- LOGIN
- MemberShip
- 2026-05-11 15:36:45
- Company
- Jemperli starts reimbursement process in Korea
- by Eo, Yun-Ho Mar 31, 2023 06:08am
- Jemperli, a new cancer immunotherapy is attempting reimbursement listing in Korea. According to industry sources, GSK Korea has recently submitted an application to list its PD-1 inhibitor Jemperli (dostarlimab) for reimbursement in Korea. With the application, whether another treatment option may be released into the market remains to be seen. Unlike other immunotherapies, Jemperli was first approved as a treatment for mismatch repair deficient (dMMR) recurrent or advanced endometrial cancer that has progressed on or following prior treatment with a platinum-containing chemotherapy regimen. Therefore, the company plans to first provide treatment opportunities to patients in need. For the benefit of these patients, GSK has been carrying out an Expanded Access Program (EAP) in 15 major medical institutions in Korea since earlier this year before filing for reimbursement. Endometrial cancer develops in the endometrium, the inner lining of the uterus, and accounts for the majority of cervical cancer. Roughly, 1 out of 4 endometrial cancer patients are diagnosed to be in the advanced stage or experience recurrence, and patients whose disease recurs after platinum-based chemotherapy have limited treatment options. Jemperli’s approval was based on the cohort A1 analysis results of Phase I multicohort GARNET trial that included a cohort of patients with relapsed or advanced dMMR/MSI-H endometrial cancer who progressed on or following prior treatment with a platinum-containing regimen. In particular, this study had the largest cohort among PD-1 inhibitor monotherapy studies for dMMR/MSI-H endometrial cancer patients among those that have been conducted so far. The major efficacy outcome measures of the study were Objective Response Rate (ORR) and Duration Of Response (DOR) as determined by a Blinded Independent Central Review (BICR) according to (Response Evaluation Criteria Solid Tumors) RECIST v 1.1. As a result of analyzing a total of 108 patients with a median follow-up of 16.3 months, Jemperli demonstrated durable antitumor activity with a manageable safety profile. In the cohort, the ORR was 43.5% (95% CI, 34.0-53.4) and the median DOR was not yet reached. Disease Control Rate (DCR) was 55.6%, and the proportions of responses lasting 6 and 12 months were 97.9% and 90.9%, respectively. Jae-Weon Kim, Professor of Obstetrics and Gynecology at Seoul National University Hospital (President of the Korean Society of Gynecologic Oncology), said, “Most endometrial cancers are diagnosed early and have a relatively good prognosis, but 7-15% of these early-stage patients experience recurrences, and a limitation existed in that area due to lack of treatments available for use in case of recurrence after platinum-based chemotherapy.” Professor Kim added, “Tumors with confirmed dMMR/MSI-H respond well to treatment using PD-1 inhibitors, and Jemperli, with its significant treatment effect, safety profile, and treatment convenience, has offered a new possibility as second-line treatment for patients with recurrent or advanced dMMR/MSI-H endometrial cancer.”
- Policy
- Started a demand survey for drugs participating in the pilot
- by Lee, Tak-Sun Mar 31, 2023 06:08am
- Due to the burden of simultaneous data submission, there are likely to be fewer participating drugs. The government started a demand survey for drugs to participate in the parallel trial project of 'Permission Evaluation Negotiation', which is being pursued rapid reimbursement. The project was planned to drastically shorten the registration period by simultaneously proceeding with benefit evaluation and negotiations from the MFDS approval application stage. According to the industry on the 30th, the Ministry of Health and Welfare is conducting a drug demand survey to participate in a parallel pilot project in 2023, including application for permission, evaluation of benefits, and negotiation of drug prices, as part of a plan to improve access to treatment for expensive severe diseases and strengthen reimbursement management. Candidate drugs are scheduled to apply for approval from the Ministry of Food and Drug Safety in 2023 and are drugs aimed at treating diseases that threaten survival (life expectancy is less than one year) or rare diseases. If there is no existing treatment or if there is a clinically significant improvement in efficacy, etc. compared to existing treatment, participation is possible. The data to be submitted are related to approval: ▲the expected date of application for domestic approval, ▲effectiveness and effect among items of application for domestic approval (scheduled) , ▲countries approved in foreign countries and items permitted, ▲consistency between items approved in foreign countries and items scheduled for application for domestic approval. Regarding disease information of approved indications, ▲ disease severity, life expectancy, survival rate, progress, etc. ▲ existing treatment methods, and treatment performance data for the disease. Regarding clinical usefulness, ▲a clinical trial summary of the therapeutic confirmatory trial (or related evidence source), ▲the expected number of patients, ▲A8 listing status and listing price, ▲benefits evaluation result in excluding countries, ▲cost when applying for decision Effectiveness application track expected data (PE, medication cost comparison, etc.) can be submitted. The data will be used as basic data for selecting drugs for pilot projects. Applications must be submitted to KPBMA by April 4th. The government plans to promote the pilot project in earnest from the second half of the year. On the 28th, Yoo Mi-young, head of the Pharmaceutical Management Office of the HIRA, said at a meeting with the Korea Special Press Association, “We are currently discussing the target selection and related procedures with the relevant departments of the Ministry of Food and Drug Safety to promote the pilot project.” Explained. It is expected that there will be not many drugs to participate in the pilot project. Since the evaluation methods and conditions of the three agencies - MFDS, HIRA, and NHIS - are different, related pharmaceutical companies are also burdened with preparing data at the same time. It is interpreted that the first step was to investigate demand before starting the pilot project in earnest in the second half of the year.
- Policy
- About 180 clinics without IRB used diagnostic drugs
- by Park,Yang-myeong Mar 30, 2023 05:42am
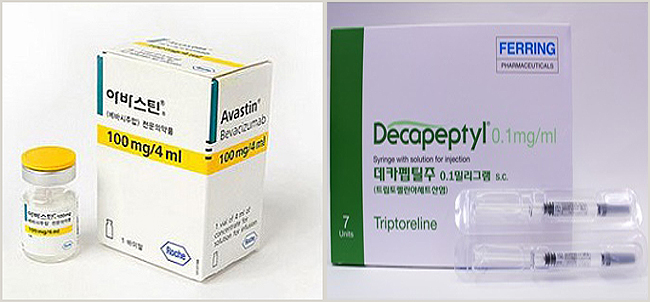
- Avastin and Decapeptyl, are drugs that allow the use of drugs beyond the scope of approval without the IRB. How much is it used in front-line medical institutions? About 230 macular degeneration treatment drugs Avastin and precocious puberty drug Decapeptyl injection were found to be using these drugs without IRB at 180 hospitals and clinics. The Pharmaceutical Management Office of the Health Insurance Review and Assessment Service disclosed the current status at a meeting with the Special Press Association held on the 28th. In 2019, the relevant notice was changed so that drugs exceeding the permitted scope could be used without reimbursement even without an IRB, but it was only last year that the system was actively applied to the field. The first drug approved by an institution not implemented by the IRB is Avastin, which is used for ophthalmic diseases. Avastin, as a targeted anticancer drug, has been used for various cancers, including colorectal cancer, breast cancer, non-small cell lung cancer, renal cell cancer, ovarian cancer, peritoneal cancer, and cervical cancer. In February 2020, the Korean Academy of Ophthalmology applied for the use of Avastin over the permitted scope to the ophthalmologist, and the HIRA prepared guidelines after discussing infection control measures. The guidelines include measures to prevent infection during the dispensing process. The HIRA received applications for use from non-IRB organizations since February of last year, and until last year, about 230 places used Avastin for ophthalmologic diseases. The second approved drug is Decapeptyl 0.1mg, which is used to diagnose precocious puberty. Decapeptyl is a prescription drug approved for hormone-dependent prostate cancer, endometriosis, uterine fibroids, and central premature onset of puberty in girls under 9 years old and boys under 10 years old. It was not approved as a diagnostic reagent for precocious puberty, but as the supply of the only diagnostic reagent was stopped, medical institutions are continuing to apply for approval for excessive use. It was applied by the Korean Hospital Association in October 2021, and it was confirmed that about 180 hospitals and clinics without an IRB applied for use between July last year and just five months. On the other hand, for the use of drugs over the scope of permission by institutions that have not implemented the IRB, the Korean Medical Association, the Korean Hospital Association, the Korean Dental Association, and related academic societies for each specialty apply for use over permission, and the HIRA reviews for approval. ▲ More than one-third of approved drugs compared to all drug clinical trial institutions for the same case, ▲approval of more than 3,000 drugs for the same case based on the previous year, ▲ Need for expansion Approval can be made if it meets at least one standard, such as medicines for rare diseases and pediatric diseases that are recognized.
- Company
- Viatrice Korea appoints Bill Schuster as new CEO
- by Jung, Sae-Im Mar 30, 2023 05:42am
- Viatris Korea announced on the 28th that it would appoint Bill Schuster as its new CEO. New CEO Bill Schuster is an expert with more than 30 years of diverse business experience in the healthcare and pharmaceutical industries. Based on his career as a senior leader in Asia, the Americas, and Europe, he is highly regarded for his understanding of the diverse healthcare systems and cultural backgrounds of each region. Since the launch of Viatrice in 2020, Bill Schuster, the new CEO, has led the growth of the diverse product portfolio as Head of the Beatrice Brand Division in Japan. He also contributed to Beatrice Japan's strong growth by playing a pivotal role in Japanese digital channel innovation as well. Bill Schuster, the new CEO, was born in Ireland and received a Bachelor of Science and Ph.D. from Dublin University and an MBA from INSEAD Business School in France. He held key positions in marketing and business strategy at Shionogi Pharmaceutical, Takeda Pharmaceutical International, and Mylan Japan. He has worked as a marketing manager in charge of Japan and Korea at Boehringer Ingelheim and Novo Nordisk Pharmaceuticals in Japan. New CEO Bill Schuster said, "I am very pleased to serve as CEO of Viatrice Korea, one of the leading companies in the pharmaceutical industry in Korea." We will further solidify our position in the Korean market and continue to contribute to Korean society by realizing the company's mission of supporting.
- Policy
- New Pompe disease drug Nexviazyme is approved in Korea
- by Lee, Hye-Kyung Mar 30, 2023 05:41am
- The long-term enzyme replacement therapy for Pompe disease, Nexviazyme Inj (avalglucosidase alfa-ngpt), has received marketing authorization in Korea. On the 29th, the Ministry of Food and Drug Safety (Minister Yu-Kyung Oh) announced that it had approved Sanofi-Aventis Korea’s Nexviazyme, a treatment for a rare condition called Pompe disease. Nexviazyme is a recombinant enzyme used as a long-term enzyme replacement therapy in patients confirmed with Pompe disease, which is caused by a genetic deficiency or dysfunction of the lysosomal enzyme acid alpha-glucosidase (GAA). Pompe disease is a rare genetic disorder that causes progressive weakness in the heart and skeletal muscle, leading to respiratory failure and cardiomyopathy. It is a rare condition reported in 1 in 40,000 around the globe and affects about 1,300 people in Korea. The disease presents in two main forms: infantile-onset Pompe disease (IOPD) which presents during infancy, and late-onset Pompe disease (LOPD) which is characterized by onset at all ages and progressively weakens the patients’ muscles. The muscle damage also leads to heart failure, respiratory failure, movement disorders, and sleep disorders. In particular, as Pompe disease is caused by a disorder in the GAA gene, a drug that targets the mannose-6-phosphate (M6P) receptor that allows the transportation of GAA enzymes into the lysosome is administered for its treatment. The drug is a biobetter that has improved dosage and administration over the company’s previous Pompe disease treatment, ‘Myozyme (alglucosidase alfa).’ The company had changed the sugar structure of Myozyme to improve cell absorption of Nexviazyme's active pharmaceutical ingredient. Patients who did not see an effect with Myozyme may use Nexviazyme Inj and see an effect. The MFDS said, “We will continue to make efforts to allow the prompt provision of treatments with confirmed safety and efficacy based on regulatory science."
- Opinion
- [Reporter’s View] Accepting drugs used by very few patients
- by Eo, Yun-Ho Mar 30, 2023 05:41am
- It's the same 'cancer', but different. So how should we regard these new anticancer drugs that target only a very few among all patients that have the same type of cancer? The cancer types that we commonly refer to, such as liver cancer, stomach cancer, and lung cancer are just major categories used, and the specific condition of each patient is classified in more detail. According to the specific class of cancer, the difficulty of treatment may vary even for tumors that originate from the same organ and affect a varying number of patients. The development of precision medicine is already heralding a shift in the prescription standards from organs to genes. In other words, the era of personalized healthcare has now arrived but is still regarded as an unfamiliar concept by many. Korea is still having difficulty accepting cutting-edge targeted anticancer therapies that show an effect regardless of cancer type according to each patient’s identified gene mutation. In this sense, targeted anticancer therapies and cancer immunotherapies that have previously been listed are experiencing considerable difficulties in the process of extending their reimbursement. This is due not only to the high price of the drugs but also because the drugs need to undergo value assessments again and reestimate their amount of use. This is the key framework that sustains Korea's national health insurance system. However, one of the characteristics of the recently developed new drugs is that the number of subject patients, that is, the number of patients confirmed with the specific genetic mutations that allow the use of such new drugs is very small. In other words, there are not many people who are eligible to use the new drugs. Less than 1% of all solid cancer patients have a rare type of solid cancer, and in terms of diagnosis, around 200 are diagnosed with such rare conditions in Korea. The typical standard-of-care therapies (existing drugs) do not work well in these patients. This was why industry voices have been on the need to redefine rare diseases. In other words, the number of patients corresponding to each treatment option should be reflected rather than the number of patients that are affected by the same disease. Of course, such redefinition would require systematic and detailed discussions. However, the time has now come for Korea to consider how to reimburse the new targeted anticancer drugs whose use has increased and targets reduced. Korea's current system is not the issue. Korea just need to consider how to solve the problem regarding the increasing number of anticancer drugs difficult to reimburse through the current system.
- Policy
- Obizur will be evaluated for reimbursement upon approval
- by Lee, Tak-Sun Mar 30, 2023 05:41am
- Obizur, a treatment for acquired hemophilia A for adults, approved on the 20th, immediately began a benefit evaluation. This drug is attracting attention as it is the first treatment to replace blood coagulation factor 8 with an indication for acquired hemophilia A. According to the industry on the 29th, the HIRA received an application for Obizur benefits and entered the evaluation. Obizur received approval from the MFDS on the 20th. This drug was designated as an orphan drug in July 2021. US FDA approval was obtained in 2014. Acquired hemophilia A is a rare disease in which autoimmune antibodies cause antibodies to coagulation factor VIII, and bleeding cannot be stopped like in hemophilia patients. The prevalence rate is 0.2 to 1.48 per year per 1 million people, and it is known that there are about 60 patients in Korea. Takeda explains that Obizur is a genetically engineered product made by removing the B-domain from pig coagulation VIII, which is similar to human, and replaces inactivated human VIII, which is not easily recognized by autoimmune antibodies, to help blood coagulation and help control bleeding. did. In phase 2/3 for 28 patients with acquired hemophilia A, Obizur stopped or reduced bleeding in all early bleeding episodes, showed clinical improvement, or showed VIII activity above the target 24 hours after the first administration. The treatment success rate at the time of final administration was 85.7%, and the success rate was higher in the patient group using Obizur as the first treatment at 94%. The patient group using the second-line medicine showed a 73% treatment success rate. It seems that Obizur applied for reimbursement to the HIRA immediately after passing the safety and efficacy verification of the Ministry of Food and Drug Safety.
- Policy
- Govt seeks to improve the drug price reduction system
- by Lee, Tak-Sun Mar 29, 2023 06:02am
- The Korean government will be working together with the pharmaceutical industry to come up with plans to improve the price reduction system that discounts the actual transaction price of drugs. The Health Insurance Review and Assessment Service plans to hold a roundtable meeting with the pharmaceutical industry to improve the actual drug transaction price reduction system based on the recently disclosed research service results. However, whether the improvements will be received with content by both the authorities and the industry remains in question as some of the research service results may be difficult for the pharmaceutical industry to accept. According to industry sources on the 21st, the results of the 'Study to Prepare a Comprehensive Improvement Plan through an Effect Evaluation of the Transaction Drug Price Reduction System' that was led by principal investigator Jin-Hyun Kim, a Seoul National University professor, was disclosed through the public institution management disclosure system on the 9th. The research team proposed short-term, mid-term, and long-term measures for improvement in its final report. As for the short-term measures, ▲abolishing the 10% upper limit in the price ceiling system and ▲reflecting the low purchasing price paid by national and public hospitals in the drug price cuts were proposed. As mid-term measures, ▲ identifying the actual transaction price including rebates ▲ tougher punishments for false reporting, ▲ real financial savings, and ▲ promoting low-price purchases, etc. were proposed. As long-term measures, ▲ switching to a list price reimbursement system rather than the currently ineffective actual transaction price reimbursement system, ▲reducing the insurance price ceiling based on the actual transaction price, ▲changing the drug price structure including the dispensing fee, and ▲ maximizing pharmaceutical expense saving effect on outpatient pharmaceutical expenditures were proposed. As the proposed mid-to long-term measures are difficult to apply immediately, the short-term plans proposed by the research team are highly likely to be discussed with the pharmaceutical industry as a promotion task. However, predictions are that it will be difficult to reflect the low purchasing price paid by national and public hospitals in the drug price cuts due to strong opposition from the industry. Therefore, realistic measures such as the abolition of the 10% upper limit in the price ceiling system are expected to be discussed. A HIRA official said, “The actual drug transaction price reduction system has been in effect for a long time, and there had been criticism on its low effect, therefore, we plan to come up with an improvement plan. If discussions progress well, measures may even be derived within the year.” The reductions to the actual drug transaction price had been carried out every year in line with the separation of prescribing and dispensing system in 2000 to reflect the actual transaction price of drugs in the drug price, and then has been conducted every two years since 2016. However, the prevailing opinion was that an improvement was needed as its drug cost reduction effect was poor and it was difficult to identify the actual transaction price. This was why HIRA set out to derive an improvement plan through a research service last year.
- Company
- JW Pharma registers the first overseas patent for JW0061
- by Mar 29, 2023 06:02am
- A researcher at JW Pharmaceutical is conducting a candidate substance test. (Photo by JW Pharma)JW Pharmaceutical announced on the 27th that it has obtained a patent for 'JW0061', a treatment for hair loss targeting Wnt, from the Russian Intellectual Property Office. This patent is for JW0061, a new drug candidate for hair loss treatment based on the Wnt signaling pathway. The patent protects the composition of JW0061. This is the first time that the JW0061 material patent has been registered. JW Pharmaceutical has applied for patents in more than 10 countries including the US, Europe, Japan, and China as well as in Korea. JW0061 is a first-in-class candidate substance that promotes hair follicle proliferation and hair regeneration by activating the Wnt signaling pathway in skin and hair follicle stem cells. It is effective for hair loss symptoms such as androgenetic alopecia and alopecia areata and is expected to have excellent hair loss prevention effects. JW Pharmaceutical participated in the 'Wnt2022' conference held in Japan last November and disclosed the results of the preclinical study of JW0061. JW Pharmaceutical also disclosed the results of animal experiments that confirmed the superior hair growth and hair follicle generation effect of JW0061 compared to the placebo group. As a result of animal experiments where JW0061 was applied, the time to enter the hair growth phase, which took an average of more than 50 days, was advanced by more than 15 days (30%). JW Pharmaceutical is conducting GLP non-clinical toxicity evaluation with the goal of starting clinical trials of JW0061 in the first half of 2024. We are also conducting joint research with medical staff in the field of dermatology in the United States.
- Company
- Bridge Biotherapeutics applies to FDA for the phase 1/2 plan
- by Mar 29, 2023 06:02am
- Bridge BiotherapeuticsBridge Biotherapeutics announced on the 27th that it had applied to the FDA for a phase 1/2 clinical trial plan for 'BBT-207', a 4th generation non-small cell lung cancer drug candidate. BBT207 is a new drug candidate that targets the C797S mutation that can occur after using third-generation non-small cell lung cancer treatments such as Tagrisso. Bridge Biotherapeutics plans to conduct clinical trials at 15 to 20 institutions in the US and Korea when the phase 1/2 clinical trial plan is approved. This clinical trial is a study to evaluate the safety, pharmacokinetic/pharmacodynamic properties, and efficacy of BBT-207 in patients with advanced non-small cell lung cancer with EGFR mutation after EGFR TKI treatment. The dosage will be increased through a phase 1/2 study targeting 112 patients, and an appropriate phase 2 dose will be explored. Capacity expansion will follow. Bridge Biotherapeutics plans to attend AACR on the 18th (local time) and present the preclinical data of BBT-207 as a poster. Based on the results of additional animal experiments, the company plans to publish data related to antitumor efficacy, survival rate improvement in brain metastasis animal models, and brain metastasis inhibitory activity, which study the potential of BBT-207 for targeted treatment for C797S-positive mutations.