- LOGIN
- MemberShip
- 2026-05-13 11:45:43
- Curacle resumes licensing-out activities after 2 years
- by Cha, Ji-Hyun | translator Alice Kang | 2026-05-13 09:10:30
Curacle, a biotech company specializing in new drug development, has successfully licensed out a preclinical-stage bispecific antibody candidate in a deal valued at over KRW 1 trillion. This marks its first licensing-out achievement in 2 years, following a notice of rights termination regarding a retinal disease treatment from a French ophthalmology-focused pharmaceutical company.
The deal is expected to significantly reduce Curacle’s risk of being designated as an administrative issue stock due to revenue requirements. However, concerns remain due to limited disclosure about the counterparty.
MT-103 global deal worth KRW 1.5 trillion… Curacle’s share KRW 781.8 billion
According to filings with the Financial Supervisory Service, Curacle recently signed a global licensing agreement with US-based Memento Medicines for the development and commercialization of its bispecific antibody candidate ‘MT-103.’ Under the agreement, Memento gains exclusive worldwide rights to develop, manufacture, and commercialize MT-103.
The total deal value is USD 1.077 billion (KRW 1.56 trillion). Of this, the non-refundable upfront payment is USD 8 million (0.7%), with USD 82.25 million in development and approval milestones and up to USD 987.5 million in royalties upon commercialization.
Curacle and MabTics will share revenues 50:50 under a joint R&D agreement. Reflecting this, Curacle’s share amounts to USD 538.875 million (KRW 781.8 billion). The company’s upfront payment share is USD 4 million.
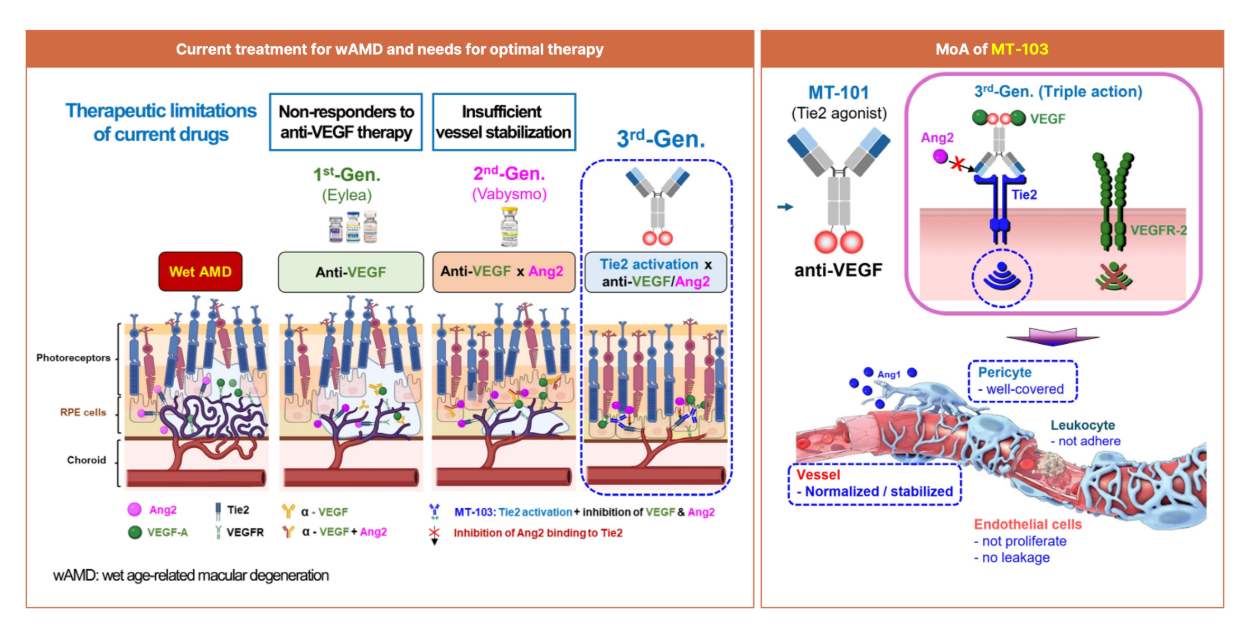
MT-103 is a bispecific antibody candidate for retinal diseases co-developed by Curacle and MabTics. Curacle secured the antibody pipeline through a partnership signed in June 2023 and then signed a joint R&D agreement in July of the following year. MT-103 is one of the key antibody pipeline assets secured through this process.
MT-103 is a bispecific antibody that combines an antibody that inhibits vascular endothelial growth factor (VEGF), which induces angiogenesis, with an antibody that activates the Tie2 receptor, which stabilizes blood vessels. While the existing bispecific antibody therapy ‘Vabysmo’ indirectly induces Tie2 activation by inhibiting VEGF and angiopoietin-2 (Ang-2), MT-103 is designed to directly activate Tie2. The company explains that this is expected to deliver differentiated therapeutic effects in patient groups that do not respond sufficiently to existing anti-VEGF therapies or Vabysmo.
Preclinical data presented at ARVO 2026 by Curacle and MabTics, MT-103 induced Tie2 receptor phosphorylation and inhibited VEGF-induced signaling in cell experiments. In endothelial cell-based vascular leakage assays, it also demonstrated an effect of reducing vascular permeability. In animal models, the drug was shown to reduce pathological neovascularization, inhibit vascular leakage, improve retinal vascular remodeling, and alleviate inflammatory responses.
First deal since CU06 return… expected to ease revenue-related administrative issue risk
This deal marks Curacle’s first licensing success since its previous retinal therapy candidate CU06 was returned in 2024 by a French ophthalmology-focused pharmaceutical company. In May 2024, Curacle received a notice of termination of the technology export agreement and a return of rights regarding the candidate drug ‘CU06’ for the treatment of diabetic macular edema and wet age-related macular degeneration from Théa Open Innovation, a French ophthalmology-focused pharmaceutical company.
The contract, signed in October 2021 for a total of USD 163.5 million, transferred the global development and commercialization rights for CU06-RE (excluding Asia) to Théa. Although this contract was effectively Curacle’s only technology export achievement, it became void following the return of rights. This MT-103 agreement is considered significant as it marks Curacle’s first technology export achievement since then.
With this agreement, Curacle is expected to fill the revenue gap that had effectively been left open. Curacle’s revenue has plummeted over the past 3 years. The company’s revenue, which stood at KRW 10.3 billion in 2023, fell by 84.5% to KRW 1.6 billion in 2024. Last year, revenue amounted to just KRW 7.1 million as the recognition of subsequent revenue was halted due to the termination of the CU06 technology export agreement. In effect, this means there was no revenue generated from its core business of new drug development.
Consequently, the risk of being designated as an administrative issue stock due to revenue requirements has been largely alleviated. The company, which went public in 2021, saw the grace period for the designation criteria, triggered by revenue falling below KRW 3 billion, expire last year. The company faced the possibility of being designated as an administrative issue stock if it failed to meet this year’s revenue requirements; however, if the KRW 5.8 billion upfront payment from this contract is received as planned, that burden is expected to be significantly reduced.
The company has also moved to secure a separate revenue base. Following the establishment of an API business division last year, the company completed a merger-by-absorption with the API firm Daesung Pharmtech in January of this year. A Curacle official stated, “We expect to receive the upfront payment for this contract at a time similar to that of a typical technology export contract. We will be able to meet the revenue requirements without issue through the absorption merger of the API that the company completed earlier this year.”
Information on the contracting party is overly limited… in stark contrast with the D&D Pharmatech-Metsera case
However, some market observers have expressed skepticism regarding the substance of this agreement due to the limited information available about the counterparty. In its public disclosure, the company only stated that the counterparty is a US-based firm named Memento, without providing any further details. Basic information about Memento, such as its date of establishment, location, representative, and investors, cannot be verified, and the company does not even have a separate website.
The company maintains that Memento is a NewCo-type entity established for the development of a specific pipeline and that it cannot disclose information on investors or management due to confidentiality clauses in the contract. The NewCo model involves establishing a separate new corporation for the purpose of developing a specific new drug candidate, attracting external investment, and pursuing clinical development and commercialization. Typically, such entities operate in “stealth mode” in the early stages, with confidentiality clauses preventing disclosure.
A Curacle official stated, “The company cannot arbitrarily disclose information designated as confidential in the contract. It is a fact that the counterparty is a newly established entity in which a top-tier global venture capital (VC) firm has participated. We will be able to provide additional information once Memento begins its official promotional activities.”
Industry observers point out that Curacle’s refusal to disclose information is excessively secretive, even considering the nature of the NewCo model. Even under the NCO model, the contract’s validity can only be recognized if it is backed by credible human and material resources. In particular, they argue that since major technology export contracts by listed companies directly impact stock prices and investment decisions, a minimum level of information disclosure is necessary to verify the identity of the contracting party.
In the case of the contract between D&D Pharmatech and Metsera, cited as a successful example of a “Newco-style” technology export in the biotech industry, details such as the company’s purpose, headquarters location, major investors, founding entities, management team, and funding details were disclosed relatively specifically, even though the counterparty was a newly established entity.
Metsera is a biotech startup established in New York, USA, in 2022 with the goal of developing an obesity treatment. Major U.S. biotech-focused venture capital firms ARCH Venture Partners and Population Health Partners (PHP) participated in its founding. PHP is an investment firm established by Ian C. Read, former chairman and CEO of Pfizer, and Clive Meanwell, founder of the Medicines Company. Meanwell is known for growing the Medicines Company and selling it to Novartis. These key figures are also positioned on Metsera’s board of directors and executive team. Clive Meanwell serves as Chairman, while Whit Bernard, formerly of PHP, serves as CEO, and co-founder J. Visioli serves as CFO and CBO.
Metsera was acquired by the US big pharma company Pfizer last November, joining the ranks of global pharmaceutical firms just 3 years after its founding. Pfizer agreed to pay Metsera shareholders USD 65.60 per share in cash and an additional USD 20.70 in contingent value rights (CVR) based on clinical and regulatory milestones. The base enterprise value is USD 7 billion, and including the contingent payments, the total transaction value amounts to up to USD 86.3 per share.
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.
- [Reporter's View] Contradiction of "K-passing" and a new drug powerhouse
- Reporter's view | Lee, Jeong-Hwan





