- LOGIN
- MemberShip
- 2026-05-13 23:39:07
- Policy
- HIRA, established a management system for high-priced drugs
- by Lee, Tak-Sun Dec 19, 2022 04:34am
- Medical institutions submit administrative information and response evaluation results to the Board of Audit and Inspection. The HIRA (Director Kim Sun-min) announced that it has been operating a "high-priced drug management system" since the 12th to increase the work efficiency of analyzing the results of response evaluations for patients receiving high-priced drugs and to facilitate data submission. It is explained that the high-priced drug management system was promoted to analyze and monitor the entire administration process from administration information of ultra-high-priced drug administration patients such as Kymriah and Zolgensma to response evaluation of drugs. Medical institutions that claim expensive drugs log in to the "care institution work portal" and fill out and submit "administration information and response evaluation results" according to the drug-specific evaluation form, and the application will be completed after inspection by the evaluation agency. Looking at the evaluation period by drug, Kymriah submits it for one year every six months and Zolgensma for five years every six months. Medical institutions that administer ultra-high-priced drugs such as Kymriah and Zolgensma, which were registered this year, were cumbersome and uncomfortable by submitting patient-specific administration information by e-mail, but it was improved by directly entering or uploading data according to the system. It is explained that the administrative convenience of medical institutions' submission of data has been increased, and the HIRA has improved work efficiency as accurate data can be collected through real-time data checks. Recently, as social demands for expensive new drugs with high treatment effects are increasing, there is a need to strengthen patient access and secure the sustainability of health insurance through proper management of expensive drugs. In response, the HIRA Pharmaceutical Management Office established a salary management roadmap for the proper management of high-priced drugs in July and formed a dedicated organization to prepare a driving force for the post-management of high-priced drugs. In the future, it will lay the legal foundation for the stable promotion of high-priced drug performance management by specifically defining the targets for high-priced drug management, follow-up management period, and billing method. "The establishment of a high-priced drug management system is the first step to establish a performance-based follow-up system for ultra-high-priced new drugs," Yoo Mi-young, head of the drug management office, said. "The performance evaluation results will be used as data to calculate the NHIS's drug refunds and contribute to securing health insurance sustainability."
- Policy
- Discard Moon Care Policy
- by Lee, Jeong-Hwan Dec 19, 2022 04:34am
- President of Yoon Suk YeolPresident Yoon Suk Yeol announced a normalization policy, emphasizing the necessity and urgency of reforming the health insurance system. Analysts say that it has virtually formalized the abolition of the policy to strengthen health insurance coverage, which is the core of the so-called "Moon Jae-in Care" promoted by the previous government. "We urgently need to normalize health insurance, the last bastion of protecting public health," President Yoon Suk Yeol said in a statement at a Cabinet meeting held at the presidential office in Yongsan this morning (13th). "Health insurance reform is not an option, but a necessity," he said. President Yoon criticized, "We have spent more than 20 trillion won on strengthening coverage over the past five years, but the government has neglected medical abuse and free riding of health insurance," adding, "The burden is being passed on to the majority of the people." This is a criticism of the Moon Jae In government's policy to strengthen coverage, and the Ministry of Health and Welfare has recently decided to review whether to apply benefits to MRI and ultrasound tests suspected of abuse among salary items covered by health insurance. President Yoon said, "Popular populism that wastes taxpayers' money is supposed to destroy finances, harm the foundation of the health insurance system, and eventually force the public to make great sacrifices," and stressed, "We need to strengthen health insurance benefits and qualification standards and prevent waste and leakage of health insurance." "We will strongly support those suffering in medical blind spots with reduced funds," he said. "The key to the health insurance system is to ensure that essential medical care that is expensive like serious diseases but must be included."
- Company
- Severe asthma treatment Fasenra seeks reimb in KOR
- by Eo, Yun-Ho Dec 19, 2022 04:34am
- Another severe asthma treatment is attempting insurance reimbursement in Korea. According to industry sources, AstraZeneca is undergoing the reimbursement process for its monoclonal antibody for asthma, Fasenra (benralizumab). Other biological drugs including GSK’s ‘Nucala (mepolizumab),’ and Teva-Handok’s ‘Cinqair (reslizumab)’ have also been reattempting reimbursement in Korea as well. Fasenra was approved in Korea in 2019 as an add-on maintenance treatment in adults with severe eosinophilic asthma inadequately controlled with existing treatment options. The drug is administered once every 4 weeks for the first 3 months, then every 8 weeks thereafter. One of the main purposes of asthma management is to reduce the risk of asthma exacerbations. Fasenra directly binds to the alpha subunit of the interleukin-5 receptor (IL-5Rα) to induce apoptosis. The drug demonstrated efficacy in reducing asthma exacerbation and improving lung function. In the global SIROCCO trial that was conducted to evaluate the effect of Fasenra in treating asthma exacerbations in 1,205 asthma patients including 122 Korean patients, the annual rate of clinically significant asthma exacerbations was reduced by 45% in patients who were treated with Fasenra once every 4 weeks, and by 51% in patients who were treated with Fasenra once every 8 weeks. Also, in the CALIMA trial, Fasenra demonstrated a significant reduction in asthma exacerbations compared to the placebo. In the trial, the annual rate of asthma exacerbations was reduced by 36% in patients who were treated with Fasenra once every 4 weeks, and by 28% in patients who were treated with Fasenra once every 8 weeks compared with placebo. In both trials, a change from baseline in mean FEV1 (forced expiratory volume in 1 second) was observed using Fasenra, and a consistent improvement compared with placebo. The long-term efficacy and safety of Fasenra in severe eosinophilic asthma were evaluated through the BORA trial, a long-term extension trial on 1,926 patients that participated in the SIROCCO and CALIMA trials. In terms of safety, Fasenra showed no significant difference compared to the placebo, and 72% of the patients that were administered Fasenra did not experience asthma exacerbations and were able to maintain their FEV1. Surprisingly enough, asthma is associated with a high mortality rate. The mortality rate of hospitalized patients due to asthma exacerbation is nearly 1/3, and the expenses spent by patients that require emergency treatment or hospitalization due to asthma exacerbation account for more than 80% of the total asthma-related cost, representing a high burden of social cost. Eosinophilic inflammations are found in 50% of all asthma patients and may lead to reduced lung function or asthma exacerbations. In particular, eosinophilic asthma patients that show increased blood eosinophil levels despite appropriate ICS therapy are inadequately controlled with existing treatment therapy including ICS and LABA therapy. Therefore, the quality of life of these patients is threatened by the pain caused by the symptoms and the frequent exacerbation of the disease. Meanwhile, AstraZeneca sought to expand its indication to uncontrolled Fasenra to chronic rhinosinusitis with nasal polyp (CRSwNP), but FDA turned down the final approval and requested additional data.
- Policy
- MFDS approves Jemperil, the third PD-1 inhibitor in Korea
- by Lee, Hye-Kyung Dec 15, 2022 05:55am
- The Ministry of Food and Drug Safety (Director Oh Yoo-kyung) approved GlaxoSmithKline's new drug endometrial cancer treatment Jemperil on the 14th. Jemperil will be the third PD-1 inhibitor to be licensed in Korea after Opdivo of Ono and BMS and Keytgruda of MSD. Jemperil is a monoclonal antibody targeting cell predetermined history 1 (PD-1), an immune gateway receptor for immune cells (T cells). PD-1 is an inhibitory immune checkpoint receptor belonging to the CD28 family and is expressed in activated T cells, B cells, NK cells, and bone marrow cells, and plays a pivotal role in immune regulation. The drug has been approved as an indication for the treatment of adult patients with recurrent or progressive inconsistency recovery defects (dMMR)/high-frequency microsatellite instability (MSI-H) endometrial cancer, which has been treated or progressed after treatment. It uses PD-1 expressed on the surface of immune cells (T cells) to block the survival mechanism of cancer cells avoiding immune cell attacks, and helping immune cells remove cancer cells. The Ministry of Food and Drug Safety said, "We will continue to do our best to quickly supply treatments that have been sufficiently confirmed for safety and effectiveness based on regulatory science."
- Company
- Asthma treatment Cinqair attempts reimb again in Korea
- by Eo, Yun-Ho Dec 15, 2022 05:55am
- The asthma treatment Cinqair for asthma is again attempting reimbursement listing in Korea. According to industry sources, Teva-Handok has recently submitted an application for the reimbursement listing of its interleukin-5 antagonist monoclonal antibody Cinqair (reslizumab). Following Cinqair’s steps, other biological drugs for asthma including GSK Korea’s ‘Nucala (mepolizumab) are also moving to start their reimbursement listing processes as well. As an interleukin-5 antagonist, Cinqair reduces levels of blood eosinophils, a type of white blood cell that is involved in the development of asthma exacerbation. The drug had first been first approved in Korea in 2017 as an add-on maintenance treatment for adult patients with an eosinophilic phenotype of asthma (those who have a blood eosinophil count of at least 400/㎕ before treatment) that were not adequately controlled with existing treatments. The company had applied for its reimbursement being launched without reimbursement in 2018 but failed. Teva-Handok has been jointly marketing Cinqair with Handok Pharmaceutical since its release. Meanwhile, Cinqair’s efficacy had been demonstrated through five placebo-controlled clinical studies that evaluated the safety and efficacy of Cinqair 3mg/kg in 1,028 adult and adolescent asthma patients that were uncontrolled with currently available therapies. In three Phase III clinical trial programs that were conducted on asthma patients with high blood eosinophil counts, Cinqair reduced the frequency of asthma exacerbations by up to 59% and significantly improved lung function, symptoms, and the asthma-related quality of life. Also, Cinqair received attention for releasing the post-hoc analysis results of asthma patients that require Step 4 and Step 5 treatment among all patients that participated in the Phase III trials. Cinqair reduced the clinical degree of asthma exacerbations in patients classified as Step 4 or 5 under the Global Initiative for Asthma guidelines by 53% and 72%, respectively, and increased the level FEV1 (forced expiratory volume in 1 second) by 103ml in Step 4 patients and by 237ml in Step 5 patients, demonstrating that the benefit was found to be greater in Step 5 patients.
- Company
- Verzenio expects to address the unmet demand
- by Eo, Yun-Ho Dec 15, 2022 05:55am
- Professor Son Joo-hyuk Expectations are high for securing early breast cancer indications for Verzenio, a breast cancer treatment with CDK4/600 inhibition mechanism. Lilly Korea held a press conference on the 14th to commemorate the expansion of permission for early breast cancer indications at high risk of recurrence of the anticancer drug Verzenio. Verzenio obtained approval from the Ministry of Food and Drug Safety on the 18th of last month as a treatment used in combination with hormone receptor-positive, human epithelial cell growth factor 2 negative (HR+/HER2-), early breast cancer adult patients at high risk of recurrence of lymph node-positive, and endocrine therapy. Verzenio's approval is meaningful in that the first CDK 4&6 inhibitor for early breast cancer patients at high risk of HR+/HER2-lymph node-positive recurrence was introduced in Korea. Sohn Joo-hyuk, a professor of oncology at Severance Hospital, shared the medical unfulfilled needs of HR+/HER2-lymph node-positive recurrence early breast cancer patients and the clinical value of Verzenio confirmed through the monarch clinical study, which was the background of approval of MFDS. In the first topic of the presentation, "HR+/HER2-High-Risk Early Breast Cancer Patients' Medical Unfulfilled Demand," Professor Sohn said, "Most of the female cancers in Korea are diagnosed early due to screening activation, especially in the standard treatment of HR+/HER2- patients, to prevent recurrence after surgery. He added, "The prognosis of HR+/HER2-early breast cancer is generally known to be good, but high-risk patients are highly likely to recur, making it difficult to expect long-term survival." According to domestic statistics, the survival rate of general early breast cancer patients is more than 90%, which is higher than that of other diseases. However, patients with recurrence risk factors such as lymph node-positive, high tumor grade, large tumor size, and fast cell proliferation are known to have a higher risk of remote recurrence and death than general patients. Studies have shown that if the tumor size exceeds 5cm, the 5-year survival rate of breast cancer patients decreases from 57% (without lymph node metastasis) to 21% (with lymph node metastasis). Professor Son said, "The recurrence of early breast cancer patients after the first treatment is usually one to two years, and more effective postoperative auxiliary treatment is needed to reduce the risk of recurrence and death." However, since the introduction of aromatase inhibitors in the early 2000s, the absence of new treatment options for HR+/HER2-early breast cancer patients has led to a medical non-fulfillment demand." Verzenio's monarch clinical trial is the only study that has confirmed successful results in about 20 years as a combination of endocrine therapy as an adjunct treatment for HR+/HER2-early breast cancer. In monarch cohort 1, which served as the basis for this indication expansion permit, Verzenio+ endocrine therapy confirmed remote recurrence risk reduction results through Distance Relapse-Free Survival (DRF) indicators as well as Invisible Disease-Free Survival (IDFS) indicators compared to endocrine therapy alone.
- Product
- Menarini Korea is expected to distribute Viagra
- by Kim JiEun Dec 15, 2022 05:55am
- Viagra, which was sold by Jeil, is likely to be distributed at Menarini from next month. According to the pharmaceutical industry on the 12th, Menarini is in charge of distributing urological treatments such as Viatris Korea's Viagra, Cadura, and Detrusitol. The move follows the termination of Viatris and Jeil's sales rights contract for these products. In addition to Viatris Korea and Viagra, Jeil has been conducting joint promotions of Lipitor Plus, and it is known that the promotion of Lipitor Plus is maintained. Medications that stop distribution through Jeil are 50 mg of Viagra tablet, 100 mg of Viagra L film, 100 mg of Viagra L film, 100 mg of Detrusitol SR 2 mg, 4 mg of Detrusitol SR, and 4 mg of Cadura XL. The date of discontinuation of distribution is December 23rd. There is a rumor in the industry that Viatris Korea has already decided on its next partner. Menarini is said to be the most likely related company. Prior to this, Menarini recently announced to trading drug distributors that it would change the OX-2 inhibitor Arcoxia 30mg seller. Organon Korea will take over Arcoxia's sales rights. Arcoxia was sold by Menarini under a global partnership agreement after MSD ended its green cross and co-marketing in 2018. In the industry, some analysts say that Viatris Korea's right to sell urology-related treatments such as Viagra may have been chosen as a follow-up measure following Nemarini's termination of Arcoxia sales rights. An official from the distribution industry said, "Viatris Korea has Menarini in mind as Jeil's next partner, and I know that only the final decision is left," adding, "It is expected to be a measure following the end of Arcoxia sales rights." 정품 비아그라 https://xoad79.com
- Policy
- MFDS “Increasing AAP manufacturing well in progress”
- by Lee, Hye-Kyung Dec 14, 2022 05:57am
- To the issue that had been raised on how the supply shortage is continuing in the field even after increasing the price of acetaminophen, the Ministry of Food and Drug Safety replied that it has reviewed the monthly production schedule and the increase in production is progressing according to plan. Also, the MFDS added that the acetaminophen stock shortages found in some online malls may not be an actual lack of stock, and that it would be necessary to check with the distributors. In other words, some online malls may be leaving the category as out of stock to prevent the large pharmacies to preoccupy the whole stock in online malls at once and allow small pharmacies the purchasing opportunity. Eun-hee Moon, Director of the Pharmaceutical Policy Division Eun-hee Moon, Director of the Pharmaceutical Policy Division at MFDS, said so at a briefing with the medical trade journal correspondents on the 13th. Director Moon said, “Even at the increased price, it is said that the companies cannot manufacture more because they are already at their maximum capacity, and some pharmacies are saying there isn’t enough supply. However, when checking with the manufacturers and reviewing their monthly production schedule, no company has yet said they are having difficulty matching the demand.” The Ministry of Health and Welfare raised the ceiling price of 18 products that contain acetaminophen 650mg to at least KRW 70 to a maximum of KRW 90 as of December 1st. The different ceiling prices have been set according to the manufactured amount. After holding drug pricing negotiations with pharmaceutical companies, the National Health Insurance Service and the companies agreed to increase the monthly average manufacture amount by 50% more for 13 months until September of next year. Also, in consideration of the increased demand and exhaustion of market inventory in the winter and in-between seasons, the period from this month to April next year will be set as an intensive management period, and the monthly target manufacture amount set by 60% more during that period. The average monthly supply will rise the previous 45 million tablets per month to 67.6 million per month for the 13 months, and to 72 million tablets per month during the intensive management period. Also, the MFDS issued an ‘Emergency management and import order for medical products,’ ordering companies that supply prescription acetaminophen 650mg to report their manufacture and import plans, expected monthly manufacture and import and manufacture and import results from December 1, 2022, to April 30, 2023. Director Moon said, “The monthly manufacture amount submitted by the pharmaceutical companies has been set by adjusting the manufactured volume of other drugs. It was not vaguely made." For companies that consign productions, the companies agreed on the price increase after making the production plans for all of its products and confirming their production schedule with their factories. Director Moon emphasized, “Hanmi Pharmaceutical and Chong Kun Dang switched their consigned acetaminophen products to self-manufacturing and submitted their specific manufacture schedules before signing the price increase agreement. The companies are working hard to achieve the goal of expanding production by 50% and 60% more from the previous period according to the plans set by the government.” The MFDS believes that the supply shortages raised in the field will not occur as the contracts that were signed mandate companies to refund the drug price if the production volume does not reach the target amount. Director Moon said, “The MFDS’ manufacture and import order was issued to address the concern that not enough acetaminophen will be supplied in the file even after the NHIS signed agreements with companies that link the production and supply amount to the drug price. As the MFDS and NHIS are managing the issue together, I believe the production of acetaminophen will be carried out as planned." Moon added, “Companies that cannot fulfill the manufacture/import orders were directed to consult with the MFDS, but there has been no talk of companies being unable to meet the volume yet. I have reviewed the production volume until early December, and more acetaminophen is being supplied compared to November." However, as the specific production volumes of acetaminophen in November and December need to be compared, the actual difference can only be found at the end of the year. The MFDS is also preparing a plan to prepare for the supply shortage of raw materials for cold medicine that may arise due to China’s recent lifting of COVID-19 restrictions. Director Moon said, “When checking the raw material suppliers, most were from the US, China, and India, and we think we can use Indian suppliers if there is a disruption in the supply of raw materials in China. While receiving plans for increased production of acetaminophen from companies, we are also checking the current status of raw material supply” The exemptions from the mandatory small-package packaging of prescription cold medicine also continue to be in effect. Moon explained, "We had originally decided to apply the small packaging exemption for the period when we monitor the supply and demand, which is conducted once every two weeks."
- Company
- Samsung Epis's Soliris biosimilar bioequivalent to original
- by Dec 14, 2022 05:56am
- Study results that show Samsung Bioepis’s Soliris biosimilar is bioequivalent to the original drug have been presented. On the 13th, the company announced that its biosimilar, SB12 has demonstrated equal effect to its original drug Soliris at the American Society of Hematology (ASH) Annual Meeting that is being held in New Orleans. Soliris is a rare disease treatment used to treat incurable rare diseases like paroxysmal nocturnal hemoglobinuria (PNH) that was developed by the US pharmaceutical company Alexion. SB12 is a biopharmaceutical drug developed using the same active ingredient contained in Soliris, eculizumab. Samsung Bioepis presented statistical analysis results in the form of a poster to add credibility to the clinical results through additional analysis of its Phase III clinical trial. The company had previously demonstrated the bioequivalence of SB12 to Soliris in terms of clinical efficacy and safety through a Phase III trial that had been conducted from 2019 to last year. The primary efficacy endpoint of the SB12 Phase III trial was the level of lactate dehydrogenase (LDH) at Week 26 and the time-adjusted area of under the effect curve (AUEC) of LDH from Week 14 to Week 26 and from Week 40 to Week 52. The red blood cell of patients are destroyed and LDH is released into the blood when a patient develops PNH. LDH is one of the biomarkers that can diagnose this. Elevated LDH levels are usually associated with liver disease and increased fatigue, etc. The trial looked at the time-adjusted area under the effect curve (AUEC) of LDH from Week 14 to Week 26 and from Week 40 to Week 52 to observe whether these levels were similar to the original. Results showed that the LDH level lay within the pre-defined equivalence margin, indicating that the SB12 was bioequivalent to the original drug. Samsung Bioepis conducted a further sensitivity analysis to add reliability to the accuracy of the results of the Phase III trial. Statistical analysis derived the same conclusions as the primary efficacy endpoint results that were observed in the Phase III trial. An official from Samsung Bioepis said, “SB12 contains the core purpose of biosimilars that seek to expand patient access to expensive medicines. We will continue to make efforts to provide more treatment opportunities to patients suffering from rare diseases.”
- Company
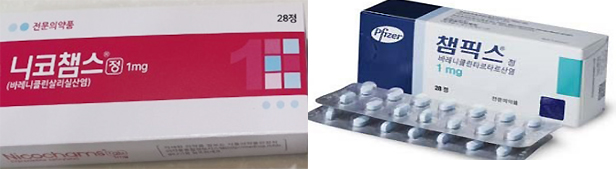
- Champix supply interruption
- by Kim, Jin-Gu Dec 14, 2022 05:56am
- Jeil Pharmaceutical's Nico Chams has dominated the market for anti-smoking treatment aids with Varenicline ingredients. Analysts say that while sales of major products, including Pfizer Champix, were suspended due to the detection of impurities in NNV components in September last year, it succeeded in digging into the gap and raising its influence to the level of market monopoly. ◆ First Pharmaceutical's Nico Chams share is 1% to 87% According to IQVIA, a pharmaceutical market research firm on the 13th, Jeil Pharmaceutical's Nico Chams' cumulative sales in the third quarter were 7.3 billion won. It increased more than nine times from 800 million won accumulated in the third quarter of last year. Sales have risen vertically since a series of impurities were detected in the anti-smoking treatment aid containing Varenicline in September last year. At that time, the Ministry of Food and Drug Safety announced the results of the NNV detection test on Varenicline-based drugs and decided to suspend sales only for products exceeding 185 ng/day. Products with a detection amount of 733 ng/day or more were ordered to be recovered. Among the products that were distributed in Korea, sales of seven products that CTC Bio had commissioned and one product that Pfizer Pharmaceutical Korea imported have been suspended. Nineteen manufacturing number items from three companies produced by CTC Bio have been ordered to be recovered. Pfizer Pharmaceutical Korea voluntarily recalled it. In the case of products produced by Jeil, impurities were detected for 16.70 to 43.28 ng/day, and they survived the recovery as well as the suspension of sales. Nico Chams' share also increased significantly around this point Until the second quarter of 2021, just before the suspension of sales, Nico Chams' market share was only 0.7%. However, the market share jumped to 24% in the third quarter of 2021, right after the suspension of sales. It then increased to 68% in the fourth quarter, and further expanded to 87% in the third quarter of this year. Based on sales in the third quarter, Nico Chams is actually over-exclusive with 2.4 billion won, and the rest is less than 200 million won. ◆Varenicline Market 34% in a Year↓ The lack of smoking cessation business overlaps with the prolonged suspension of sales. With the withdrawal of major products, the size of the anti-smoking treatment aid market for Varenicline ingredients decreased by 34% from 13 billion won in the third quarter of last year to 8.6 billion won in a year. This market has been on the decline for the past four consecutive years. As the government launched an anti-smoking treatment support project in 2015, the market expanded to 65 billion won in 2017 but has since shrunk to 16.2 billion won last year as it has steadily declined. This is the result of a combination of a reduction in drug prices and a decrease in the number of participants in the anti-smoking treatment project. On top of that, it is expected to decrease further this year due to the prolonged suspension of sales of impurity drugs that occurred in the third quarter of last year. Champix, which previously led the market, has still been suspended from sales and distribution as the impurity issue has not been resolved. Champix's sales plunged from 4.6 billion won in the second quarter of last year to 600 million won in the third quarter. Since then, sales of less than 10 million won have been recorded every quarter. Pfizer Pharmaceutical Korea said, "We are waiting for the resumption of supply at the global headquarters," adding, "We do not know the plans for the resumption of sales at this time."