- LOGIN
- MemberShip
- 2026-04-28 11:08:22
- Policy
- How Trodelvy was applied flexible ICER for innovativeness
- by Lee, Tak-Sun Jul 14, 2025 06:04am
- The ADC breast cancer drug Trodelvy (sacituzumab govitecan, Gilead), which was reimbursed in June for triple-negative breast cancer, was the first case in which the ICER (incremental cost-effectiveness ratio) threshold was flexibly applied in recognition of its innovation. So how did the Drug Reimbursement Evaluation Committee (DREC) of the Health Insurance Review and Assessment Service at the time evaluate the innovativeness of this drug? According to the recently released evaluation results, Trodelvy was found to satisfy all criteria for innovative treatment, including the absence of alternative treatments, clinical improvement, and the criteria for expedited review. According to industry sources on the 11th, in the recently released DREC evaluation results for Trodelvy, the committee members recognized its reimbursement as appropriate despite the high cost-effectiveness ratio, taking into account the drug’s innovativeness." In August last year, the Health Insurance Review and Assessment Service established new requirements for the innovation of drugs subject to flexible ICER threshold assessment. According to the newly established criteria, the innovation of a new drug is recognized when ▲there are no substitute products or treatments with equivalent therapeutic positions, ▲significant clinical improvement can be recognized in the final outcome indicators, such as prolonged survival, ▲the new drug is a new drug approved by the Ministry of Food and Drug Safety through an expedited review process in accordance with Article 35-4, Paragraph 2 of the Pharmaceutical Affairs Act, or a drug recognized by the committee as equivalent to such a drug. All three requirements must be met for a new drug to be recognized as innovative. Trodelvy became the first drug to be recognized as innovative under these criteria. At the time, the DREC members evaluated that Trodelvy satisfied all three requirements. First, they determined that it satisfied the first requirement as the first antibody-drug conjugate (ADC) approved for triple-negative breast cancer, with no products or treatments with equivalent therapeutic positions. In addition, they evaluated that the second requirement was also satisfied, as clinical improvement (mOS HR=0.51) was recognized in the final outcome indicators, such as prolonged survival. Finally, considering that it was approved by the FDA as a breakthrough therapy and designated as a Global Innovative product for Fast Track item by the MFDS, the committee deemed that the third requirement was also satisfied. In terms of cost-effectiveness, the committee concluded that Trodelvy is significantly more cost-effective than existing anticancer drugs and breast cancer drugs that have been reviewed in the past. However, considering that it is a drug used for diseases that threaten survival in the third or further line of administration, that it has been recognized for improving the social burden of disease and quality of life, and that it is the first antibody-drug conjugate (ADC) approved for triple-negative breast cancer, the committee accepted its reimbursement as adequate. In other words, the high price was accepted in recognition of its innovation. Another factor was that the number of patients eligible for treatment was small, so the drug’s financial impact was not significant. According to the DREC evaluation results, Trodelvy is a drug used by approximately 200 patients, and the decision was made after comprehensive consideration of factors such as the number of patients eligible for treatment and the financial impact. In the end, Trodelvy was listed for reimbursement under two types of risk-sharing agreements (RSA) with the National Health Insurance Service. Among the types of RSA contracts that were applied, one was a Refund-Type agreement, where the pharmaceutical company reimburses a certain percentage of the claimed amount to the NHIS, and the other was the Utilization Cap/Fixed Cost per Patient Refund Type agreement, where the usage limit per patient is set in advance, and a certain percentage of the excess amount is reimbursed to the NHIS if the limit is exceeded. The listed price (maximum amount) is KRW 1,052,300 per vial, but due to the dual pricing system applied under the reimbursement-type contract, the actual price is higher. The estimated number of patients receiving Trodelvy annually is approximately 282, with an estimated fiscal expenditure of approximately KRW 12.5 billion. However, the actual fiscal expenditure is expected to be lower if the risk-sharing agreement is applied. At a 5% coinsurance rate, patients will only need to pay approximately KRW 2.21 million per year for its administration.
- Company
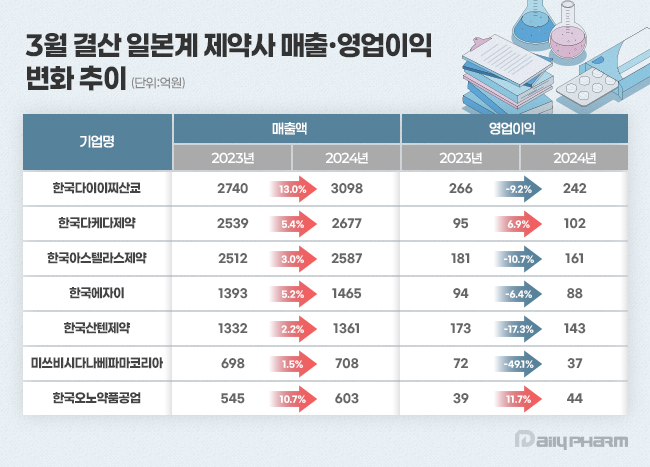
- Daiichi Sankyo and Ono Pharmaceutical see surge in sales
- by Son, Hyung Min Jul 14, 2025 06:03am
- Japanese pharmaceutical companies operating in South Korea saw overall sales growth last year. All seven companies surveyed achieved year-on-year sales growth, with Daiichi Sankyo Korea and Ono Pharma Korea leading the way with double-digit growth rates. However, in terms of operating profits, the companies showed mixed emotions. According to the Financial Supervisory Service's electronic disclosure system on the 11th, the performance of 7 Japanese pharmaceutical companies with March fiscal year-ends totaled at KRW 1.2499 trillion, up 6.3% from KRW 1.1759 trillion the previous year. During the same period, operating profit decreased by 11.2% from KRW 92 billion to KRW 81.7 billion. Major Japanese pharmaceutical companies' Korean subsidiaries follow the Japanese fiscal year, recognizing the period from April last year to March this year as their 2024 annual sales. #SB Daiichi’s sales surpass KRW 300 billion for the first time... Ono’s sales double in four years Last year, Daiichi Sankyo Korea's sales increased by 13.0% year-on-year to KRW 309.8 billion. During the same period, operating profit decreased by 9.2% from KRW 26.6 billion to KRW 24.2 billion. Daiichi Sankyo Korea's sales have been steadily increasing since 2020. The company surpassed KRW 200 billion in sales for the first time in 2020 with KRW 217.9 billion, followed by KRW 245.4 billion in 2021, KRW 253.2 billion in 2022, and KRW 274 billion in 2023, continuing its upward trend. In particular, it is analyzed that the company achieved synergistic effects by collaborating with the domestic pharmaceutical company Daewoong Pharmaceutical on certain cardiovascular products such as Lixiana and Sevikar. Daiichi Sankyo Korea signed a co-marketing agreement with Daewoong Pharmaceutical for Sevikar in 2013 and Lixiana in 2015, and has maintained its partnership with the company to date. Among these, the highest-selling product is the direct-acting oral anticoagulant (DOAC) Lixiana. According to market research firm UBIST, Lixiana's prescription sales last year reached KRW 117.5 billion, an 11.6% increase from KRW 105.3 billion in 2023. The three-component hypertension combination drug Sevikar HCT also maintained its growth momentum. Last year, Sevikar HCT's prescription sales reached KRW 42.1 billion, up 4.0% from the previous year. Daiichi Sankyo Korea generated approximately KRW 140 billion in prescription sales from olmesartan-based hypertension treatments, including Sevikar HCT (KRW 42.1 billion), Olmetec (KRW 30.6 billion), and Sevikar (KRW 68.8 billion). Daiichi Sankyo Korea is working to expand its business from cardiovascular drugs to anticancer drugs. In particular, the company is focusing on new growth areas such as antibody-drug conjugates (ADCs) by concentrating its R&D capabilities. Following the approval of Enhertu, the company is preparing to launch various treatments, including five ADC strategies such as Datroway, Patritumab deruxtecan, DS-7300, and DS-6000. Last year, Takeda Pharmaceutical Korea's sales increased by 5.4% year-on-year to KRW 267.7 billion. Operating profit rose by 6.9% to KRW 10.2 billion. Anticancer drugs such as Zejula, Alunbrig, and Ninlaro contributed to the sales growth through steady growth. According to the market research firm UBIST, Alunbrig recorded prescription sales of KRW 12.1 billion last year. Since its launch in 2019, its prescription sales have continued to increase. This treatment is gaining market share for its indication in ALK-positive non-small cell lung cancer. Alunbrig is in fierce competition with Pfizer's Lorviqua and Roche's Alecensa. However, However, Alunbrig offers the advantage of once-daily, single-tablet dosing, and can be taken regardless of food intake. This simplicity reduces the number of factors patients need to consider when taking the medication. The ovarian cancer treatment Zejula also recorded KRW 14.3 billion in sales last year, a 51.6% increase in prescriptions from the previous year. In October last year, Zejula was added to the health insurance reimbursement list for the treatment of HRd-positive ovarian cancer. Until then, Zejula had been reimbursed only as maintenance therapy for BRCA-mutated ovarian cancer patients who responded to platinum-based therapy as a first-line treatment for ovarian cancer. The growth of Ono Pharma Korea was also noteworthy. Ono’s sales last year were KRW 60.3 billion, up 10.7% year-on-year. Operating profit in the same period increased 11.7% from KRW 3.9 billion to KRW 4.4 billion. The sales and operating profit recorded by Ono last year are the highest figures since the company began submitting audit reports in 2017. Ono recorded sales of over KRW 40 billion in 2021 and surpassed KRW 50 billion for the first time the following year. Comparing the KRW 60.3 billion recorded by Ono last year with the KRW 31 billion in sales in 2020, sales increased by 94.5% over four years. The immune-oncology drug Opdivo drove Ono’s sales growth. Opdivo is an anti-PD-1 class immuno-oncology drug jointly developed by Ono and BMS, and was approved in Korea in 2015. Ono holds development and sales rights in Asia, including South Korea, Japan, and Taiwan. In particular, the combination of Opdivo and BMS's CTLA-4 targeted immunotherapy Yervoy has demonstrated efficacy in various solid tumors such as melanoma, renal cell carcinoma, and hepatocellular carcinoma, contributing to the increase in market share. According to market research firm IQVIA, Opdivo surpassed KRW 100 billion in domestic sales in 2022. All showed external growth, but with mixed operating profits Astellas Pharma Korea, Eisai Korea, and Santen Pharmaceutical Korea all saw slight increases in sales, but their operating profits all declined. Astellas Pharma Korea reported sales of KRW 258.7 billion last year, a 3.0% increase from the previous year. Harnal, Betmiga, Prograf, and Xtandi performed well, driving sales growth. Harunal, a treatment for benign prostatic hyperplasia, recorded prescription sales of KRW 66.2 billion last year, a 0.8% increase from the previous year. Betmiga, a treatment for overactive bladder, and Prograf, an immunosuppressant, have consistently recorded prescription sales of over KRW 30 billion even after their patents expired. Last year, prescription sales for Betmiga and Prograf were KRW 33.4 billion and KRW 33.6 billion, respectively, up 2.6% and 3.9% year-over-year. Operating profit decreased by 10.7% year-on-year to KRW 16.1 billion. A slight increase in the cost of sales contributed to the decrease in gross profit. Astellas Pharma Korea is pinning its hopes on its next-generation anticancer drugs. The company's Padcev, its ADC anticancer drug, and Vyloy, its new gastric cancer targeted therapy targeting Claudin 18.2, have entered the domestic market. Both drugs have shown remarkable results in clinical trials. Currently, Astellas Pharma Korea has completed its application for insurance reimbursement for both drugs. Eisai Korea's sales rose 5.2% from KRW 139.3 billion in 2023 to KRW 146.5 billion last year. Operating profit for the same period decreased 6.4% to KRW 8.8 billion. Sales of existing products such as the anticancer drug Lenvima, the gastroesophageal reflux disease treatment Pariet, and the Alzheimer's disease treatment Aricept remain steady. The three products—Aricept (KRW 95.9 billion), Pariet (KRW 17.9 billion), and Lenvima (KRW 12.9 billion)—combined for over KRW 100 billion in sales last year. Operating profit decreased slightly due to factors such as increased cost of sales. EisaiEisai is expecting strong performance from its new Alzheimer's disease drug, lecanemab. Lecanemab is an Alzheimer's disease treatment developed by Eisai and Biogen that selectively binds to amyloid beta, a substance believed to cause the disease, and has been proven to slow the progression of the disease and delay cognitive decline. It is reported that lecanemab is currently being prescribed at various general hospitals and semi-general hospitals in Korea. Santen Korea reported sales of KRW 136.1 billion last year, a 2.2% increase from the previous year. However, operating profit decreased by 17.3% to KRW 14.3 billion. Santen specializes in ophthalmic products and holds various formulations, including diquafosol. However, in South Korea, hyaluronic acid eye drops are primarily used, and the company has not significantly increased its prescription volume in Korea. As of last year, the product with the highest sales among Santen Pharmaceutical's portfolio was Cosopt S eye drops, used for glaucoma. Cosopt S’s prescription volume last year was KRW 33.9 billion, an increase of 2.6% compared to the previous year. Mitsubishi Tanabe Pharma Korea's sales last year were KRW 70.8 billion, up 1.5% from 2023, but operating profit fell 49.1% in the same period. The decrease in operating profit was due to an increase in selling, general, and administrative expenses, such as reimbursement and retirement benefits, as well as an increase in cost of sales.
- Policy
- Orphan drug 'Bylvay Cap' wins reimb approval after reeval
- by Lee, Tak-Sun Jul 14, 2025 06:03am
- 'Bylvay Cap,' which is being considered for the expedited reimbursement listing process as a designated drug for 'Pilot Project for Integration of Product Approvals, Reimbursement Coverage Reviews, and Drug Price Negotiations,' received reimbursement appropriateness decision after reevaluation. Consequently, Bylvay Cap will be subjected to negotiation with the National Health Insurance Service (NHIS) before its inclusion in the reimbursement list. Health Insurance Review and Assessment Service (HIRA) stated that it has held the 7th Drug Reimbursement Evaluation Committee (DREC) meeting on July 10 and decided as such. During the meeting, the DREC decided on the reimbursement appropriateness for Bylvay Cap 200, 400, 600, and 1200 μg (odevixibat sesquihydrate). Drug Reimbursement Evaluation Committee (DREC) Bylvay Cap is used to treat progressive familial intrahepatic cholestasis (PFIC). This drug was designated as the first concurrent approval-evaluation-negotiation pilot project in 2023. The criteria for the concurrent approval-evaluation-negotiation pilot project were drugs with sufficient efficacy intended for treating cancer or rare diseases with a life expectancy of less than one year, as well as those proving superior survival·treatment effectiveness for over two years when alternative treatments are not available. Bylvay Cap obtained marketing authorization from the Ministry of Food and Drug Safety (MFDS) in August 2024 and entered into the reimbursement evaluation process. Yet, the reimbursement evaluation process has been uneasy. A complaint was heard from an expert who attended the reimbursement review meeting from the evaluation stage. In the DREC meeting held last April, a conclusion was not reached, and it was decided to re-evaluate. It was only today, three months later, that it passed the DREC review. As drugs under the concurrent approval-evaluation-negotiation pilot project undergo a one-stop process without following separate stages, Bylvay is likely to have already entered negotiations with the NHIS. If the NHIS negotiations are completed, patients will be able to receive National Health Insurance coverage for this drug immediately after review by the Health Insurance Policy Review Committee. Meanwhile, the DREC decided on HLB Pharma's Citrelin ODT to receive a conditional approval, stating that reimbursement appropriateness if the company accepts a price below the evaluated amount. This drug is used for 'improving ataxia caused by spinocerebellar degeneration.'
- Company
- Bimzelx may be prescribed at general hospitals in KOR
- by Eo, Yun-Ho Jul 11, 2025 06:12am
- The new psoriasis drug Bimzelx may be prescribed at general hospitals in Korea. According to industry sources, UCB Pharma Korea's Bimzelx (bimekizumab) has been approved by the Drug Committees (DCs) of major hospitals nationwide, including tertiary hospitals like Seoul Asan Medical Center and Severance Hospital as well as major hospitals including Kyungpook National University Hospital, Pusan National University Hospital, Seoul National University Bundang Hospital, Chonnam National University Hospital, Jeonbuk National University Hospital, and Hanyang University Hospital. The company appears to be rapidly working to expand Bimzelx prescriptions following its reimbursement listing in June. i1Bimzelx is the first plaque psoriasis treatment that dually inhibits interleukin-17A and 17F (IL-17A and 17F). IL-17A and IL-17F are key cytokines that trigger the inflammatory process in psoriasis, and Bimzelx selectively and directly targets and inhibits both simultaneously. This dual inhibition mechanism against IL-17A and 17F is what makes the drug’s introduction significant. Despite the emergence of various psoriasis treatments, there remained an unmet need in practice due to resistance and other factors, which is why health professionals saw the introduction and reimbursement of Bimzelx encouraging. Yong-Beom Choi, President of the Korean Society for Psoriasis (Department of Dermatology, Konkuk University Medical Center), said, “Psoriasis is an intractable disease that recurs and improves repeatedly, and the quality of life of patients with severe psoriasis in particular tends to deteriorate significantly, affecting their mental health. Therefore, the reimbursement and launch of Bimzelx, which has been proven to be highly effective, is very meaningful for both medical professionals and patients.” He added, “Based on its next-generation mechanism of action, Bimzelx is expected to be an excellent treatment option for both new patients and those who have not seen sufficient results with existing treatments.” Meanwhile, in the Phase III BE READY trial, 90.8% of patients in the Bimzelx group achieved PASI 90 at Week 16, and 68.2% of patients achieved PASI 100. In another clinical trial that compared Bimzelx with another biological agent, there was a clear difference in the percentage of patients who achieved complete clearance of skin lesions at Week 16, or 'PASI 100'. Specifically, PASI 100 for the drug and control drug were as follows: ▲BE VIVID: Bimzelx 59%, ustekinumab (Stelara) 21% ▲BE SURE: Bimzelx 60%. 8%, adalimumab (Humira) 23.9% ▲BE RADIANT: Bimzelx 61.7%, secukinumab (Cosentyx) 48.9%, etc.
- Opinion
- [Reporter's View] US tariffs…variable that must be faced
- by Whang, byung-woo Jul 11, 2025 06:12am
- Tension is growing in the domestic pharmaceutical and biotech industry as President Donald Trump recently announced the potential imposition of tariffs on imported pharmaceuticals. If the tariff hike is imposed, it is expected to directly impact not only the procurement costs of raw materials and sub-materials for Korean companies but also their price competitiveness when exporting finished products. According to the announcement, the tariffs are projected to be imposed approximately 1 to 1.5 years from now. While opinions within the industry are divided on the realistic feasibility of applying tariffs as high as 200%, the burden companies would face, even if tariffs are partially imposed, would be substantial. In particular, the U.S. market is crucial for Korean pharmaceutical and biotech companies as an export destination. For domestic companies that have rapidly grown in areas such as biosimilars and active pharmaceutical ingredients (APIs), securing global competitiveness, tariff barriers pose a significant threat. If a deterioration in profitability and a decline in price competitiveness materialize, significant disruptions to companies' global strategies are inevitable. Furthermore, Korean companies that have relied on the Contract Manufacturing Organization (CMO) and Contract Development and Manufacturing Organization (CDMO) industries as key growth pillars could face direct pressure, such as demands to relocate production sites or re-evaluate contract structures. Although specific implementation plans have not yet been released, long-term and strategic responses are required. Thus, there is a growing need to establish various tariff imposition scenarios and prepare stepwise countermeasures starting now. This tariff issue is likely to affect not only the pharmaceutical and biotech industries but also industries across the board, including automobiles, electronics, and steel. Therefore, individual corporate responses alone have apparent limitations. Ultimately, the government and businesses must collaborate to develop various scenarios based on potential tariff increases and formulate corresponding phased countermeasures. At the government level, while diplomatic efforts are necessary to reduce the likelihood of tariff increases, it is also essential to implement policy support measures, such as financial assistance and tax benefits, to minimize the impact on companies. Especially given that the government has identified the pharmaceutical and biotech sector as one of the future growth engines, neglecting this anticipated threat or delaying a response would be a strategic misstep. The pharmaceutical and biotech industries, by their nature, require long-term preparation and investment. The pharmaceutical and biotech industries, which are highly susceptible to the negative effects of trade conflicts, demand even more concrete and proactive preparation. It is crucial to recognize that this President Trump-initiated tariff issue is not merely a political discussion but a significant variable that will determine the overall competitiveness and survival of the industry. Therefore, discussions on a pan-governmental response must be initiated fully. The pharmaceutical and biotech industries should use this tariff issue as an opportunity to comprehensively re-evaluate the structural risks associated with global market entry and formulate new strategies. The government must actively consider and implement consultative bodies in collaboration with the private sector to prepare for tariff increases and long-term competitiveness strategies.
- Policy
- Will drugs subject to reimbursement reevals be expanded?
- by Lee, Tak-Sun Jul 11, 2025 06:11am
- The government is reportedly planning to tighten the selection criteria ahead of the second phase of the drug reimbursement adequacy reevaluations, which is set to begin next year. As a result, it is expected that the number of ingredients subject to reassessment may be expanded. According to industry sources on the 10th, the Health Insurance Review and Assessment Service (HIRA) is currently collecting feedback from the pharmaceutical industry before finalizing its second-phase drug reimbursement adequacy reevaluation plan. The first phase will conclude this year. Since the pilot reevaluation was conducted for choline alfoscerate in 2020, HIRA has completed four full reevaluation rounds from 2021 to 2024, and the fifth round is currently underway. Thus far, the reassessment has focused on ingredients listed for reimbursement from 1998 to 2006, before the positive listing system was implemented, and ingredients currently undergoing clinical reevaluation by the Ministry of Food and Drug Safety. Also, ingredients with an annual total reimbursement claim exceeding 0.1% (approximately KRW 20 billion), and those reimbursed in only one foreign country among the A8 reference countries was the criteria. Industry observers anticipate that the selection criteria stated above will change significantly from the second phase of reevaluations. In particular, the strengthened criteria may lead to the inclusion of ingredients that were previously excluded from reevaluations. It is reported that authorities are considering expanding the annual claims threshold to identify ingredients that have remained in a regulatory blind spot. The criteria under discussion include lowering the reimbursement claim threshold from KRW 20 billion to KRW 10 billion, and expanding the eligibility from being listed in only one A8 country to fewer than three. As in the first phase of reevaluations, the second phase will also continue to target drugs listed before the implementation of the positive listing system, making it likely that previously excluded products will be included. If the criteria are expanded as proposed, pharmaceutical companies will likely begin reviewing their product portfolios to identify those that may be subject to reevaluation. Unlike in the first phase, drugs with lower claims may also be included, posing new challenges for the companies preparing for reevaluations. However, as the selection criteria are still under review, companies are expected to limit their response until the final criteria are confirmed. Given the delay in finalizing the criteria and scope for the second phase, the industry is calling for the 2025 revisions to be postponed to allow sufficient preparation time.
- Policy
- Pfizer discontinues the supply of 'Viviant' in KOR
- by Lee, Tak-Sun Jul 11, 2025 06:09am
- Product photo of ViviantPfizer announced of discontinuing the supply of its 'Viviant Tab (bazedoxifene acetate)' in Korea. They stated that supply discontinuation is due to the difficulty in securing a stable and continuous supply. Viviant had previously been out of stock in May due to delays in the manufacturing site. An analysis suggests that Viviant's market competitiveness has also weakened following the emergence of generics and fixed-dose combinations in the Korean market, as well as the arrival of biologics. According to industry sources on July 10, Pfizer Korea recently sent an official letter to its distributors and other trading partners, stating its decision to discontinue the supply of Viviant Tablet 20mg (28BLP formulation). Pfizer stated, "This supply discontinuation was an unavoidable decision due to difficulties in securing a continuous and stable supply," and explained, "The estimated depletion date for the current inventory is by December." The inventory depletion date could be brought forward depending on market demand. Viviant obtained domestic product approval in November 2011. Pfizer Korea launched it in the Korean market the following year. This drug is a once-daily selective estrogen receptor modulator (SERM) class osteoporosis treatment, which garnered attention as Pfizer's first osteoporosis drug. Until its patent expiration in 2018, Viviant gained significant success with approximately KRW 10 billion in prescription sales. However, its performance has been declining since the introduction of domestic pharmaceutical companies' generics and fixed-dose combinations. Currently, 15 fixed-dose combinations containing bazedoxifene acetate, Viviant's active ingredient, and Vitamin D are listed for reimbursement, and 5 generic drugs with the same active ingredient are also reimbursed. Last year, Viviant's outpatient prescription sales amounted to KRW 2.4 billion, a 9.4 percentage point decrease from the previous year. The osteoporosis treatment market is now dominated by biologics like Prolia, which offer both efficacy and convenience of administration, leading to a diminished market competitiveness for Viviant compared to previous years. Consequently, analysis suggests that not only the stated reason of supply source instability but also a decline in performance in Korea likely influenced Pfizer Korea's withdrawal from the market.
- Policy
- Hanmi’s BTK inhibitor poseltinib approved for P1T in KOR
- by Lee, Hye-Kyung Jul 11, 2025 06:09am
- Hanmi Pharmaceutical's BTK inhibitor ‘poseltinib,’ which was licensed out to Nobo Medicine last year, will enter Phase I clinical trials in Korea as a monotherapy. On the 7th, the Ministry of Food and Drug Safety approved an open, multicenter, monotherapy, dose escalation Phase I clinical trial of NB02 (poseltinib) for patients with relapsed or refractory non-Hodgkin lymphoma. Following the approval granted last year to initiate a Phase II clinical trial for NB02 (poseltinib) in combination with rituximab and lenalidomide as a salvage therapy for patients with relapsed or primary central nervous system lymphoma (PCNSL), this marks the first Phase I clinical trial for poseltinib as a monotherapy. The Phase II trial is expected to enroll 18 patients in South Korea and will be conducted at Seoul National University Hospital until June 2028. Poseltinib is an oral drug candidate for autoimmune diseases and blood cancers that selectively inhibits BTK (Bruton's Tyrosine Kinase), an enzyme related to B lymphocyte activation signals in the human body. Hanmi Pharmaceutical first developed the drug and licensed it out to Eli Lilly in 2015 for USD 690 million, but the rights were returned in January 2019 after the drug failed to demonstrate efficacy in a Phase II clinical trial for rheumatoid arthritis patients. Hanmi Pharmaceutical continued developing poseltinib after the rights were returned and in October 2021, the company signed a joint development agreement with Genome Opinion, which became Nobo Medicine. In June last year, Hanmi Pharmaceutical signed a license-out agreement with Nobo Medicine for poseltinib. The total contract value and license fee are undisclosed, as agreed by both parties. Under the agreement, Nobo Medicine secured the global exclusive rights to poseltinib. Nobo Medicine was established in October 2017 under the name Genome Opinion and changed its name to Nobo Medicine in April last year. In addition to poseltinib, the company’s pipeline includes candidates for cardiovascular and ophthalmic diseases.
- Company
- Gastric cancer-targeted therapy 'Vyloy' seeks reimb again
- by Eo, Yun-Ho Jul 10, 2025 06:10am
- Product photo of Vyloy Vyloy, a gastric cancer-targeted therapy, is once again vying for inclusion in the National Health Insurance reimbursement list. According to industry sources, Astellas Pharma Korea recently submitted a reimbursement application for Vyloy (zolbetuximab), a targeted treatment for Claudin 18.2-positive gastric cancer. Vyloy did not pass the Health Insurance Review & Assessment Service's (HIRA) Cancer Disease Review Committee (CDRC) in February. Consequently, it remains to be seen whether it will achieve success in this second attempt. Vyloy, approved in Korea last September, is the world's first approved Claudin 18.2-targeted therapy. It is an immunoglobulin monoclonal antibody that acts by binding to Claudin 18.2, a protein expressed and exposed in the stomach. The basis of Vyloy's approval was the SPOTLIGHT Phase 3 study, which showed that the median progression-free survival (mPFS) for the Vyloy and mFOLFOX6 (oxaliplatin, leucovorin, fluorouracil) combination therapy was 10.61 months, higher than the placebo group's 8.67 months. The median overall survival (mOS) for the treatment group also surpassed that of the placebo group, which was 15.54 months, reaching 18.23 months. The GLOW study also found that the Vyloy and CAPOX (capecitabine and oxaliplatin) combination therapy recorded a median PFS of 8.21 months, reducing the risk of disease progression or death by approximately 31%. However, Vyloy is currently an unreimbursed drug. It was submitted to HIRA's CDDC in February but failed to establish reimbursement criteria. Additionally, due to a companion diagnostic (CDx) issue last year, Vyloy was officially launched in Korea only in March. To use Vyloy, it's necessary to identify patients who are Claudin 18.2-positive. This was complicated because the companion diagnostic device used for Claudin 18.2 diagnosis was being considered for the evaluation of new medical technology. In response, Astellas initiated an Expanded Access Program (EAP) even before Vyloy's approval to ensure that patients in need could access the treatment quickly. Currently, 51 patients are enrolled in 10 institutions through this program. Professor Sun Young Rha of Yonsei Cancer Center's Division of Medical Oncology stated, "Approximately 90% of metastatic gastric cancer patients are HER2-negative, creating a critical need for new biomarker-targeted therapies." Rha added, "In a situation where about 40% of HER2-negative patients are reported to be Claudin 18.2-positive, the introduction of Vyloy, which selectively binds to Claudin 18.2, presents new treatment possibilities."
- Opinion
- [Reporter’s View] Time to verify K-radiopharmaceuticals
- by Son, Hyung Min Jul 10, 2025 06:09am
- Radiopharmaceuticals are special preparations that combine drugs with radioactive isotopes for diagnostic or therapeutic use. In particular, radioligand therapy (RLT), which delivers radiation directly to tumor cells, is known for its high precision, being referred to as "moving radiation surgery." As beta particle-based radioligand therapy enters the commercialization stage, the global industry is shifting its focus to alpha particle-based therapies, which offer higher energy, shorter range, and more precise killing power. Leading candidates include actinium-225 and astatine-211. These agents, which have a shorter range and higher linear energy transfer (LET) than the existing beta-particle-based lutetium-177, are gaining attention as next-generation targeted therapies for their ability to precisely target cancer cells while minimizing damage to surrounding tissues. Multinational pharmaceutical companies are also making bold investments. Novartis, which developed Lutathera and Pluvicto, acquired a radioligand therapy developer Mariana Oncology last year. AstraZeneca has begun joint development with Actinium Pharmaceuticals. The United States, Canada, and Europe have already incorporated the development of actinium production and purification infrastructure into their national strategies. In contrast, Korea is still in its early stages. Although the Korea Atomic Energy Research Institute (KAERI) and the Korea Institute of Radiological & Medical Sciences (KIRAMS) have isotope production and some research platforms, vertical integration encompassing isotope purification, labeling technology, ligand design, and clinical development has not yet been achieved. Above all, the connection between hospitals, nuclear power, and pharmaceutical companies remains weak. While platforms exist, they rarely lead to the development of pharmaceuticals. Radiopharmaceuticals for diagnostic use have been commercialized, but the development of therapeutic agents is still lagging behind. Nevertheless, companies in Korea have also begun to explore this field. Currently, Cellbion is completing Phase II clinical trials for prostate cancer and is expected to apply for approval. They expect therapeutic effects comparable to or better than those of the already commercialized Pluvicto. FutureChem has completed a Phase II clinical trial for prostate cancer and plans to proceed with Phase III trials. The company plans to apply for conditional approval as soon as it receives authorization for the Phase 3 Investigational New Drug (IND) application. In this sense, the rumored development of K-radiopharmaceuticals is finally beginning to show some tangible results. However, the companies’ pipelines still have gaps in key technological elements such as target protein design, clinical strategy, and formulation optimization compared to global standards. Therefore, structural government support is necessary to secure independent competitiveness. Radiopharmaceuticals are a complex technology with a high barrier to development. However, as the entry barriers become extremely high, some experts believe that successful localization could enable market dominance even beyond that of antibody-drug conjugates (ADCs) once a company enters the field. The solution for domestic production of radiopharmaceuticals lies not in isolated technologies but in “strategic integration.” It is now time to establish a “Korean RLT Alliance” that encompasses everything from target proteins to hospital networks, clinical design, and isotope supply. Otherwise, domestically produced radiopharmaceuticals will remain mere objects of technology transfer rather than becoming market leaders.