- LOGIN
- MemberShip
- 2026-05-14 23:06:14
- Medipost's Cartistem Phase 3 clinical trial succeeds in Japan
- by Cha, Ji-Hyun | translator Hong, Ji Yeon | 2026-05-14 09:28:19
Medipost has met all dual primary endpoints in its Japanese Phase 3 clinical trial for Cartistem, a stem cell therapy for knee osteoarthritis. By securing statistical significance in both subjective symptom improvement and arthroscopic cartilage regeneration compared to the active control group, the company’s efforts for commercialization in Japan and the advancement of its U.S. Phase 3 trials are expected to speed up.
On the 13th, Medipost held a press conference at the Four Seasons Hotel in Jongno-gu, Seoul, to disclose the major results of the Japanese Phase 3 trial for Cartistem and its future global development strategy. Key attendees included Medipost CEO Wonil Oh, Global Business Head and CEO of Medipost K.K. (Japan) and co-CEO of Medipost Inc. (USA) Seung Jin Lee, and Vice President Hoon Sik Cho.

CEO Wonil Oh remarked, "This achievement is a major achievement of Medipost's journey since its founding, driven by the goal of improving patients' quality of life through stem cell therapy innovation," and added, "We will achieve entry into the Japanese market for Cartistem and complete the ongoing U.S. Phase 3 trial to emerge as a global leader in the stem cell industry."
Cartistem is an allogeneic umbilical cord blood-derived mesenchymal stem cell therapy developed by Medipost. It is a treatment aimed at regenerating damaged cartilage and improving pain and function in patients with knee cartilage defects caused by degenerative changes or repeated trauma. Since receiving marketing authorization from the Ministry of Food and Drug Safety (MFDS) in 2012, it has accumulated over 10 years of prescription experience. Cartistem recorded sales of KRW 19.5 billion last year.
Medipost conducted Japanese clinical trials to overcome domestic market growth limitations and secure its first overseas commercial hub. The Japanese Phase 3 trial was designed as a pivotal study for Japanese marketing authorization. Based on its experience in authorization and commercialization, as well as existing clinical data accumulated in Korea since 2012, Medipost was able to skip Phase 1 and 2 trials in Japan and enter directly into Phase 3.
The trial was a randomized, active-controlled comparative study conducted at 13 medical institutions in Japan involving 130 patients with knee osteoarthritis. The actual administration and surgery groups consisted of 59 patients in the Cartistem group and 61 in the hyaluronic acid (HA) group. Patients were followed for 52 weeks after administration to observe efficacy and safety.
The control group received a hyaluronic acid injection, which is the standard of care for patients with knee osteoarthritis in Japan. Global Business Head Seung Jin Lee explained, "In the process of discussing the clinical design with the Japanese Pharmaceuticals and Medical Devices Agency (PMDA), we reflected the fact that Japanese patients generally receive HA injections for knee osteoarthritis treatment and set that product as the control."
The primary subjects were patients with knee osteoarthritis aged 20 to 80. The radiographic severity of osteoarthritis corresponded to K&L grades 2 to 3, and the degree of cartilage damage corresponded to ICRS grades 3 to 4. The size of the cartilage defect ranged from approximately 2 to 9 square centimeters, and the trial included patients with a pain score of 40 or higher on a 100mm Visual Analog Scale (VAS) and a Body Mass Index (BMI) of less than 35. Furthermore, the study targeted patients who had not shown sufficient symptom improvement despite receiving existing treatments, such as exercise therapy, physical therapy, or hyaluronic acid injections, for at least 3 months.
According to the presentation, Cartistem met both dual primary endpoints in the Japanese Phase 3 trial. The dual primary endpoints were ▲the change in the WOMAC total score ▲the improvement rate of one or more ICRS grades. WOMAC is a patient-reported outcome that evaluates knee pain, functionality, and stiffness as perceived by the patient. ICRS is a structural evaluation metric used to confirm whether the degree of cartilage damage has actually improved through arthroscopy. The design required both metrics to be met for the trial to be considered a success.
Lee explained, "Since Cartistem is administered via surgery and HA via injection, patients inherently know which treatment they received, which can allow for a placebo effect in patient-reported metrics," and added, "To compensate for this, we included the ICRS evaluation, which directly checks the cartilage state via arthroscopy at the 52-week mark, as a co-primary endpoint. To ensure the treatment group information remained undisclosed, evaluations were conducted by a separate central adjudication committee after blinding the data."
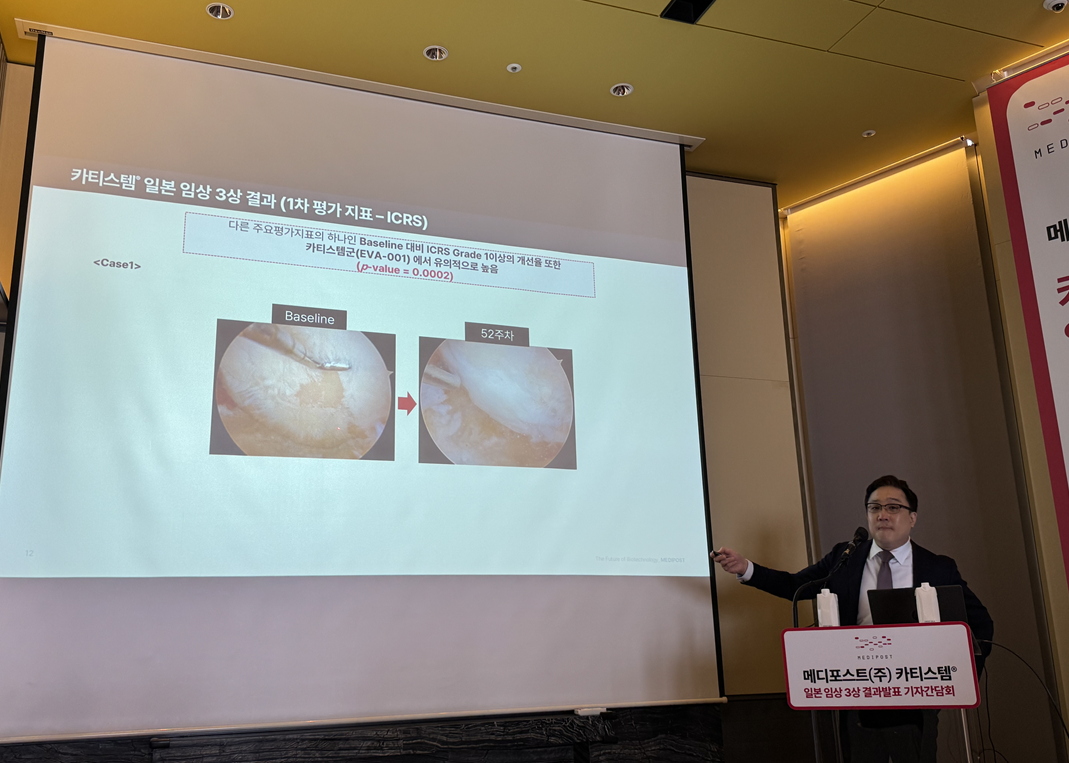
Clinical results showed that Cartistem demonstrated statistically significant superiority in changes in the WOMAC total score compared with the HA group. The WOMAC p-value was reported as <0.0001. Cartistem also showed a significant improvement over the active control group in the rate of improvement in one or more ICRS grades, with an ICRS p-value of 0.0002.
Consistent results were also confirmed in the secondary endpoints. The VAS for pain level and the IKDC and KOOS for knee functionality all recorded p-values of less than 0.0001. Significant improvements were also observed across all WOMAC sub-items, including pain, functionality, and stiffness.
"This Japanese Phase 3 study was designed so that it had to meet both dual primary endpoints," Lee said. "It is highly significant in that we confirmed not only the improvement in pain and function felt by the patient but also the regeneration of cartilage via arthroscopy," and added, "In the U.S. or Europe, a design that performs arthroscopy on patients again is not easy due to the high patient burden and ethical considerations, as it involves anesthesia and invasive procedures. Therefore, the ICRS data secured in the Japanese Phase 3 will be highly valuable data to be utilized overseas."
Medipost plans to apply for Japanese marketing authorization in the second half of this year based on these clinical results. Given the PMDA review period, the company aims to obtain approval by the end of 2027. It plans to pursue a launch in the Japanese market, followed by subsequent processes for price setting and insurance reimbursement.
The company expressed strong confidence regarding the possibility of Japanese marketing authorization. Lee stated, "Due to the nature of cell and gene therapies, CMC (which refers to manufacturing process management and quality consistency), inspections of our GMP facility in Guro, Korea, and our local production partner's facility in Japan, remain." However, Lee said, "After reviewing these results with external experts, including advisory panels and former PMDA officials, we received the opinion that with clinical results indicate there should be no major issues in gaining marketing authorization."
The local partner, Teikoku Pharma, will handle sales in Japan. Previously, Medipost signed an exclusive distribution agreement for Cartistem in Japan with Teikoku Pharma in December last year. The company has already received a non-refundable upfront payment of USD 8 million, with an additional USD 10 million payable upon marketing authorization. Lee explained, "The marketing authorization holder will be Medipost K.K., the Japanese subsidiary of Medipost, and Medipost will also manage the responsibility for manufacturing and shipping. Teikoku Pharma will be responsible for transportation, sales, and marketing within Japan."
Considering the price-setting and insurance reimbursement procedures following Japanese marketing authorization, Medipost is also conducting a Real-World Evidence (RWE) study in South Korea. The company explained that it has secured data from approximately 550 patients who underwent Cartistem procedures at 13 Korean medical institutions, with an average follow-up period of about 6.9 years.
"Since Cartistem received marketing authorization in Korea in 2012, approximately 36,700 patients have undergone surgery as of the end of last month," Lee emphasized. "Among them, we are collecting real-world evidence data targeting patients who have passed at least three years since their procedure."
Lee continued, "The endpoint we consider most important is the rate of conversion to total knee artificial joints. From the data of 550 patients secured so far, we have identified that less than 1% of patients have converted to artificial joints." He added that detailed information would be disclosed at the end of the year. The company intends to use this data as reference material for the Japanese marketing authorization application and as evidence during the price-setting and insurance reimbursement negotiations with the Ministry of Health, Labor and Welfare after approval.
Lee noted, "In Japan, the process of price setting and insurance reimbursement with the Ministry of Health, Labour and Welfare proceeds after marketing authorization. How many years a patient maintains the effect after receiving Cartistem, and the rate at which they transition to other treatments or artificial joint surgery, can be vital data for economic evaluations," and stated, "This real-world evidence study will serve as an important basis not only for Japan but also for discussions with insurance companies in the U.S. after obtaining approval there."
Regarding the company's U.S. Phase 3 clinical trial, patient enrollment is currently underway following the U.S. Food and Drug Administration (FDA) approval of the IND application earlier this year. The company plans to use the results of the U.S. Phase 3 trial, along with the Japanese Phase 3 data and Korean RWE, as materials for the U.S. marketing authorization application.
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.
- [Reporter's View] The dark side of improved diabetes med convenience
- Reporter's view | Son, Hyung Min
- [Reporter's View] Contradiction of "K-passing" and a new drug powerhouse
- Reporter's view | Lee, Jeong-Hwan






