics in 2024 to strengthen its urology portfolio, and in January, decided to acquire neurovascular treatment company Penumbra.In addition, it has also recently fully integrated Valencia Technologies, a urinary incontinence treatment company, rapidly expanding its portfolio.As the company expands its business scope to include new disease areas in addition to those that generate synergies with existing businesses, its business scale in the Korean market is also expected to grow.
- Policy
- Drug pricing premium to hinge on innovative status
- by Jung, Heung-Jun Apr 10, 2026 08:26am
- As interest in innovative pharmaceutical company certification rises following the government’s drug pricing reform, pharmaceutical companies are keeping a close eye on the changed review requirements.With the required R&D investment ratio set to rise by 2 percentage points in three years, companies are particularly focused on the certification process scheduled for the second half of this year.According to industry sources on the 9th, whether a company receives the Innovative Pharmaceutical Company certification has become a key factor determining whether a company is a winner or a loser under the revised drug pricing system. Although all companies are subject to the same structural changes, they are effectively being handed very different report cards because of differences in treatment regarding existing price cuts and pricing premiums.An official from a domestic pharmaceutical company stated, “For Innovative Pharmaceutical Companies, price reductions for existing listings are spread out over 10 years, and there are pricing premiums and reduction clauses, so the impact of the reform plan is likely to be less severe. The extent of the impact will inevitably differ from what non-innovative companies experience.”The Korea Pharmaceutical and Bio-Pharma Manufacturers Association held two explanatory sessions on the pricing reform, one online on the 3rd and another on the 8th, and many of the questions were reportedly related to innovative and quasi-innovative company certifications.There was also interest in when and how the price adjustment method would be applied if a non-innovative company’s status were to change to innovative or quasi-innovative.The introduction of the quasi-innovative category has broadened interest beyond companies that had already been focused on the certification system to a wider range of firms.However, with the Ministry of Health and Welfare revising the Enforcement Decree and Enforcement Rules of the “Special Act on the Promotion and Support of the Pharmaceutical Industry,” companies now face the need to redesign their certification strategies.In particular, as the government has signaled a shift toward more microscopic scrutiny, such as the introduction of quantitative evaluations, while simplifying review criteria, more thorough preparation has become necessary.One of the most significant changes is that four items among the detailed evaluation criteria for certification, which include R&D investment, the number of clinical trials, and export volume, have been converted into quantitative indicators.Given the high level of interest from the pharmaceutical industry, the Ministry of Health and Welfare is holding a briefing session today (the 9th) for pharmaceutical companies to explain the improvements to the certification system.Another official from a domestic pharmaceutical company stated, “Although we have avoided an immediate increase in the R&D ratio, since the review criteria have changed, even companies that were already preparing will likely be re-evaluating their plans.”An official from a pharmaceutical company preparing for certification this year said, “Companies with an R&D ratio in the 5% range are particularly interested in the revised certification system. We have already completed our preparations to a certain extent, but we will still attend the briefing session.”
- Opinion
- [Reporter's View] Dilemma of off-label prescribing
- by Son, Hyung Min Apr 10, 2026 08:26am
- For patients who have used all cancer treatment options, the only remaining path is often clinical trials. This is because a system has become rigid, so that even if a treatment with expected efficacy exists, institutional constraints prevent it from being prescribed.Theoretically, off-label prescribing is permitted. In South Korea.To use an off-label drug, a hospital's Institutional Review Board (IRB) must first deliberate on the case, after which the Health Insurance Review and Assessment Service (HIRA) determines whether to approve. However, critics point out that IRBs are limited to specific hospitals, and because deliberation standards are applied at a very high level, significant hurdles remain before actual approval is secured.Furthermore, the problem is that this pre-approval system and the list of available treatments operate differently across hospitals. Even under identical patient conditions, a prescription may be possible at one specific hospital but impossible at another.Consequently, the decision to treat depends not on the patient’s clinical status but on the medical institution's decision. Patients must search for a hospital that can provide treatment, rather than choosing the treatment itself.As a result, it is difficult for patients even to know under what circumstances treatment is possible or to what extent it is allowed. This structure, in which treatment availability varies by hospital and situation despite the same disease and conditions, causes significant confusion.This issue is even more pronounced among patient groups outside approved indications. It is not uncommon for patients with the same biomarker to be excluded from treatment simply because they do not fall within the authorized indication. In a system where off-label use is effectively blocked, the options available to these patients become extremely limited.Within this framework, the only option left for patients is to participate in clinical trials. However, clinical trials are not a realistic option for everyone. Participation conditions are stringent, and accessibility varies widely by region and institution. Most importantly, for patients who might be missing their treatment window, a clinical trial is less a choice and more a last resort.Under these circumstances, the discussion on expanding off-label prescribing is emerging as an alternative rather than simply an option. Instead of dismissing this as a risky or unrealistic approach, it is necessary to consider it as a solution to guaranteeing treatment opportunities within a limited fiscal environment. At the very least, restructuring is required to ensure that the system does not block treatment opportunities.This is not a regulatory issue, but it is a minimum requirement to reduce confusion caused by a lack of standards and to provide patients with a predictable treatment environment. If the structure where clinical trials are the only remaining option persists, fair treatment opportunities can hardly be guaranteed.
- Policy
- Obesity drugs likely to be designated as drugs at risk of misuse or abuse
- by Lee, Jeong-Hwan Apr 10, 2026 08:26am
- The Ministry of Food and Drug Safety’s Central Pharmaceutical Affairs Council has issued an advisory opinion that GLP-1-based obesity treatments such as Wegovy and Mounjaro should be designated as drugs at risk of misuse and abuse.As GLP-1 obesity injections have gained popularity nationwide, indiscriminate prescribing and sales in areas exempt from the separation of prescribing and dispensing, as well as a sharp increase in pediatric prescribing, are said to have influenced the recommendation.According to the pharmaceutical industry on the 9th, following discussions on the validity of designating obesity treatments as drugs of concern for misuse or abuse, the Central Pharmaceutical Affairs Council agreed on the need to designate GLP-1 injectables as such.If regulations are finalized in line with the council’s recommendation, GLP-1 injections are expected to be designated as drugs of concern for misuse or abuse in the near future.Industry sources say that this decision was driven by a sharp rise in cases where GLP-1 obesity drugs are prescribed and sold for cosmetic purposes beyond obesity treatment, by exploiting areas exempt from the separation of prescription and dispensing, as well as growing risks associated with off-label prescriptions for children without approved indications.In fact, the Ministry of Health and Welfare has been working with the MFDS since last year to address misuse and abuse of newer obesity injections such as Wegovy and Mounjaro.To regulate indiscriminate prescriptions for cosmetic purposes, they have established an administrative policy to discuss designating these drugs as “drugs of concern for misuse or abuse” and to strengthen crackdowns on in-house dispensing by medical institutions that violate the principle of separation of prescribing and dispensing.GLP-1 obesity drugs have been associated with adverse effects ranging from relatively mild gastrointestinal side effects such as nausea, vomiting, and diarrhea to severe complications, including pancreatitis and intestinal obstruction.In particular, some medical institutions have also been criticized during NA audits for undermining the prescribing-dispensing separation principle by directly selling GLP-1 injections within hospitals through irregular in-house dispensing practices.If GLP-1 injectables are designated as drugs of concern for misuse or abuse in the future, they will be subject to much stricter distribution, prescription, and sales regulations compared to general prescription drugs.First, GLP-1 injectables will no longer be sold without a prescription, even in areas exempt from the separation of prescription and dispensing.In addition, product containers, package inserts, and outer packaging will be required to clearly display wording indicating that they are drugs at risk of misuse and abuse, so that consumers and healthcare professionals can immediately recognize the risk.Distribution oversight and surveillance will also be strengthened. The MFDS and the Ministry of Health and Welfare will more closely monitor distribution records among pharmaceutical companies, wholesalers, medical institutions, and pharmacies. If abnormal bulk purchases or prescribing patterns are identified, entities may face administrative penalties following intensive on-site inspections.A pharmaceutical industry official stated, “As Wegovy, Mounjaro, and similar agents have become extremely popular, usage has risen sharply, and GLP-1 prescriptions surged in conjunction with telemedicine during the early stages when regulations were absent. Even now, telemedicine continues to serve as a conduit linking medical institutions with patients seeking GLP-1 obesity prescriptions. In this context, the Central Pharmaceutical Affairs Council appears to have concluded that designation as drugs with a risk of misuse or abuse is unavoidable in order to control adverse effects.”Meanwhile, drugs designated as being at risk of misuse and abuse have so far included erectile dysfunction treatments such as sildenafil and tadalafil, premature ejaculation treatment dapoxetine, diuretics, and anabolic steroids. No obesity drug has previously been designated.
- Opinion
- "Preventing itch-scratch cycle…Dupixent for prurigo nodularis"
- by Son, Hyung Min Apr 10, 2026 08:26am
- Changes are emerging in the treatment landscape for prurigo nodularis, a condition of patients with an itch so intense it persists until they bleed, destroying their lives.With the introduction of the biologic 'Dupixent (dupilumab),' which targets the mechanism of the disease, there is a growing call for a reestablishment of treatment strategies, moving beyond the traditional symptom-focused approach.Tae Young Han, a professor of dermatology at Nowon Eulji University HospitalTae Young Han, a professor of dermatology at Nowon Eulji University Hospital, said during a recent meeting with Daily Pharm, "Prurigo nodularis accompanies the most severe itch among all dermatologic conditions, significantly deteriorating quality of life of a patient," adding, "With the introduction of a targeted therapy, such as Dupixent, changes to the treatment paradigm is anticipated."Prurigo nodularis is characterized by tens to hundreds of hard nodules accompanied by chronic, extreme itching. In over 80% of cases, the itch lasts more than six months, and for more than half, it persists for over two years. The psychological burden, including sleep disorders and depression, is so severe that quality of life is significantly compromised. The problem is the low awareness of the disease. It is often confused with atopic dermatitis, leading to diagnostic seeking where the correct diagnosis is significantly delayed. Missing the optimal treatment window can lead to chronicity and an increased risk of comorbidities.The mechanism of the disease differs from simple skin conditions. The key is a Type 2 inflammatory response where the immune and nervous systems interact. Interleukins such as IL-4, IL-13, and IL-31 trigger and amplify the itch, forming a vicious cycle. Sanofi’s Dupixent, developed based on this pathological mechanism, has emerged.Dupixent inhibits both IL-4 and IL-13 signals. This approach blocks the root cause of the disease rather than merely alleviating inflammation. It reduces itch, leading to decreased scratching and eventual improvement in skin lesions.Global clinical trials showed that the proportion of patients achieving significant itch reduction (WI-NRS reduction of 4 points) was approximately 3 times higher with Dupixent than with placebo, with rapid improvement starting in week 3. By week 24, the proportion of patients achieving clear or almost clear skin was significantly higher than that of the control group.Professor Han stated: "While existing treatments only non-specifically suppress inflammation, Dupixent is significant in that it directly inhibits the key cytokines of Type 2 inflammation to reach the root cause," and that "Reducing the itch leads to less scratching, which eventually improves the lesions."Q. How severe is the itch and the resulting decrease in quality of life?The most prominent feature is the itch's extreme intensity. The scale of itch is evaluated with the WI-NRS (0–10 scale): 0 means no pruritus and 10 means very severe pruritus.Many patients score 8 or higher, indicating very severe itch. Patients often describe not just itching, but stinging, pain, and a stabbing sensation.In terms of the DLQI (Dermatology Life Quality Index), prurigo nodularis patients experience a much greater decline in quality of life than even psoriasis patients. Many suffer from anxiety, depression, and sleep disorders. Some patients even express suicidal ideation, saying they "want to die because it itches so much."Q. How is the current treatment carried out?Previously, there were no treatments directly targeting the Type 2 inflammatory response. We typically start with topical steroid ointments. If the nodules are too firm so that ointments cannot be absorbed, direct steroid injections are made into the nodules. If the nodules are too numerous, phototherapy is used, and if that is insufficient, immunosuppressants are used.However, many patients are elderly (60s–80s), making these treatments difficult. Phototherapy requires standing naked in a closed booth for several minutes, which is a burden.Immunosuppressants carry risks for patients with decreased kidney function or a history of cancer. Consequently, treatment is often limited to ointments, making it hard to achieve a sufficient response.Under international guidelines (IFSI, US, and EU), biologics like Dupixent are recommended immediately if phototherapy or immunosuppressants are difficult to apply. In fact, biologics are considered the most effective options.Q. What is the clinical value of Dupixent compared to existing treatments?While conventional treatments focus on nonspecifically controlling overall inflammation, Dupixent differs mechanistically by directly inhibiting IL-4 and IL-13, the primary mediators of Type 2 inflammation, which is the core mechanism underlying the development of prurigo nodularis. By addressing the underlying cause of the disease, Dupixent approaches the condition at its root.Because Dupixent targets the fundamental cause to effectively reduce itching, the natural result of decreased pruritus is reduced scratching behavior, which subsequently improves skin lesions. When these immunological abnormalities are controlled, it is possible to break the itch-scratch cycle that forms between the nervous system and the inflammatory response.Furthermore, Dupixent has a robust safety profile from other indications. In fact, prurigo nodularis is strongly associated with repetitive scratching. Some patients even present with both atopic dermatitis and prurigo nodularis, as severe scratching from atopic dermatitis can progress to similar lesions. For such patients, including the elderly or those with underlying medical conditions, Dupixent can be used with relatively little burden, as it holds multiple indications for conditions such as atopic dermatitis, asthma, and chronic rhinosinusitis. Q. What improvements, in terms of patient symptoms, are seen after Dupixent treatment?Clinical studies report a rapid reduction in itching within three weeks of starting treatment. In actual clinical practice, significant relief of pruritus is observed within three to four weeks, leading to a noticeable improvement in the patient’s quality of life. In particular, it serves as a critical treatment option for patients who find it difficult to use immunosuppressants due to medical histories such as hepatitis, dialysis, or tumors.The administration protocol is specified as a continuous cycle every two weeks. By breaking the vicious itch-scratch cycle, Dupixent reduces scratching and improves skin lesions. Once this fundamental cycle is broken, cases have been observed in which the improved condition is maintained even after treatment is discontinued.Q. Would you like to provide advice for patients with prurigo nodularis and for medical staff treating them?Because the name prurigo nodularis is unfamiliar, many patients don't recognize their condition. Patients who do not seek dermatology often mistake their symptoms for simple eczema, being left without a correct diagnosis. They often wander between clinics. If a patient gets an accurate diagnosis from a dermatologist, symptoms can be improved. My advice is that with the emergence of new options such as biologics, patients should pursue treatment.