- LOGIN
- MemberShip
- 2026-06-06 12:33:12
- Company
- ‘Lower and Faster’…Dyslipidemia treatment strategy evolves
- by Son, Hyung Min Apr 21, 2026 07:33am
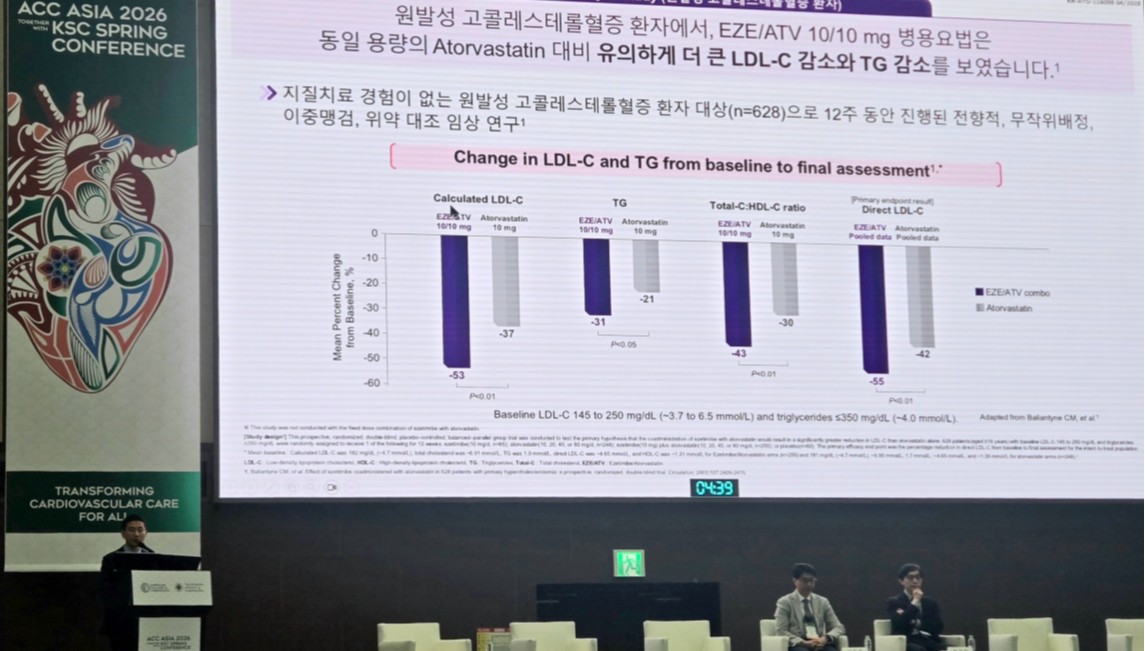
- Beyond the question of “how low to lower” LDL cholesterol, “how quickly to reach the target level” is emerging as a key variable in the treatment of dyslipidemia.As the target has been tightened to below 55 mg/dL, an approach aimed at reaching the goal quickly through initial combination therapy is emerging as an alternative.According to industry sources on the 21st, treatment strategies for dyslipidemia based on early combination therapy were presented as a major topic at the ACC Asia 2026 Together With KSC Spring Conference held recently in Gyeongju.Cardiovascular disease is one of the major causes of death in Korea, and dyslipidemia is considered a representative risk factor that can be corrected through treatment.Nevertheless, the 30-day and 1-year mortality rates of acute myocardial infarction patients in Korea still stand at approximately 9% and 16%, respectively, indicating that existing treatment strategies alone have not been sufficient to adequately reduce early mortality risk. This, together with the fact that the LDL cholesterol target attainment rate in very high-risk patients remains limited, suggests the need for a more aggressive treatment approach.Against this backdrop, the need for a combination therapy that can overcome the limitations of statin monotherapy has been consistently raised. Notably, the IMPROVE-IT study reported that adding ezetimibe to statins resulted in an additional approximately 24% reduction in LDL-cholesterol, along with a 24% and 32% reduction in the risk of myocardial infarction and ischemic stroke, respectively.With long-term safety and efficacy subsequently confirmed, ezetimibe has become established in major guidelines as a preferred combination and second-line treatment option.Professor Jin Wi of the Division of Cardiology at Gachon University Gil Medical Center emphasized the importance of statin + ezetimibe combination therapy at the ACC Asia 2026 Together With KSC Spring Conference.Professor Jin Wi of the Division of Cardiology at Gachon University Gil Medical Center said, “Dyslipidemia treatment is shifting from a high-intensity statin-centered approach to a more aggressive cholesterol-lowering strategy. It is important to reach LDL cholesterol targets quickly through early combination therapy.”In particular, he emphasized the importance of dyslipidemia management from a CKM (Cardio-Kidney-Metabolic) perspective, where cardiovascular, kidney, and metabolic diseases are interconnected, saying, “In these patients especially, LDL cholesterol should be controlled aggressively at an early stage.”He added, “Atorvastatin can be used in chronic kidney disease patients without dose adjustment, which gives it high clinical utility.”Professor Suk Min Seo, Division of Cardiology at Eunpyeong St. Mary’s Hospital, also noted, “In actual clinical practice, it is difficult to fully implement stepwise therapy due to limitations in patient follow-up. Applying high-intensity statin and ezetimibe combination therapy from the outset is a realistic strategy in terms of achieving the target within a short period and managing risk.”At the session that day, the ezetimibe/atorvastatin combination product ‘Atozet’ was also introduced as one of the initial combination therapy options.Beyond ‘lower’ to ‘faster’… guidelines and clinical evidence alignRecent guidelines are demanding changes not only in treatment goals but also in the treatment approach itself.The 2025 European Society of Cardiology / European Atherosclerosis Society (ESC/EAS) guidelines presented the LDL cholesterol goal for patients with atherosclerotic cardiovascular disease (ASCVD) as below 55 mg/dL and at least a 50% reduction from baseline, and in some very high-risk groups, recommended below 40 mg/dL.Furthermore, for treatment-naïve ACS patients, the guidelines explicitly recommend considering combination therapy with high-intensity statins and ezetimibe from the outset, emphasizing aggressive intervention at the start of treatment.The 2026 American College of Cardiology / American Heart Association (ACC/AHA) guidelines likewise presented an LDL cholesterol goal of below 55 mg/dL for very high-risk patients in secondary prevention, and recommend early addition of non-statin agents when the target is difficult to achieve with statin monotherapy alone. Accordingly, the treatment paradigm appears to be shifting from the existing stepwise approach to an early combination-centered strategy.Professor Suk Min Seo, Division of Cardiology at Eunpyeong St. Mary’s Hospital, presenting at the ACC Asia 2026 Together With KSC Spring ConferenceThis shift is supported by clinical research. The Ez-PAVE trial was a study designed to verify the clinical validity of lowering the LDL cholesterol target from the conventional 70 mg/dL to 55 mg/dL.In this study, which enrolled 3,048 Korean ASCVD patients, the group targeting an LDL-cholesterol level below 55 mg/dL achieved a statistically significant reduction of approximately 33% in major adverse cardiovascular events (MACE) over three years compared to the group targeting below 70 mg/dL. This suggests that a strategy of controlling LDL cholesterol to a lower level can lead not only to numerical improvement but also to an actual reduction in clinical events.Furthermore, the BETTER TRIAL study showed that early combination therapy resulted in significant improvements in both the magnitude of LDL-cholesterol reduction and the rate of target achievement compared to monotherapy. In this study of Korean patients with very high-risk ASCVD, early combination therapy with ezetimibe and atorvastatin, administered before reaching the maximum statin dose, demonstrated significant improvements in both the magnitude of LDL-cholesterol reduction and the rate of target achievement compared to monotherapy.In particular, the rate of achieving LDL-cholesterol levels below 55 mg/dL was 46.2% versus 9.0% at 6 weeks and 55.0% versus 15.4% at 12 weeks, clearly demonstrating the efficacy of the early combination strategy.Ultimately, the treatment of dyslipidemia is expanding beyond the question of “how low to lower” to include “how quickly to achieve” the target level. Considering changes in guidelines and clinical evidence, a strategy of applying aggressive combination therapy from the outset is expected to emerge as a key factor in improving patient outcomes.
- Company
- Amgen’s BiTE platform to become a pillar of solid tumor strategy
- by Son, Hyung Min Apr 20, 2026 04:58pm
- Bispecific antibody-based immuno-oncology platforms are expanding beyond hematologic malignancies into solid tumors, emerging as a new axis of treatment strategy transformation.Amgen is rapidly expanding its position as a next-generation immuno-oncology strategy by broadening the application scope of its ‘BiTE (Bispecific T-cell Engager)’ technology based on a research and development system that combines genetics and artificial intelligence (AI).Meejin Cho, Hematology-Oncology TA Lead, Medical Affairs, Amgen KoreaOn the 17th, Amgen Korea held a media session at its Seoul headquarters under the theme “AMGEN INNOVATION TALK BiTE – Innovation Driving Changes in Patient Treatment,” and unveiled its R&D strategy and direction for BiTE platform expansion.Under its mission of “To Serve Patients,” Amgen has been developing treatment options in areas of serious diseases such as cancer, cardiovascular disease, inflammation, and rare diseases. Recently, the company has been focusing on simultaneously enhancing the precision and speed of new drug development by integrating AI technology based on its understanding of human genetics and disease biology.The R&D strategy is structured around three pillars: ▲identifying the root causes of diseases and discovering new targets, ▲developing diverse treatment modalities, and ▲innovating clinical trial design.These strategies are being implemented in actual research processes. Amgen uses large-scale genetic data accumulated through its subsidiary deCODE Genetics to analyze disease causes and treatment responses with high precision. In addition, it applies the AI platform ‘Freyja,’ developed in collaboration with NVIDIA, to predict the probability of success of candidate substances in advance, thereby improving development efficiency.The use of AI is also expanding in the clinical stage. By analyzing real-world patient data through its proprietary machine learning model ‘ATOMIC’ and identifying institutions with a high likelihood of participating in clinical trials, the company claims to have increased patient recruitment speed by approximately threefold compared to previous methods. This directly contributes to shortening the drug development timeline and improving patient access.On this foundation, Amgen’s core platform is BiTE. BiTE is a bispecific antibody-based immuno-oncology platform that enables a patient’s immune T cells to directly recognize and attack cancer cells.Cancer cells evade immune surveillance through various mechanisms. While existing immune checkpoint inhibitors restore T-cell activity by blocking the PD-1/PD-L1 binding, BiTE represents a more advanced approach.In some cancers, cells evade T-cell recognition by reducing MHC class I expression, but BiTE bypasses this limitation. Its structure allows it to bind simultaneously to the T-cell’s CD3 and the target antigen on the cancer cell, thereby inducing immune cells to directly attack cancer cells without relying on the TCR-MHC class I recognition process.In other words, unlike previous approaches that only activated immune responses, BiTE physically connects T cells and cancer cells, inducing immediate cell death.Based on these mechanistic strengths, BiTE is expanding its application across various cancer types and has already proven its clinical value in hematologic malignancies.‘Blincyto (blinatumomab),’ a treatment for acute lymphoblastic leukemia (ALL), induces anti-cancer effects by linking CD19-positive B cells and T cells and has demonstrated improved survival compared to existing treatments. Based on this evidence, it is recommended as first-line consolidation therapy in the NCCN guidelines.Building on this success, Amgen is expanding the application of BiTE technology to solid tumors. A prime example is “Imdelltra (tarlatamab),” a treatment for extensive-stage small cell lung cancer.Small-cell lung cancer is a disease with rapid progression and high recurrence rates, representing an area of significant unmet medical need. Imdelltra is a BiTE-based therapy that targets DLL3 to induce T cells to directly attack cancer cells.Clinical results have also demonstrated meaningful outcomes. In patients whose disease progressed after receiving at least two prior treatments, including platinum-based chemotherapy, Imdelltra achieved an objective response rate of approximately 40% and a median overall survival (mOS) of 14.3 months. In the Asian patient population, this was extended to 19 months.Based on these data, Imdelltra was approved in the United States in 2024 and recommended as a subsequent therapy (Category 1) in NCCN guidelines. In Korea, it has been designated as a Global Innovative product on Fast Track (GIFT) and approved, with reimbursement procedures currently underway.The emergence of Imdelltra is evaluated as a turning point in small cell lung cancer treatment, which has seen little change over the past 30 years.Meejin Cho, Hematology-Oncology TA Lead at Amgen Korea, explained, “BiTE is particularly effective in diseases with clear target antigens and rapid progression. Expanding from ALL to small cell lung cancer, which has a clear target, DLL3, is an extension of this strategy.”She added, “Among our current pipeline, we view prostate cancer as the area closest to commercialization. We will continue to expand our scope of application, focusing on cancer types with high unmet medical needs.”
- Company
- GE HealthCare rebounded with 300B won sales last year
- by Hwang, byoung woo Apr 20, 2026 04:58pm
- GE HealthCare, which is pushing for an "Integrated Precision Healthcare" that combines hardware with digital solutions, successfully led a sales rebound last year.Following the medical-government conflict that hit the industry in 2024, the expansion of communication with hospitals and clinics in South Korea via digital platforms is cited as a key factor in improving performance.After the 2024 medical-government crisis…V-shaped rebound in one year According to recently released audit reports, GE HealthCare Korea's sales in 2025 reached KRW 317.1 billion, a 14.9% increase from KRW 275.8 billion the previous year.After sales dropped 6.8% in 2024 to KRW 295.9 billion from 2023, the company quickly surpassed the KRW 300 billion the following year, signaling a significant recovery.Operating profit also increased by 13.4%, from KRW 16.86 billion in 2024 to KRW 19.13 billion in 2025. During the same period, net income rose from KRW 14.57 billion to KRW 17.09 billion. However, the operating profit margin remained stable at around 6.0% (6.1% in 2023, 6.1% in 2024, and 6.0% in 2025), indicating steady maintenance rather than a drastic improvement in profitability.GE HealthCare Korea's 4-Year Sales & Operating Profits: GE HealthCare Korea's sales in 2025 reached KRW 317.1 billion. Source: FSS DART, unit: KRW 100 million (Audit report re-graphed by Daily Pharm)As the medical-government conflict issues gradually resolve, the company's efforts to strengthen capabilities and improve its internal structure have been proven through these figures, suggesting a structural rebound rather than a simple recovery.In 2024, the decline in sales was largely due to a decrease in product sale, which dropped from KRW 193.8 billion to KRW 170.8 billion.In contrast, service sales increased from KRW 102.1 billion to KRW 104.9 billion during the same period, acting as a buffer while equipment sales were shaken.In 2025, both products and services rebounded simultaneously. Product sales reached KRW 203.9 billion, and service sales rose to KRW 113.2 billion.GE HealthCare Korea's Product Sales & Service Sales Ratio: GREEN-product sales, BLUE-service sales. Source: FSS DART, Unit: KRW 100 million. (Audit report re-graphed by Daily Pharm)GE HealthCare strengthens platform strategy…Expanding hospital collaborationsGE HealthCare is shifting its weight from a structure centered on equipment sales to a platform-based strategy.The core of this is the 'D3 Strategy,' which encompasses Device, Disease, and Digital. It aims to use AI to improve quality, support customized treatment, and ultimately integrate data into a cloud-based platform.Globally, GE HealthCare has obtained FDA clearance for 100 AI-enabled medical devices, setting a record for the most listings in four consecutive years.A representative technology, 'AIR Recon DL,' which offers both reduced MRI scan times and improved image quality, has been introduced to more than 20 medical institutions in Korea.SPI index displayed on the CARESCAPE CanvasAdditionally, in February, the Surgical Pleth Index (SPI), a monitoring indicator that quantifies pain responses during surgery, was listed as a "New Medical Technology." This expanded the company's presence beyond imaging into the operating room and patient monitoring sectors.The technology portfolio is shifting from simple imaging equipment to clinical decision-support tools.Consequently, partnerships with major hospitals are expanding beyond simple supply contracts into a system of "Reference Sites" and cooperative hospital networks.For example, Dongtan City Hospital was designated as an Asian regional reference site in January. Its AI Radiology Center serves as a hub where customers from Korea, ASEAN, Australia, and New Zealand can refer to operational cases.Gumi Gangdong Hospital signed an MOU for clinical cooperation and equipment operational efficiency. Daegu Miracle Women's Clinic is partnering to advance infertility treatment based on high-end ultrasound technology.These structures integrates everything from equipment supply to operational training, protocol sharing, and the expansion of clinical utilization into a single package.Yong-duck Kim, President & CEO of GE HealthCare Korea, stated, "The designation of Dongtan City Hospital as an Asian regional reference site signifies that the equipment operation and clinical environment of medical institutions in South Korea have been recognized as excellent global reference cases," and added, "GE HealthCare will continue to actively support Dongtan City Hospital in maintaining efficient and stable equipment operation and building a sustainable medical environment."
- Company
- Pfizer Korea’s dividend returns to ₩12.48 million
- by Chon, Seung-Hyun Apr 20, 2026 04:58pm
- Pfizer Korea paid a dividend of KRW 12.48 million to its parent company last year. While the company paid KRW 160 billion in dividends in 2023 and 2024 as earnings surged due to pandemic-related benefits, it returned to its “20% of preferred stock capital” dividend policy after 3 years, resulting in a dividend payout ratio of just 0.06%.According to the Financial Supervisory Service on the 18th, Pfizer Korea’s dividends paid to its parent company last year totaled KRW 12.48 million. The dividend payout ratio was 0.06%. This means that only 0.06% of last year’s net profit of KRW 19.8 billion was distributed to the parent company.Pfizer Korea’s dividend for last year was reduced by 99.98% compared to the previous year’s KRW 60.01 billion. In 2023, Pfizer Korea paid out KRW 100.012 billion in dividends. This is the first time in 3 years, since 2022, that Pfizer Korea has set its dividend at KRW 12.48 million.Pfizer Korea’s annual revenue (left) and operating profit (right) (Unit: KRW 100 million, Source: Financial Supervisory Service)Pfizer Korea’s largest shareholder is Pfizer’s Dutch subsidiary, ‘PF OFG South Korea 1 B.V.’, which holds a 99.99% stake.Pfizer Korea has maintained a unique dividend policy of keeping the annual dividend at KRW 12.48 million. From 2018 to 2022, it paid the same dividend of KRW 12.48 million for 5 consecutive years.Pfizer Korea calculated the dividend by applying a 20% dividend payout ratio to the preferred stock capital. Pfizer Korea’s total capital stock is KRW 922.92 million. Of this, common stock (172,104 shares) accounts for KRW 860.52 million, and preferred stock (12,480 shares) accounts for KRW 62.40 million. A dividend of KRW 12.48 million , 20% of the KRW 62.40 million in preferred stock capital, was set.The high dividends in 2023 and 2024 are analyzed as a result of significantly improved performance during the COVID-19 pandemic.Pfizer Korea recorded revenue of KRW 391.9 billion in 2020, which increased more than fourfold to KRW 1.694 trillion in 2021, and surged to KRW 3.2254 trillion in 2022, marking more than an eightfold increase over two years. Revenue in 2023 decreased to KRW 1.6018 trillion, about half of the previous year, but this figure is still more than four times higher than in 2020.Pfizer Korea recorded an operating loss of KRW 7.2 billion and a net loss of KRW 21.2 billion in 2020, but in 2022, operating profit and net profit reached KRW 120.1 billion and KRW 119.5 billion, respectively. In 2023, operating profit was KRW 59.2 billion and net profit was KRW 84.9 billion.Pfizer partnered with Germany’s BioNTech in March 2020, when COVID-19 began spreading globally, to develop an mRNA vaccine. The combination of BioNTech’s mRNA technology and Pfizer’s extensive global clinical experience created synergy.Pfizer successfully developed the Comirnaty vaccine with 95% efficacy in less than a year after the outbreak. It received emergency use authorization from the U.S. FDA in December 2020 and was approved in Korea in March of the following year, leading to full-scale supply through its Korean subsidiary. Pfizer also successfully developed the COVID-19 treatment Paxlovid, which the Korean government procured and used.In 2023 and 2024, annual dividends were maintained at KRW 12.48 million, but interim dividends of KRW 100 billion and KRW 60 billion were additionally paid, respectively. Dividend payout ratios reached 117.76% and 168.17% for 2023 and 2024.As performance declined after the COVID-19 pandemic phase, the company appears to have returned to its previous dividend policy. Pfizer Korea’s revenue in 2024 was KRW 783.7 billion, down 75.7% from two years prior, and operating profit decreased by 77.4% from KRW 120.1 billion to KRW 27.2 billion. Last year’s revenue and operating profit were KRW 586.1 billion and KRW 6 billion, respectively, decreasing by 25.2% and 77.9% year-on-year. Compared to 2022, revenue dropped by 81.9% and operating profit by 95.0%Over the past 20 years since 2005, Pfizer Korea has paid dividends of KRW 12.48 million based on the “20% preferred stock” rule in all but four years.In 2017, the dividend was set at KRW 79.794 billion, exceeding net profit. At the time, both common and preferred shares had a dividend rate of 660% of par value, increasing the total payout. In 2008, a dividend of KRW 190 billion was set. Despite recording a loss of KRW 600 million that year, the company set a dividend payout ratio of 3,045% relative to par value.
- Company
- 'Fruzaqla' passes the Cancer Disease Review Committee
- by Eo, Yun-Ho Apr 20, 2026 04:58pm
- Product photo of FruzaqlaInterest is drawn to the health insurance reimbursement listing process for 'Fruzaqla,' a new treatment for colorectal cancer.According to industry sources, Takeda Pharmaceuticals Korea's Fruzaqla (fruquintinib), a colorectal cancer treatment that selectively inhibits Vascular Endothelial Growth Factor Receptor (VEGFR) 1, 2, and 3, is currently under discussion at the subcommittee level. This drug passed the Cancer Disease Review Committee of the Health Insurance Review and Assessment Service (HIRA) last March.It is to be seen whether Fruzaqla will pass the Pharmaceutical Reimbursement Evaluation Committee, the final hurdle at the HIRA stage, this year to become a standard treatment option for colorectal cancer.Approved in South Korea in March of last year, Fruzaqla is an innovative anti-cancer drug previously designated as both an Orphan Drug and the item for the Global Innovative products on Fast Track (GIFT) program.The specific indication for this drug is: "The treatment of adult patients with metastatic colorectal cancer (mCRC) who have been previously treated with fluoropyrimidine-, oxaliplatin-, and irinotecan-based chemotherapy, an anti-VEGF therapy, and, if RAS wild-type, an anti-EGFR therapy."Currently, Fruzaqla remains a non-reimbursed drug. Takeda submitted an application for reimbursement to the health authorities last year and is currently proceeding through the listing steps. It remains to be seen if Fruzaqla will successfully secure reimbursement to allow for seamless prescribing to patients.The clinical utility of Fruzaqla was confirmed through the FRESCO and FRESCO-2 Phase 3 studies.Results from these trials demonstrated that Fruzaqla extended the median overall survival (mOS) to 9.3 months in previously treated metastatic colorectal cancer patients, a 2.7-month increase compared to the placebo group. Furthermore, it reduced the risk of death by 35%.In addition to its clinical efficacy, Fruzaqla is an oral medication taken once daily without complex dietary restrictions. This convenience is expected to have a positive impact on patients' quality of life alongside its therapeutic effects.Professor Dong-hoe Koo of the Department of Oncology at Kangbuk Samsung Hospital stated, "Fruzaqla has high drug specificity because it does not target unnecessary receptors. This allows for efficient VEGFR inhibition and continuous drug exposure. Its potential for combination therapy with existing agents is also worth evaluating in future clinical settings."Meanwhile, colorectal cancer refers to malignant tumors occurring in the colon and rectum, categorized into colon cancer or rectal cancer depending on the location. Metastatic colorectal cancer (mCRC) occurs when the tumor spreads beyond the primary site to invade other organs.As of 2022, the incidence rate of colorectal cancer in South Korea was 61.1 per 100,000 people, ranking second to thyroid cancer. Notably, the incidence rate among the younger population aged 40 and under stands at 12.9 per 100,000, the highest in the world. It poses a serious public health situation.
- Company
- Multinational pharma employees in Korea earn over ₩100M on average
- by Son, Hyung Min Apr 20, 2026 04:58pm
- Last year, the average annual salary of employees at Korean subsidiaries of major multinational pharmaceutical companies exceeded KRW 100 million. Boehringer Ingelheim Korea and Viatris Korea were found to have average salaries reaching KRW 150 million.According to an analysis of audit reports from 27 multinational pharmaceutical subsidiaries disclosed by the Financial Supervisory Service, the average employee salary was KRW 114.37 million. Of these, 21 companies paid average annual salaries of KRW 100 million or more, indicating that a high-salary structure has spread across the industry.This average salary was calculated based only on salary items listed in audit reports, excluding welfare benefits, bonuses, incentives, or retirement benefits. Additionally, there may be some discrepancy between this figure and the actual perceived annual salary due to differences in accounting treatment of labor costs across companies.Among the companies surveyed, Boehringer Ingelheim Korea recorded the highest average salary. The company paid a total of KRW 24.94 billion in salaries last year, an 8.8% increase year-over-year. The average salary per employee rose from KRW 139.8 million in 2024 to KRW 157.86 million last year, an increase of KRW 18.06 million.Viatris Korea ranked second with KRW 145.26 million per employee. This represents an increase of approximately KRW 10 million compared to the previous year’s average of KRW 135.87 million. The company’s total payroll for last year was KRW 13.28 billion, a 14.5% increase from 2023.Gilead Sciences Korea paid an average of KRW 139.94 million per employee, slightly down from KRW 141.32 million in 2024. Total salary payments decreased by 2.4% to KRW 12.97 billion.Pfizer Korea’s average salary was KRW 127.42 million, about KRW 1 million higher than in 2023.However, total salary expenses fell 6.3% to KRW 47.78 billion. This increase in the average annual salary per employee is attributed to a decline in the number of employees from 404 in 2024 to 375 last year.Pfizer has operated a voluntary retirement program since the end of the pandemic. Severance pay, which serves as a form of compensation, surged by 202.0% from KRW 5.33billion in 2024 to KRW 16.11 billion last year, reflecting changes in the labor cost structure.In addition, Sanofi-Aventis Korea, GSK, Ferring Korea, UCB Korea, Amgen Korea, Janssen Korea, Novo Nordisk, AbbVie Korea, Lundbeck Korea, Abbott Korea, Galderma Korea, AstraZeneca Korea, Handok Teva, and Servier Korea all paid employee salaries exceeding KRW 100 million.Merck Korea’s total salary rose to KRW 45.38 billion, an 8.2% increase from KRW 41.96 billion in 2024. However, the average annual salary per employee was KRW 80.96 million, a 7.2% decrease from the KRW 87.2 million in the previous year. This decrease in the average per-employee figure was due to the increase in the number of employees from 481 to 579.For BMS Korea, both total salary and retirement benefit expenses increased. The company’s total salary last year amounted to KRW 24.9 billion, an 8.4% increase from the previous year. During the same period, retirement benefit expenses rose by 169.4%, from KRW 3.3 billion to KRW 8.8 billion.BMS Korea implemented an ERP program for some employees last year. Consequently, the number of employees reported in the audit report decreased slightly from 209 to 190. The company’s average annual salary for executives and employees was KRW 130.58 million last year, ranking fifth among the surveyed companies.
- Policy
- Ginkgo biloba ·calcium dobesilate ·silymarin set for reimb re-eval this month
- by Jung, Heung-Jun Apr 17, 2026 09:03am
- Ginkgo biloba extract, calcium dobesilate hydrate, and silymarin are set to be discussed for the re-evaluation of reimbursement appropriateness during this month’s Health Insurance Policy Deliberation Committee (HIDC) meeting.Three ingredients, including the two reviewed by the Pharmaceutical Reimbursement Evaluation Committee (PREC) in January and silymarin (added this month), are expected to be considered. The HIDC's final decision process, which had been somewhat delayed following the drug pricing system reform, is anticipated to be finalized this month.According to industry sources on the 15th, the ingredients for this year's reimbursement re-evaluation are highly likely to pass through the HIDC subcommittee today (the 16th) and be presented at the plenary session on the 23rd.The list of ingredients for re-evaluation differs slightly from those discussed in last year's HIDC subcommittee. At that time, seven ingredients were under discussion, including kallidinogenase, meglumine gadoterate, diacerein, afloqualone, and octylonium bromide, as well as ginkgo biloba and calcium dobesilate.However, as the drug pricing system reform plan, which included a new set of criteria for appropriateness re-evaluation, passed the HIDC last November, re-discussions were held this year based on these updated standards.The requirements for claim amounts and registration in specific countries have been removed. The selection criteria have been changed tov include ▲ingredients for which health authorities in A8 countries have initiated clinical or reimbursement appropriateness re-evaluations ▲cases where data or clinical evidence contradicting previously reported efficacy has been published ▲drugs for which academic societies or experts have recommended a re-evaluation.Accordingly, ginkgo biloba and calcium dobesilate were reviewed as re-evaluation targets by the PREC in January. Typically, these should have been finalized at the January HIDC meeting following PREC approval, but the HIDC consideration was delayed alongside the postponement of the drug pricing reform.In the meantime, silymarin was added to the list of targets for review at this month's PREC meeting. Although a court had previously ruled to cancel the notification of silymarin's removal from the reimbursement list following an administrative lawsuit by pharmaceutical companies, the ruling was limited to procedural issues, leaving re-evaluation likely.The Health Insurance Review and Assessment Service (HIRA) plans to proceed with the re-evaluation again, this time acknowledging the literature previously pointed out by the court. Consequently, silymarin, which underwent reimbursement re-evaluation in 2021, will return to the evaluation table alongside ginkgo biloba and calcium dobesilate.Due to the government's reform of the drug pricing system, the follow-up measures following a reimbursement re-evaluation have been simplified to either exclusion from reimbursement or selective reimbursement. The bypass that allowed companies to maintain reimbursement by voluntarily lowering drug prices to demonstrate cost-effectiveness relative to alternative drugs has effectively been blocked.
- Policy
- New reimb criteria established for growth hormone Sogroya
- by Jung, Heung-Jun Apr 17, 2026 09:03am
- The government is establishing new reimbursement criteria following the new listing of growth hormone treatments. Additionally, self-injectable formulations of severe asthma treatments have been newly added to the list, allowing for switching between formulations.On the 16th, the Ministry of Health and Welfare issued an administrative notice regarding the amendment to the “Detailed Provisions on the Application Criteria and Methods for Medical Care Benefits.” The amendment is scheduled to take effect on May 1.Novo Nordisk’s Sogroya prefilled pen (somapacitan) will have new reimbursement criteria established for growth hormone deficiency in both pediatric and adult patients.The drug is administered at a maximum of 8 mg per week in accordance with the approved indications, and patients must bear the full cost if they exceed specific height criteria (153 cm for females, 165 cm for males). Coverage may be extended if growth hormone deficiency is confirmed after 3 years of treatment. Self-injection prescriptions are also permitted.Reimbursement criteria have also been newly established for emergency imported drugs supplied through the Korea Orphan & Essential Drug Center. The skeletal muscle relaxant Takeda Lysthenon Inj is now covered for use during general anesthesia to alleviate spasms associated with endotracheal intubation or electroconvulsive therapy (ECT).For the anti-atherosclerotic agent Epic Cholestyramine Powder for Susp, coverage criteria have been established solely for the purpose of 'Reducing serum cholesterol levels’ within the scope of approval by the Ministry of Food and Drug Safety. However, patients must bear the full cost when the drug is administered for other purposes, such as treating pruritus.Reimbursement criteria have been established for the vasodilator Viatris Isoptin Inj for the treatment of angina and arrhythmia, and Centa 5-HT-P Cap is now covered for the treatment of patients with BH4-deficient phenylketonuria. For GSK’s severe asthma treatment Nucala, the self-injectable autoinjector formulation Nucala Autoinjector was added, allowing switching with the existing formulation.Additionally, for Nucala Autoinjector, long-term prescriptions of up to 24 weeks are permitted for patients who have shown stable disease activity and no side effects 6 months after the initial dose.’With the listing of new combination drugs for diabetes and hypertension/dyslipidemia, general reimbursement principles have been partially revised. The empagliflozin + linagliptin combination has been added for diabetes with the new listing of K-Glito Tab, and the fimasartan + atorvastatin + ezetimibe combination has also been added for hypertension and hyperlipidemia with the new listing of Kanarbzet Tab.Gliptide Tab, which failed to demonstrate usefulness during the Ministry of Food and Drug Safety’s clinical reevaluations, will be removed from the reimbursement list.
- Opinion
- [Reporter's View] NHI "red light", societal consensus needed
- by Lee, Jeong-Hwan Apr 17, 2026 09:03am
- South Korea’s National Health Insurance (NHI) finances are expected to enter a deficit in 2026. While the fund was in surplus for five consecutive years since 2021, the end of the COVID-19 pandemic, the transition to endemic status, and the onset of a super-aged society have led to a sharp increase in medical expenditures, causing the surplus to plummet annually.The government has established and implemented various policies to ensure the soundness and sustainability of NHI finances. One example is the reduction of pharmaceutical costs through generic drug price cuts, which recently sparked a conflict with the domestic pharmaceutical industry.However, health insurance experts argue that the government must resolve larger fundamental issues rather than focusing on administrative measures to create fiscal room through drug price cuts.While the increase in medical utilization and benefit expenditures due to a super-aged population is an unavoidable natural growth factor, experts criticize that the failure to aggressively resolve distortions in the medical delivery system or the lack of measures to eradicate excessive medical care ("medical shopping") is a man-made disaster that exacerbates the problem through inadequate administration despite being preventable.Furthermore, health insurance experts view the solutions for securing the soundness of NHI finances as having been consistently discussed as an agenda for over a decade, and the correct answers are already somewhat determined.Ultimately, this means that procedures must be established for the government, medical providers (doctors), and medical consumers (patients and the public) to form a consensus and agree on a policy direction that boldly lowers or suspends the benefit coverage rate for excessive medical care and overhauls the payment system centered on the fee-for-service model from its roots.Of course, there is a high probability that opinions will differ regarding the priorities for allocating NHI finances. However, before coordinating such detailed disagreements, the government should take the lead in establishing societal consensus.The etymology of "consensus" originates from the Latin words con- (together) and sentire (to feel). It refers to members of society sharing common feelings and thoughts.NHI finances are a social insurance system operated by the state with public consent, and the key lies in how to create, maintain, and distribute limited public resources. Ultimately, it means that the NHI can be operated without conflict only when it is based on consensus.At this crossroads, where NHI finances are being depleted and the deficit is set to increase significantly, the Ministry of Health and Welfare (MOHW), the ministry in charge, must engage in bold, active administration. This is necessary so that the entire public and medical providers can share the common view that NHI sustainability must be significantly strengthened before social conflict is triggered by fiscal deterioration.This implies that the Ministry of Health and Welfare (MOHW) should take the lead in improving the perception that a "red light" has been turned on for NHI finances and that all members of our society must share some of the burden to solve the problem.
- Policy
- Free HPV vaccination for 12-year-old boys to begin in May
- by Lee, Jeong-Hwan Apr 17, 2026 09:03am
- Starting this May, 12-year-old boys will also be eligible for free human papillomavirus (HPV) vaccinations.The Korea Disease Control and Prevention Agency announced on the 16th that it will launch a new national HPV vaccination support program targeting 12-year-old male adolescents (born in 2014) starting May 6.With this change, the existing HPV free vaccination program for female adolescents aged 12–17 and low-income women aged 18–26 will now include male adolescents, who will receive two HPV vaccine doses at six-month intervals.Starting with those born in 2014 this year, those born in 2015 will be newly supported next year. Even if those born in 2014 do not complete both doses this year, they will still be eligible for free vaccination next year.The KDCA plans to expand the program gradually, broadening the target group by one age group each year.HPV vaccination is included in national immunization programs in 147 countries worldwide, including 37 of the 38 member nations of the Organization for Economic Cooperation and Development (OECD).According to the U.S. Centers for Disease Control and Prevention (CDC), 90% of cervical cancers and 70% of anogenital and oropharyngeal cancers are caused by HPV infection. The KDCA believes that expanding free HPV vaccination will help prevent related diseases such as cervical cancer, anal cancer, genital warts, and anal intraepithelial neoplasia.The KDCA maintains that since HPV can infect not only women but also men, and since the HPV vaccine’s efficacy in preventing related diseases has been confirmed in men as well, vaccination can reduce future infections and the incidence of related diseases.The Advisory Committee on Immunization also determined that HPV vaccination is effective in preventing diseases such as genital warts and anal cancer in men. Based on domestic and international research and vaccination experience, which have confirmed the safety and efficacy of the HPV vaccine, the agency recommended expanding support for HPV vaccination among male adolescents.Eligible individuals can visit a nearby designated medical institution or public health center to receive the HPV vaccine (quadrivalent HPV vaccine) free of charge. The KDCA plans to provide information on medical institutions through the Vaccination Assistant website and support vaccination history tracking and management.Commissioner Seung-Kwan Lim stated, “HPV vaccination is an effective means of preventing future cancers and related diseases. We hope that this expansion of the target group will enable more adolescents to receive the vaccination in a timely manner.”