- LOGIN
- MemberShip
- 2026-05-03 23:15:15
- Company
- Biological drug options for psoriasis treatment increase
- by Son, Hyung-Min Sep 02, 2024 05:48am
- Domestic and foreign pharmaceutical companies are expanding psoriasis treatment options through the development of biological agents. Recently, Korea's UCB Pharma succeeded in obtaining domestic marketing authorization for a biological drug with a new mechanism of action, sparking competition in the field. Other domestic pharmaceutical and biotech companies such as HK Inn.N and AprilBio have also entered the market by developing new biologics. Celltrion, Samsung Bioepis, and others are planning to expand their influence in the psoriasis treatment market by manufacturing biosimilars of biological drugs. #According to industry sources, the Ministry of Food and Drug Safety approved Korea's UCB Pharma’s interleukin-17 dual inhibitor Bimzelex on the 29th. Bimzelex is the first biologic to simultaneously target interleukin (IL)-17A and F. While biologics targeting IL-17A, such as Lilly's Taltz and Novartis' Cosentyx, have been available in the market, Bimzelex is the first drug to target IL-17F as well. IL-17 is an inflammatory cytokine that causes psoriasis. By targeting both IL-17A and IL-17F simultaneously, it is believed to be able to inhibit the inflammatory cytokines in higher doses and with greater potency. Psoriasis is a chronic, systemic skin disease caused by an abnormality in the immune system, and is characterized by erythema, a reddening of the skin, and psoriasis, which is white, scaly patches of the skin. Bimzelex’s approval is based on the Phase III BE READY study. The study compared the efficacy and safety of Bimzelex versus placebo in patients with plaque psoriasis. Results showed that the primary endpoint, a Psoriasis Area and Severity Index (PASI) score of 90 or greater at Week 16, was 90.8% in the Bimzelex arm. The proportion of patients achieving a PASI score of 100 was 68.2%, and the proportion of patients achieving an overall clinical response (IGA) score of 0 or 1 was 92.6% in the Bimzelex arm, significantly higher than the 1.2% in the placebo arm. Bimzelex also achieved significant PASI score improvements compared to other biologics. Bimzelex achieved significantly higher PASI 100 rates compared to Janssen's Stelara, AbbVie's Humira, and Cosentyx. Bimzelex’s PASI 100 achievement rate was also shown to have been maintained at a high level for 3 years in the BE BRIGHT open-label extension study. Bimzelex is the only IL-17 biologic introduced in Korea that can be dosed once every 8 weeks as maintenance therapy. It can also be self-injected by patients with education. Development of biologics active...competition heats up for psoriasis The domestic pharma and biotech industry is also developing biologics for psoriasis. Currently, biologics with psoriasis treatment indications include Abbvie’s Humira-Skyrizi, Cosentyx, and Taltz. Domestic biotech companies have also signaled their entry into the market by developing the drugs’ biosimilar versions. Celltrion recently received approval from the U.S. Food and Drug Administration (FDA) for a global Phase III trial of Cosentyx’s biosimilar, ‘CT-P55.’ Cosentyx is a biological drug developed by Novartis that inhibits IL-17A and is effective in a variety of inflammatory diseases, including psoriasis and ankylosing spondylitis. The study will enroll a total of 375 patients with plaque psoriasis. A comparative study will be conducted to demonstrate equivalence in efficacy and safety between the original drug and CT-P55. CT-P55 received Phase I IND approval from Japan's Pharmaceuticals and Medical Devices Agency (PMDA) in December of last year. In addition to Cosentyx, Celltrion has developed a number of other biologics, including biosimilars of Humira and Stelara. The company plans to expand its footprint in inflammatory diseases such as psoriasis with the Cosentyx biosimilar. Samsung Bioepis is challenging this market with the commercialization of its Stelara biosimilars. In April, Samsung Bioepis received approval from the European Medicines Agency (EMA) for Pyzchiva, a biosimilar of Janssen's autoimmune disease treatment Stelara. Pyzchiva is a biologic that treats plaque psoriasis, psoriatic arthritis, Crohn's disease, and ulcerative colitis, with annual global sales near KRW 14 trillion. HK Inno.N and AprilBio will develop biologics targeting novel mechanisms of action. HK Inno.N, YBiologics, and IMBiologics recently licensed-out IMB-101, a new drug candidate for autoimmune diseases, to US pharmaceutical company Navigator Medicines. MB-101 is a dual antibody drug candidate that simultaneously controls innate and adaptive immune responses by dual-targeting OX40L and tumor necrosis factor (TNF). The OX40L pathway is involved in the activation of T-cells, and immune cells, and TNF is a cell signaling protein involved in inflammatory responses. IMBiologics received IND approval from the U.S. Food and Drug Administration (FDA) in August last year and is currently conducting Phase I clinical trials. AprilBio has also successfully licensed out its technology for its autoimmune disease drug candidate 'APB-R3' to U.S. new drug developer Evommune. APB-R3 is a biological drug candidate that targets interleukin (IL)-18. IL-18-targeting candidates are known to be effective in various diseases including psoriasis, inflammatory bowel disease, atopic dermatitis associated with metabolic syndrome-related steatohepatitis, and sepsis. AprilBio has confirmed the tolerability and safety of APB-R3 in a Phase I clinical trial. Evommune plans to initiate a Phase II clinical trial for APB-R3 in the first half of next year.
- Company
- Yuhan’s Leclaza targets US mkt based on its clinical effect
- by Hwang, Byung-woo Sep 02, 2024 05:48am
- The non-small cell lung cancer drug Leclaza (lasertinib) in combination with Ryvrevant(amivantamab) has gained competitivity in the U.S., based on data presented at the World Congress on Lung Cancer (WCLC 2024). Additional data on Leclaza monotherapy and Leclaza in combination with Ryvrevantare is expected to be presented to further increase the drug’s clinical value. WCLC 2024 will be held from September 7 in San Diego, U.S. According to industry sources on the 31st, results from a study comparing the efficacy and safety of Leclaza+Ryvrevant and Leclaza monotherapy to Tagrisso monotherapy will be presented at WCLC 2024, which will be held from September 7 in San Diego, U.S. The data to be presented at WCLC 2024 are from the interim follow-up results of the study, which evaluated the clinical efficacy of Leclaza monotherapy (216 patients) versus Tagrisso monotherapy (429 patients) and Leclaza+Ryvrevant combination therapy (429 patients). In the results that were first disclosed through its abstract, the study showed that Leclaza monotherapy achieved a median progression-free survival (PFS) of 18.5 months by a blinded independent centralized review (BICR) over a median follow-up of 22 months. Also, the Leclaza monotherapy arm was associated with a 2% reduction in the risk of disease recurrence, progression, or death compared with the 16.6 months confirmed in the Tagrisso arm. In addition, an analysis of a high-risk subgroup showed that Leclaza improved PFS even further compared with Tagrisso. In patients with a history of brain metastases, the median PFS was 16.4 months with Leclaza versus 13.0 months with Tagrisso. The median PFS of patients with circulating tumor DNA (ctDNA) was also superior with Leclaza (18.4 months) versus Tagrisso (14.8 months). While more detailed results will be presented at WCLC 2024, experts believe that these results will support the drug settle in the U.S. market. “I think the argument for Janssen will be in providing a stronger rationale for why they chose Leclaza for combination therapy,” said Professor A, a hematologist-oncologist at a Big 5 hospital who asked for anonymity. ”The abstract shows that the efficacy was as expected and that Leclaza was better in terms of side effects, which is one reason why the company chose to use Leclaza as its partner.” Pic of Leclaza With the rising need to prescribe new treatments, the research presented at WCLC 2024 is expected to support the use of Leclaza. The congress will also feature a follow-up of the MARIPOSA trial presented at the European Society of Clinical Medical Oncology Annual Congress (ESMO 2023) last year. According to the abstract, at a median follow-up of 31.1 months, 44% (185/421) of patients in the Leclaza+Ryvrevant arm and 34% (145/428) in the Tagrisso arm were still on treatment. Overall survival (OS) data at a median follow-up of 31.1 months were also presented. At 24 months, 75% and 70% of patients were alive in the Leclaza+Ryvrevantand Tagrisso arms, respectively, and at 36 months, the corresponding figures were 61% and 53%, respectively. the interim analysis of the OS results showed a positive trend in favor of the Leclaza+Ryvrevantarm over the Tagrisso monotherapy arm as in last year but showed no statistical significance. On this, the researchers commented, “OS continues to show a trend toward improvement in the Leclaza+Ryvrevant arm compared to Tagrisso, reaffirming how the Leclaza+Ryvrevant combination is the standard first-line therapy for advanced EGFRm NSCLC.”
- Company
- Tevimbra may be prescribed in general hospitals in KOR
- by Eo, Yun-Ho Sep 02, 2024 05:48am
- The immuno-oncology drug 'Tevimbra' may now be prescribed in general hospitals in Korea. According to industry sources, BeiGene Korea's PD-1 inhibitor Tevimbra (tiselizumab) has passed the drug committees (DCs) of tertiary hospitals, including Samsung Medical Center and Seoul National University Hospital. In addition, the company succeeded in reapplying for Tevimbra’s reimbursement to the Health Insurance Review and Assessment Service’s Cancer Disease Deliberation Committee, raising the possibility of its insurance reimbursement. Tevimbra had previously received the ‘reimbursement standards not set' decision by the CDDC in March. The company had resubmitted its reimbursement application in May. Tevimbra (tiselizumab), which was approved in Korea last November, is an immuno-oncology drug indicated as a monotherapy for patients with unresectable, relapsed, locally advanced, or metastatic oesophageal squamous cell carcinoma who are unable to continue platinum-based chemotherapy or who have relapsed or progressed after receiving prior platinum-based chemotherapy. In the global Phase III RATIONALE-302 trial, Tevimbra prolonged median overall survival (OS) by 2.3 months compared to chemotherapy (8.6 months vs. 6.3 months), with a statistically significant 30% reduction in the risk of death, and did not show any crossover, unlike existing immuno-oncology monotherapies in the OS graph. In the trial, Tevimbra’s OS improvement was consistent across predefined subgroups, including baseline PD-L1 status, region, and race. Compared to chemotherapy, Tevimbra resulted in more than twice as many patients responding to treatment (20% vs. 10%), and showed an improvement in the median duration of response of approximately 3 months, from 4.0 months to 7.1 months, with sustained responses and a reduction in tumor size, which is directly related to quality of life for esophageal cancer patients. Furthermore, Tevimbra was associated with a 17% lower risk of disease progression or death in progression-free survival (PFS) (HR=0.83, 95% CI 0.67-1.01) and improved health-related quality of life (HRQoL) compared to chemotherapy. In April, the U.S. National Comprehensive Cancer Network (NCCN) revised its guidelines to recommend Tevimbra as a Category 1, preferred option for second-line treatment of esophageal squamous cell carcinoma.
- Policy
- Nicergoline for dementia listed at 50% of the highest-priced
- by Lee, Tak-Sun Sep 02, 2024 05:48am
- Sermion, the original drug containing the ingredient nicergoline.Before March this year, there were only two products containing the active ingredient nicergoline available. However, the number quickly increased, resulting in nearly a 50% difference between the highest and lowest prices. The prices of the listed products are getting cheaper with a stepwise pricing system applied. This reflects that more products are being listed for reimbursement listing. According to industry sources on September 1st, Korea Pharma's Pharma Nicergoline Tab 30mg was listed for reimbursement with a ceiling price of KRW 221 per tablet. Because there are already over 20 existing drugs in the same class, it was priced at around 85% of the prices of those, which is the lowest price. The price KRW 221 is nearly 50% of the highest price, KRW 424. Just last year, no one predicted that such many products containing nicergoline would be listed for reimbursement. In February, the products containing nicergoline 30mg were the original Il Dong's Sermion Tab 30 mg and Hanmi Pharm's Nicegoline Tab 30mg. After three pharmaceutical companies released their products in March, 20 products became available by March. The products listed since June have applied a stepwise pricing system, and the price is set at 85% of the lower price between the lowest price for the same medicine and the price calculated at 38.69% of the original drug price. Consequently, Hutecs Korea Pharmaceutical's Sarminon Tab 30mg, initially priced at 38.69% of the original price, was calculated at around 85%, now KRW 260 per tablet. Therefore, Pharma Nicergoline Tab 30mg's price was set at KRW 221, 85% of the lowest priced Sarminon Tab 30mg. The ceiling price will become even lower for products that will be listed and priced at 85% of the lowest price. Since 20 products became available in just under three months, latecomer products will, in turn, experience a price burden. Nearly 37 Nicergoline Tab 30mg are listed for reimbursement until now. The number of products containing the active ingredient nicergoline approvals by year (unit:#, source: the Ministry of Food and Drug Safety (MFDS), excluding products set for import or API). The products containing nicergoline is used for the first-line treatment of dementia symptoms, including memory impairment, concentration difficulties, judgment issues, and lack of initiative, related to primary degenerative vascular dementia and complex dementia. Korean pharmaceutical companies have jumped into this market this year because previously listed preventative drugs for dementia have failed to show efficacy and are being withdrawn. First, products containing acetyl-L-carnitine and oxiracetam were removed from the market after clinical trial reassessment failed. Clinical trial reassessment for the active ingredient choline alfoscerate is also underway, but the results are not promising. During the reimbursement reassessment review, it was decided that choline alfoscerate cannot be covered by reimbursement when used for mild cognitive disorder, an earlier stage of dementia. However, pharmaceutical companies have filed a lawsuit to seek nullification of reimbursement reduction for choline alfoscerate and received cancellation of the administration order, thus maintaining the reimbursement criteria. However, after losing the appeal court, pharmaceutical companies will likely face sales reductions. The annual prescription sales of choline alfoscerate amount to KRW 622.6 billion (based on UBIST). The market impact is expected to be substantial when reimbursement coverage for the mild cognitive impairment indication is no longer available because such use amounts to 80% of the sales. In countermeasures, pharmaceutical companies quickly released products containing nicergoline. The first product to be launched in South Korea was in 1978. The safety of the drug has been confirmed since it was launched 46 years ago. However, the National Health Insurance Service (NHIS) may attempt to reassess reimbursements. Even if that is the case, pharmaceutical companies can use products containing nicergoline as quick cards to fill the sales gap for drugs that provide cognitive benefits. An employee of a Korean pharmaceutical company said, "Nicergoline is categorized as an alpha-blocker and is effective in increasing blood flow in the brain and peripheral nervous system through vasodilation,' and added, "Because it is commonly used as a preventative measure for senile dementia or cognitive dementia, it is the most suitable drug to replace cognitive enhancers."
- Policy
- Lily's orphan drug 'Jaypirca' receives conditional approval
- by Lee, Hye-Kyung Aug 30, 2024 05:50am
- Product photo of Lily 'Jaypirca,' a designated orphan drug for the treatment of Mantle Cell Lymphoma that recently received marketing authorization in South Korea, is expected to submit the Phase 3 trial results showing therapeutic confirmation by March 2027. As it received conditional approval based on Phase ½ clinical trial results, the company must compare Jaypirca to existing Bruton tyrosine kinase (BTK) inhibitors and confirm clinical benefits. According to the meeting notes of the Central Pharmacist Review Committee, disclosed by the Ministry of Food and Drug Safety (MFDS) on August 27th, the committee members exchanged opinions on conditional approval of Jaypirca, an orphan drug for treating Mantle Cell Lymphoma (MCL). Jaypirca has received domestic approval for its efficacy and effects as a 'monotherapy for adult patients with relapsed or refractory MCL previously received at least two treatments.' Because this drug works differently in BTK-binding compared to existing treatments, Jaypirca has the advantage of treating MCL patients who are difficult to treat with existing treatments, such as 'Imbruvica (ibrutinib)' and 'Brukinsa (zanubrutinib).' However, similar to drugs of the same type, Jaypirca's Phase 2 clinical trial outcomes did not lead to improved survival in the Phase 3 trial. The expert opinion suggested that the decision on the clinical effectiveness of Jaypirca cannot be made based on the current outcomes. Jaypirca received marketing authorization based on Phase ½ clinical trial outcome as an orphan drug. It has been suggested that additional clinical trials are needed to confirm clinical effectiveness for the consideration of approval. A Central Pharmacist Review Committee member commented, "Although the global Phase 3 trial is being conducted to secure therapeutic confirmation, the trial's patient group differs from the target indication for approval. Thus, it is difficult to use the outcomes as the basis of approval," and added, "Objective response rate is not sufficient to decide on the final clinical effectiveness. Therefore, we need to grant conditional approval requesting the Phase 3 clinical trial showing therapeutic confirmation." Another member stated, "The drug meets the requirement for orphan drug designation. However, additional documents are needed to evaluate the final clinical effectiveness and benefits," and added, "As a condition of approval, we must require Phase 3 clinical trial showing the final clinical effectiveness and clinical benefits over existing BTK treatments." However, some argue that the drug's marketing authorization is necessary because existing BTK inhibitors target MCL patients who do not have alternative treatment options. A committed member said, "There are no existing treatment methods using BTK inhibitors in South Korea. The submitted Phase 1/2 clinical trial results show an overall response rate (ORR) of 57.8% and a duration of response (DOR) close to one year, meeting the efficacy targets and showing no significant issues in safety evaluations." Some argue that Jaypirca's approval is necessary because CAR-T for blood cancer treatments is expensive and has not been approved as an MCL treatment. A committee member said, "Previously, there were no treatment methods using BTK inhibitors in South Korea," and emphasized, "While there are no significant issues with safety evaluations, it is advisable to grant conditional approval based on the submission of Phase 3 data, as Phase 2 clinical trials alone cannot definitively establish the stability and efficacy of the treatment." Consequently, Jaypirca received conditional approval despite not having the Phase 3 document. The approval has been made due to the patient's need for treatments, with the expectations that the company will submit the clinical outcomes showing therapeutic confirmation by March 2027."
- Policy
- Reimb standards set for biliary tract cancer drug Pemazyre
- by Lee, Tak-Sun Aug 30, 2024 05:50am
- Pemazyre (pemigatinib), a targeted therapy for biliary tract cancer supplied to Korea by Handok, has successfully received reimbursement standards from the Health Insurance Review and Assessment Service's Cancer Disease Review Committee (CDDC). The reimbursement standards for the immuno-oncology drug Tevimbra were also set at the meeting. Merck's Erbitux has been granted an extended reimbursement. The CDDC held a meeting on the 28th to review reimbursement for new anti-cancer drugs and extend reimbursement for listed drugs. As a result, Handok’s Pemazyre may be reimbursed for the treatment of adults with previously treated, unresectable locally advanced or metastatic cholangiocarcinoma with a fibroblast growth factor receptor 2 (FGFR2) fusion or other rearrangement. Also, Tevimbra may be reimbursed as monotherapy in adult patients with unresectable, recurrent, locally advanced, or metastatic esophageal squamous cell carcinoma who are unable to continue prior platinum-based chemotherapy or who have relapsed or progressed after receiving prior therapy. In the case of Erbitux, which is primarily used to treat colorectal cancer, the application to extend its reimbursement as combination therapy with encorafenib (with bi-weekly Erbitux) as a treatment for adult patients with previously treated metastatic colorectal cancer with a confirmed BRAF V600E mutation was approved by CDDC. On the other hand, MSD Korea's rare disease drug ‘Welireg Tab’ failed to set reimbursement standards. In addition, Ipsen Korea's ‘Cabometyx Tab,’ Ono Pharmaceutical Korea’s ‘Opdivo Inj,’ and drugs containing anastrozole and letrozole also failed to establish reimbursement standards. In addition, the CDDC reviewed the use of prophylactic G-CSF for dose-dense MVAC/CMV therapy for urothelial cancer, TIP therapy for testicular cancer, and cabazitaxel therapy for prostate cancer with drugs such as Neulasta, reflecting the opinions of the medical community, but decided to maintain the current state.
- Company
- API competition↑ with entry of Chinese and Indian pharmas
- by Son, Hyung-Min Aug 30, 2024 05:50am
- Chinese and Indian companies developing active pharmaceutical ingredients (APIs) are accelerating their entry into Korea. Numerous Chinese and Indian companies as well as domestic companies participated in 'CPHI Korea 2024,' an exhibition for the global pharmaceutical, bio, and health functional food industry that was held at COEX in Samseong-dong, Seoul for 3 days from the 27th, to promote their API development capabilities. Domestic companies also participated in the event to showcase their technology. Domestic API companies highlighted the quality of their products as a strategy to differentiate themselves from overseas companies. A large number of Chinese and Indian API companies participate in CPHI China was the largest participating country at CPHI Korea 2024. More than 140 Chinese pharma and biotech companies participated in the event, which accounted for almost half of the 340 exhibitors. (Clockwise from the top left) Jiangxi Synergy, Bloomage Biotechnology, Zhejiang Biosan, Hubei Honch Pharmaceutical, Xieli Pharmaceutical from China As China supplies nearly half of the world's APIs, these companies are highly influential in the API market. Korea also imports the largest amount of APIs from China. These companies plan to actively target the domestic market to further increase their influence in Korea. China has been turning its eye to markets outside the U.S. after the U.S. strengthened sanctions against China. The U.S. has been implementing biosecurity legislation that would ban Chinese biotech companies from doing business with the U.S., favoring its own biotech companies. In response, Chinese companies have been eyeing the Korean market, which was why many Chinese companies participated in BIO KOREA 2024 in May to promote their company and technologies. The second largest overseas participant after China was India. India owns the second-largest API market following China. India accounts for 20% of the world's supply of APIs. India's pharmaceutical industry has been growing rapidly, focusing on biopharmaceuticals and contract development manufacturing organizations (From the left) Krishna Enzytech, Ashwagandha, Anupam Rasayan, Oceanic Pharmachem from India Various API, health functional food, and additive developers such as Krishna Enzytech, KSM-66 Ashwagandha, Anupam Rasayan, and Oceanic Pharmachem attended the event. In particular, the health functional food company Ashwagandha, participated as a title sponsor of the event. Korean API companies, including Novarex and Samoh Pharm participated at the event..."secured competitivity with quality" Many domestic companies also participated in CPHI Korea 2024. The second largest amount of companies that participated in the event were Korean companies, following China. Kukjeon Pharmaceutical, Samoh Pharm, Inist ST, Seoheung, and Novarex, among others, promoted their technologies for developing various APIs. (Clockwise from top left) Inist ST, Rihu Healthcare, Samoh Pharm, Suheung from Korea Domestic companies expressed high hopes based on the quality of their products. Currently, China and India dominate the global API market. Korea also imports most of its APIs from these countries. The problem is that domestic synthetic drugs are significantly less competitive in price than their Chinese and Indian counterparts, and the high dependence on overseas APIs limits the opportunities for domestic companies. “It is virtually impossible to keep up with China and India in terms of price competitiveness,” said an official from a domestic API company. “Domestic companies need to differentiate themselves in quality through superior manufacturing facilities and quality development capabilities to stand a chance.” However, the government's support is urgently needed to overcome the price competitiveness of overseas companies, and the industry's opinion is that no significant policies have been implemented up to now. The government's policy direction seeks to advance the self-sufficiency rate of APIs for pharmaceuticals, but the policies have not shown a significant impact until now. In particular, there has been a history of disruptions in the production of essential medicines due to difficulties in the supply of raw materials, including the deployment of THAAD, the valsartan crisis, COVID-19, and trade wars, but even after the pandemic turned endemic, the self-sufficiency rate of APIs has not improved. According to the Ministry of Food and Drug Safety, 31 of the 567 finished drugs reported to have been suspended from production, import, and supply from 2017 to June 2022 due to problems in the supply of APIs. 17 of these were national essential medicines. “To compete not only with the U.S. and Europe, but also with China, India, and Southeast Asia, that own prices competitivity, we need support policies at the finished drug level,” said an official from another domestic API company. ”The government has not clearly presented measures to support for self-sufficiency of APIs in Korea. The goal should not be to maintain a certain level of self-sufficiency, but to support the domestic pharmaceutical industry to compete with the global market.”
- Company
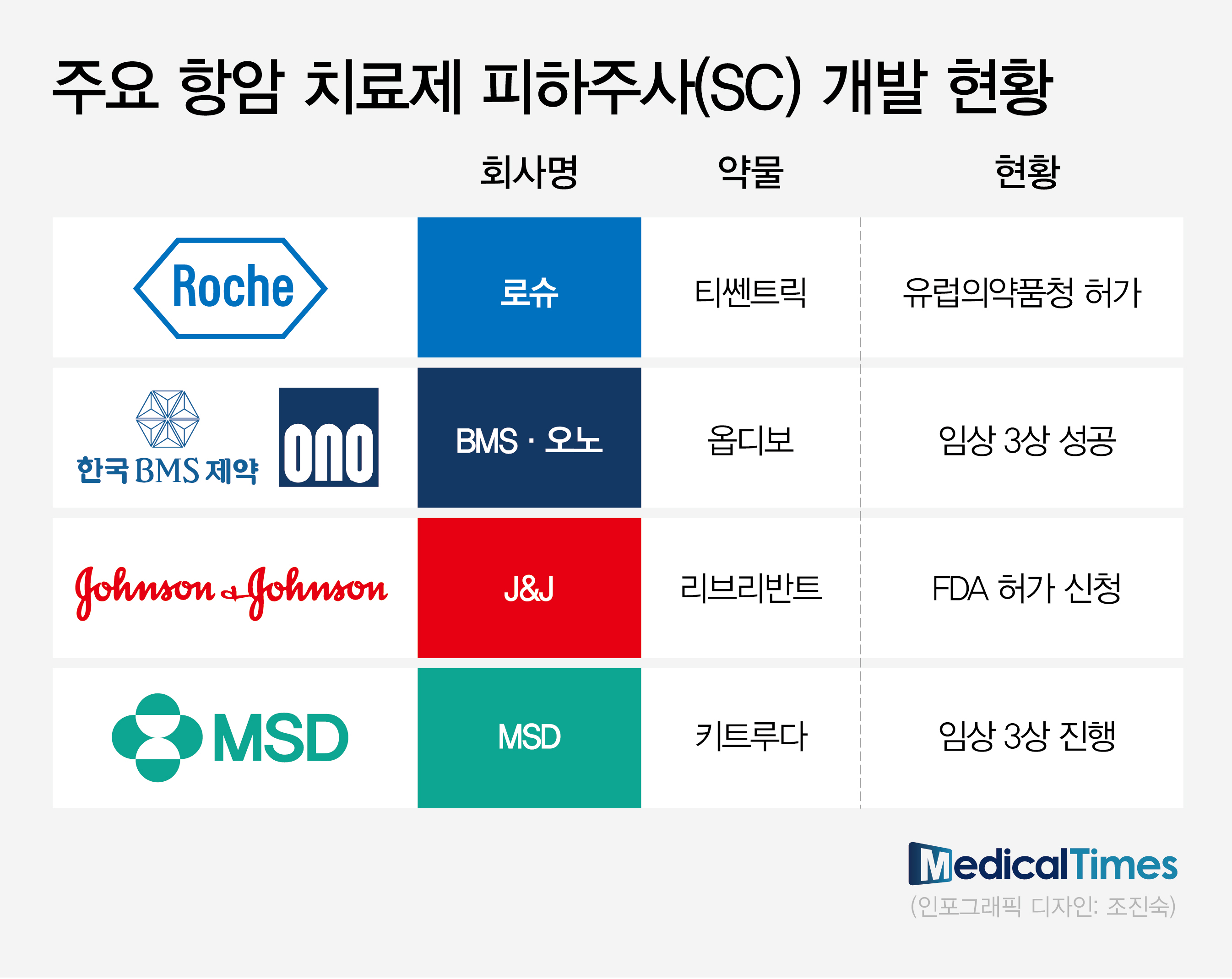
- Anti-cancer drug trends changes from IV→SC
- by Moon, sung-ho Aug 30, 2024 05:50am
- Global pharmaceutical companies speed up the development of subcutaneous (SC) formulations of their proprietary intravenous (IV) medications. To overcome the disadvantage of IV formulation medications that have long administration duration, more anti-cancer drugs are shifting to SC formulations. Following the trend, Korean companies with SC formulation technology are gaining attention. Then, what can we predict about the success in real-world clinical practices? While the shift to SC formulation of anti-cancer drugs is trending, pharmaceutical‧biotech industries focus on real-world clinical practices. Global pharmaceutical companies with IV formulations of anti-cancer drugs are proactively conducting clinical trials to transition to SC formulation products. (Table) RocheAccording to pharmaceutical and biotech companies on August 24th, global pharmaceutical companies with proprietary IV formulations of anti-cancer drugs are proactively conducting clinical trials to transition to SC formulation products. SC formulation products are injected into the subcutaneous layer of the skin. Injection sites are typically arms, thighs, and abdominal region. Until now, anti-cancer drugs were available primarily as IV formulations, which inject medications into veins. IV formulation has the advantages of fast absorption and accurate administration but has the disadvantage of taking a long time. For IV injection of anti-cancer drugs, patients were burdened by having to visit hospitals and withstand four to five hours of needle insertion. In contrast, developing anti-cancer drugs as SC formulations has the advantage of substantially improving patient convenience of administration. The administration duration was reduced from a couple of hours to a maximum of ten minutes. Therefore, patients do not need to stay in the hospital for a long time for anti-cancer drugs. As a result, global pharmaceutical companies with immune checkpoint inhibitors are proactively conducting clinical trials and applying for approvals to transition to SC formulations. Following Roche's 'Tecentriq (atezolizumab)' SC obtaining marketing authorization from the European Medicines Agency (EMA) in January, BMS and Ono Pharmaceutical's R&D of 'Opdivo (nivolumab)' and 'Rybrevant (amivantamab)' is nearing the end. These immune checkpoint inhibitors share the same goal to defend against their sales decrease due to patent expiration. Furthermore, the development of SC formulation products has gained more attention since SC formulations have a greater advantage in patient access in the global market, especially in the U.S. market. Recently, J&J confirmed the non-inferiority of SC formulations compared to IV formulations through the Phase 3 PALOMA-3 study, presented at the American Society of Clinical Oncology (ASCO) meeting. Based on the results, J&J has recently applied for additional U.S. FDA approval on Rybrevant SC formulation. Professor Byoung Chul Cho (Director of the Lung Cancer Center at Yonsei Cancer Hospital) said, "The United States provides incentives to using injections, and the amount of incentives is the same between IV injectable or SC injectable," and explained, "There is no need to maintain IV formulation injectables, which commonly induce injection-associated adverse reactions." Professor Sun Min Lim (Division of Medical Oncology, Department of Internal Medicine, Yonsei Cancer Hospital) also said, "SC injectables only take 1-2 minutes for Rybrevant administration. The common adverse reactions of IV injectables are fever and lowered blood pressure," and added, "In my opinion, SC injectables can reduce such adverse reactions." In the global market trend, clinical practices in South Korea are accelerating the introduction of SC formulation products, which overcome the previous disadvantages of IV formulation. For example, Roche's Phesgo has been recently introduced with reimbursement. Phesgo is an anti-cancer drug developed by changing the IV formulations of Herceptin (trastuzumab) and Perjeta (pertuzumab) to SC formulations. It received approval in September 2021 as the first investigational biopharmaceutical. Phesgo was developed by combining two IV formulation products into SC formulation. It is known to decrease the administration duration for patients with breast cancer substantially. It has been designated as an innovative new drug (IND) and is reimbursable, leading to its recent use in clinical practices. If patients with metastatic HER2-positive breast cancer who have been receiving the maintenance therapy, IV formulations of Herceptin plus Perjeta every three weeks, were to change to Phesgo SC, administration and monitoring duration is expected to decrease from over four hours to 20 minutes. The remaining step is whether the SC formulation can be used in clinical practices in South Korea. While there are clear advantages in patient administration, there are opinions that, unlike in the global market, such as the U.S., it is challenging to quickly replace the existing market in South Korea due to geographic accessibility. Additionally, many believe it will be challenging for healthcare professionals to readily switch to the SC formulation, which they have yet to experience. However, some argue that the fact that most cancer drug administrations are carried out primarily in large hospitals and are performed in the same way in injection rooms is more positive. The large number of patients who can receive treatment may facilitate the rapid establishment of the SC formulation. Professor Park Yeon Hee (Division of Hematolology-Oncology at Samsung Medical Center) said, "Korean patients tend to wait in the hospital, and there are long waits in large hospitals. Therefore, patients may prefer switching to SC formulations," and stated, "In clinical practices, patients may prefer IV injection despite the wait as SC prescriptions, other than clinical studies, have only been made recently."
- Company
- K-bio jumps into developing new drugs for pancreatic cancer
- by Son, Hyung-Min Aug 30, 2024 05:50am
- Korean pharmaceutical and biotech companies have jumped into the development of new drugs for pancreatic cancer, which is categorized as refractory cancer. Prestige Biopharma, New Cancer Cure Bio (NCC-Bio), LigaChem Biosciences, and Aptamer Sciences are conducting clinical trials to challenge the field. These companies plan to investigate the commercialization potential of their candidates through antibody-drug conjugates (ADCs), targeted anti-cancer agents, and new antibody drugs. New ADC drugs target TROP2 for treating pancreatic cancer According to industry sources on August 29th, Aptamer Sciences applied for a patent for its 'Aptamer-Drug Conjugate (ApDC)' and commenced treatment development. ApDC is a next-generation ADC new drug development platform with its proprietary branched linker-payload technology. Pancreatic cancer is known to have the lowest survival rate among cancer disorders. The five-year survival rate of pancreatic cancer is merely 15.9% from 2017 to 2021, according to the National Cancer Center. The early detection rate of pancreatic cancer is less than 10% due to the location of the organ, and the cancer readily metastasizes to peripheral organs. Until now, several domestic and global companies have jumped into developing new drugs for pancreatic cancer but mainly failed in clinical trials. Aptamer Sciences and latecomer companies plan to develop new drugs for pancreatic cancer using their new drug candidates, such as ADC. Aptamer Sciences derived 'AST-203' using the ApDC platform. AST-203 is made by conjugating TROP2-targeting antibody with 'MMAE,' a microtubule disruption agent, with linker 'VC-PAB.' TROP2 is an intracellular calcium signal transducer that regulates cell proliferation and survival. Although this protein is found in healthy cells, it is often overexpressed in cancer cells and is associated with drug resistance. The only TROP2-targeting new drug available in the market is Gilead Sciences' ADC Trodelvy, which is approved for treating triple-negative breast cancer. TROP2 is commonly found in breast cancer, non-small cell lung cancer (NSCLC), colorectal cancer, and pancreatic cancer. The clinical trials for latecomer agents are being conducted to target major solid cancers. In pre-clinical trials, Aptamer Sciences has confirmed the potential of AST-203 in a tumor spheroid model (3D cell culture of spheroids). According to the company, AST-203 showed a 6.7-fold higher tumor penetration rate than an existing therapy, Trodelvy. Additionally, in a pancreatic cancer animal model, AST-203 demonstrated dose-dependent tumor growth suppressive effects and tumor regression, reducing tumor sizes in all experimental animal groups. Aptamer Sciences aims to commence a clinical trial for AST-203 in two years. LigaChem Biosciences (hereafter referred to as LigaChem Bio) is developing TROP2-targeting ADC LCB84. Last year, LigaChem Bio successfully signed a deal with Janssen, a subsidiary of U.S.-based Johnson & Johnson (J&J) to license-out its LCB84. LCB85 consists of LigaChem Bio's proprietary ConjuAll linker and four MMAE, a microtubule disruption agent. ADC consists of a linker, payload (drug conjugate), and antibody. The ConjuAll linker is known to overcome the issue of releasing cytotoxic drugs into the blood and attacking healthy cells. In preclinical studies, LCB84 demonstrated effects in solid tumors not responding to topoisomerase enzymes-based TROP2 ADC payloads. Major ADCs like Enhertu (trastuzumab deruxtecan) incorporate the technology of topoisomerase enzymes. LigaChem Bio received Investigational New Drug (IND) approval from the U.S. Food and Drug Administration (FDA) in June, and the company is currently conducting Phase1/2 studies in the United States. In clinical trials, LigaChem Bio plans to investigate the preventive efficacy of LCB84 monotherapy and the combination therapy of LCB84 plus immune checkpoint inhibitor. Prestige Biopharma·NCC-Bio successfully enter clinical trials In addition to ADCs, various new drug candidates are being investigated for potential treatment of pancreatic cancer. Prestige Biopharma is developing a new antibody drug candidate, PBP1510. PBP1510 works by neutralizing pancreatic adenocarcinoma upregulated factor (PAUF), a protein target for the treatment of pancreatic cancer. The PBP1510 Phase 1/2a trials are being conducted in Spain, the United States, Singapore, and Australia. Through the trials, Prestige Biopharma plans to investigate the safety and tolerability of PBP1510 plus gemcitabine combination therapy. Prestige Biopharma aims to expand PBP1510's indication to ovarian cancer and prostate cancer in addition to pancreatic cancer. PAUF is known to be associated with ovarian cancer and prostate cancer. NCC-Bio will commence the development of a new drug for treating pancreatic cancer using 'KN510713,' a targeted anti-cancer agent and fatty acid oxidation (FAO) inhibitor. The company is conducting clinical trials after receiving approval for the KN510713 Phase 1 trial last year. NCC-Bio, founded by Kim Soo Youl, who used to work at the National Cancer Center, is a biotech company specializing in developing new anti-cancer drugs. KN510713 is being developed as an anti-cancer drug candidate that inhibits catabolism. Its clinical trial is the first anti-cancer agent trial to target inhibiting fatty acid oxidation (FAO) metabolism in cancer. KN510713 works by decreasing cancer cell growth by blocking the energy supply to tumor cells without affecting healthy ones. NCC-Bio is studying the last cohort of the Phase 1 trial and plans to enter the Phase 2 trial next year.
- Policy
- Prices of Dukarb, Rosuzet cut 3 yrs through PVA
- by Lee, Tak-Sun Aug 30, 2024 05:49am
- The insurance ceiling price of 6 drugs has been cut for 3 consecutive years through the price-volume agreement ‘Type C’ negotiations. According to the industry on the 28th, 6 drugs, including Boryung Pharmaceutical’s 'Dukarb', Dong-A ST 'Growtropin II Inj’, Hanmi Pharmaceutical’s 'Rosuzet Tab', Janssen Korea’s 'Concerta Oros ER Tab’, Boehringer Ingelheim ‘Jardiance Duo Tab', and Daewoong Bio’s ‘Gliatamin’, have had their insurance price ceiling lowered for 3 consecutive years from 2022 to 2024 through the price-volume agreement Type C negotiations. In this year's Type C negotiations, the prices of Boryung’s four Dukarb Tab items, which are combination antihypertensive drugs, were cut by -1.0% to -0.6%. In the case of Dukarb Tab 30/10mg, the ceiling price had been KRW 715 in June 2022 but will become KRW 686 from the first of next month. The company’s revenue from Dukarb has also increased significantly. Based on UBIST, outpatient prescriptions of Dukarb Tab rose from KRW 40.5 billion in 2021 to KRW 54.3 billion last year. Growth hormone drug Growtropin II Inj is another case where sales have increased significantly in recent years. Based on IQVIA, its sales increased from KRW 36.7 billion in 2021 to KRW 69.8 billion in 2023. five Growtropin II Inj items received a -5.0% to 0% reduction in their insurance ceiling price. (Source: IQVIA (Growtropin II Inj only), UBIST for the rest) Hanmi Pharmaceutical's four Rosuzet Tab items, a combination drug for hyperlipidemia, received a -1.5% to -1.3% reduction in their insurance ceiling price. The outpatient prescription of Rosuzet Tab (UBIST) also surged from KRW127.8 billion in 2021 to KRW178.8 billion in 2023. (Source: IQVIA (Growtropin II Inj only), UBIST for the rest) Janssen Korea's ADHD drug Concerta Oros ER Tab also received a reduction in their insurance ceiling price for the third consecutive year through PVA negotiations. This time, four of its items received a high rate of price reduction from -5.3% to -5.0%. The drug also showed strong growth in outpatient prescriptions from KRW 15.1 billion in 2021 to KRW 22.8 billion last year (UBIST). The insurance price ceiling of six Jardiance Duo Tab items, which is a combination used for diabetes, was reduced by -4.1% to -3.9%. The drug recorded a high growth rate, from KRW 24.1 billion in 2021 to KRW 39.4 billion in 2023 in outpatient prescriptions (UBIST). Sales of Gliatamin, a brain function enhancer, have continued to soar despite the reimbursement cut and clinical re-evaluations. Outpatient prescriptions increased from KRW 114.3 billion in 2021 to KRW 154.5 billion last year (UBIST). The price ceiling of Gliatamin Soft Cap and GliataminTab received a reduction of -3.3% this time (UBIST). In the case of Gliatamin Soft Cap, the price was KRW 504 per tablet on January 1, 2022, but will drop to KRW 476 next month. Over a three-year period, the reductions made in the insurance price ceiling amounted to -5.6%. On the other hand, starting this year, the National Health Insurance Service will reduce the reduction rate in the reference formula by 30% for innovative pharmaceutical companies or companies with an R&D ratio of more than 10% for drugs that have been subject to PVA negotiations 3 times in 5 years. This year, 17 items received a 30% reduction.