- LOGIN
- MemberShip
- 2026-05-15 14:19:33
- Company
- 10 years since stem cell therapy was introduced
- by Chon, Seung-Hyun Jun 03, 2022 06:36am
- 4 types of domestic approved stem cell therapyTen years have passed since stem cell treatments appeared in Korea, but they have yet to produce commercial results. Medipost's CARTISTEM is recording annual sales of 10 billion won in Korea, but there is virtually no overseas sales. According to the Financial Supervisory Service on the 2nd, Medipost's sales of stem cell treatments in the first quarter were 4.5 billion won, up 3.5% from a year earlier. Medipost's stem cell therapy is the only treatment for osteoarthritis, CARTISTEM. CARTISTEM is a drug mainly composed of stem cells derived from cord blood of the same kind. Medipost succeeded in developing the world's first cord blood-derived stem cell therapy using cord blood stem cells from others, and received an item approval from the MFDS in January 2012. It is used for the treatment of knee cartilage defects in ICRS grade IV. After surpassing 10 billion won in sales for the first time in 2017, CARTISTEM posted 10 billion won in sales for five consecutive years until last year. Last year, CARTISTEM's sales reached 17.2 billion won. However, the recent growth has slowed down somewhat. CARTISTEM's sales grew 36.1% and 16.6% year-on-year in 2018 and 2019, respectively, but in 2020 and 2021, they increased only 3.2% and 3.8% year-on-year, respectively. Looking at sales in the first quarter, CARTISTEM increased 95.1% and 40.1% year-on-year in the first quarter of 2018 and 2019. However, CARTISTEM's sales in the first quarter of 2020 and 2021 grew less than 1% year-on-year and recorded 3% growth this year. Medipost is seeking to enter the global market of CARTISTEM, but no overseas sales have occurred yet. The sales performance of other stem cell treatments other than CARTISTEM is even lower than expected. Currently, there are a total of four types of stem cell treatments approved in Korea. After Pharmicell's acute myocardial infarction treatment Hearticellgram-AMI was approved in Korea as the world's first stem cell treatment in 2011, CARTISTEM and Antrogen's Crohnistem were approved for market in 2012, and CORESTEM's Lou Gehrig's fourth stem cell treatment in 2014. In the first quarter of last year, Pharmicell's bio business sales were only 572 million won. Pharmicell's bio business includes cosmetics along with Hearticellgram-AMI. Last year, Pharmicell's bio business sales were only 800 million won. Cupistem posted 700 million won in sales in 2020, including stem cell culture. It recorded 359 million won in sales in the first quarter of last year. Antrogen has not disclosed sales by business units since the second quarter of last year. Total sales of anthrogen in the first quarter were 1.7 billion won. CORESTEM's Neuronata recorded sales of 1.9 billion won and 1.2 billion won, respectively, in 2020 and last year. It made 300 million won in sales in the first quarter. Although it received great attention at the time of domestic approval, it is not in high demand due to the characteristics of rare diseases, and its preference is rarely increasing at the prescription site. Stem cell therapy was once in the spotlight as a next-generation growth engine, but news of the recent emergence of new products is also far from clear. Since Neuronata's approval in 2014, there are no stem cell treatments approved for eight years. In 2016, the Ministry of Food and Drug Safety revised the regulations for product licensing screening such as biological drugs, allowing cell therapy used for life-threatening diseases or severe irreversible diseases to receive conditional approval only with the results of phase 2 clinical trials. The intention is to shorten the time to enter the market for cell therapy products whose safety has been confirmed and treatment effects have been searched. A life-threatening disease is a disease that is likely to die if it is not accompanied by appropriate treatment and is defined as a disease in which there is no alternative to treatment. Serious irreversible disease refers to a disease or condition in which irreversible pathological conditions deteriorate for functions necessary for daily life if appropriate treatment is not accompanied. After easing regulations on cell therapy approval, bio companies such as Kang Stem Biotech, Naturecell, and Pharmicell attempted conditional approval of stem cell treatments, but all failed.
- Company
- Dukarb of Huons won the patent battle
- by Kim, Jin-Gu Jun 03, 2022 06:36am
- Given that Boryung won a series of previous referees, the pharmaceutical industry is paying keen attention to the decision, which is the opposite of the previous one. ◆Patent judge decides opposite to previous judgment According to the pharmaceutical industry on the 2nd, the Korean Intellectual Property Tribunal made a claim citation decision on the 31st of last month in a passive judgment on the scope of rights for patent of Dukarb filed by Huons against Boryung. For generic companies, including Huons, they won the Dukarb patent dispute for the first time. The Dukarb patent dispute began in March last year when 40 pharmaceutical companies filed a series of passive judgment on the scope of rights, starting with Arlico. The first decision was made in March this year. Arlico, Shinpoog, HLB, and Union Korea, Futechs Pharmaceutical lost after receiving a decision to dismiss the claim. In April, Hana, Whan In Pharm, and Union Korea also lost. The pharmaceutical industry judged that the rest of generic companies are likely to lose in the first trial. This is because the Patent Tribunal tends to cite the preceding trial as it is for cases with the same content. ◆ Huons wins first trial by changing target to non-core dose product Dukarb consists of a total of four items depending on the content. There are 30/5 mg, 30/10 mg, 60/5 mg, and 60/10 mg doses. Among them, patent of Dukarb protects only 30/5mg. Boryung has registered a patent only for its core dose of 30/5mg among the four Dukarb products. The generic companies requested a passive scope of rights confirmation trial targeting the core capacity of 30/5mg. However, Huons changed the target to 60/5mg. In the case of 60/5mg, non-infringement is clear because it is not protected by patents in the first place. For this reason, the Patent Tribunal made a decision citing Huons' claim. ◆Despite the victory of the first trial, patent avoidance of core capacity products failed However, since this trial decision is not about 30/5mg products, Huons cannot release 30/5mg generics even if it wins the first trial. 30/5 mg-related generic for exclusivity cannot be obtained. An official from the pharmaceutical industry said, "Huons met the requirement of winning the first trial, but the core capacity has not been avoided." "For Boryung, we are still protecting the Dukarb patent like the previous judges." Dukarb is a combination product that combines Amlodipine with Fimasartan, a hypertension treatment developed by Boryung. Among Kanarb-based combinations, the prescription performance is the highest. According to UBIST, a pharmaceutical market research firm, the prescription amount was 41.1 billion won last year. It increased by 14% compared to 36.1 billion won in 2020.
- Policy
- Zerbaxa passed evaluation committee after 4 yrs of challenge
- by Lee, Tak-Sun Jun 03, 2022 06:36am
- MSD Korea's antibiotic drug Zerbaxa has been recognized for its benefit adequacy by the HIRA Drug Reimbursment Evaluation Committee. As a result, it is likely to be listed on health insurance benefits through NHIS negotiations. The HIRA released the results of the 6th Drug Reimbursment Evaluation Committee review on the 2nd, and determined that Zerbaxa was appropriate for complex intra-abdominal infection, complex urinary tract infection, and hospital infection pneumonia. Zerbaxa is a super antibiotic approved in Korea in 2017, and is evaluated as a drug that can overcome antibiotic resistance. Dr. Low, who has challenged since 2018, succeeded in passing the Drug Reimbursment Evaluation Committee for the first time in four years. In 2019, the Drug Reimbursement Evaluation Committee recognized the clinical necessity but non-reimbursed because it was difficult to see that the clinical treatment rate was inferior. As the MOHW expanded the anti-bacterial system for treating symptomatic infections to PE in 2020, Zerbaxa also seems to have benefited.
- Policy
- Lioresal was newly listed
- by Kim, Jung-Ju Jun 02, 2022 05:59am
- With the new registration of 10mg/5mL of the skeletal muscle relaxant, the standard was newly established this month. Jakabi 5mg, which belongs to an anti-malignant tumor drug, has been clearly set in accordance with the change in permission from the MFDS. The MOHW announced that it partially revised the "details on the criteria and methods for benefits" as of the 1st. Looking at the change table, 10mg/5mL of Lioresal was newly registered, and the standard for this ingredient was newly established by applying the permission of the MFDS. According to the MFDS' recognition, this drug is used to treat severe, chronically increased muscle tension that has not been treated with standard drugs for progressive neurological diseases in the brain and spinal cord accompanied by physical and mental symptoms. The scope of benefit has been clarified as the MFDS' permission for Jakabi 5mg has been changed. The added indication is a non-antibiotic therapy. Benefits are expanded in the general principle of oral chronic hepatitis B treatment. The MOHW and the Korea Appraisal Board deleted the taboo and expanded the benefit, considering that the permission for "administration of patients with mild, moderate, and severe" of the drug was changed and that Besifovir was recommended as an initial treatment if there was a decrease in renal function. The MOHW and the HIRA made the decision in consideration of domestic and foreign permits, textbooks, clinical medical guidelines, clinical research documents, opinions of related academic societies, and approval for the use of non-reimbursement exceeding the permission of the MFDS. Looking at the newly established contents in detail, Voriconazole injections that exceed the permission can be recognized if they are administered according to the standard. Subject patients are only adult patients diagnosed with Fungal Endophthalmitis who are confirmed to have fungal infection in the blood or eyeball, and are recognized when administered in the anterior injection (50 μg/0.1ml) and in the vitreous cavity (50 μg/0.1ml~100 μg/0.1ml). Anti-inhibitor coagulant complex injection Faiva is paid more. The MOHW and the HIRA have a need for preventive therapy because antibody patients themselves have a very high tendency to bleed, and other hemophilia drugs have recently recognized preventive therapy for antibody hemophilia patients, so they decided to expand their benefits. Specifically, it is recognized up to six times per visit, and a total of 12 times per four weeks. However, if bleeding occurs after administering 12 doses every 4 weeks and visits the hospital, it can be recognized up to 2 times per visit, and a doctor's note must be attached. In the case of in-house administration, the in-house administration is included when calculating the number of administrations.
- Opinion
- [Reporter’s View] Differing indication and reimb standards
- by Eo, Yun-Ho Jun 02, 2022 05:58am
- There are some cases where the insurance reimbursement standards and the indication for a drug differs. This gap emerges as not all the uses of a drug approved by the regulatory authorities may be deemed appropriate by the insurance authorities to spend their limited insurance finances on. This is why voices of complaint often arise in the field. It is impossible for the authorities to address all the complaints, but there exist cases where the non-reimbursement measure seems unreasonable, no matter how ‘financial’ the reason may be. One representative example of this is the restrictions set on switching and the period of administration. Let us look at the reimbursement standards set for the acute myeloid leukemia (AML) treatment Xospata (gilteritinib) which was recently listed for insurance benefit. The reimbursement standards for Xospata state that patients who show partial remission or higher after 2 cycles of administration and have received prior approval to receive allogeneic hematopoietic stem cell transplantation (or have presented evidence equivalent to the condition above) are allowed to receive 2 additional cycles of treatment in consideration of the preparation period for allogeneic hematopoietic stem cell transplantation. In general, the restrictions on a drug’s dosing cycle are set based on the design of the drug’s clinical trial or guidelines set by overseas authorities. However, there is no reason to limit the period of administration for Xospata. The ADMIRAL study that was conducted on Xospata shows that the drug was designed without limitation in the administration period, and the NCCN guidelines also recommend the drug as ‘Category 1’ without restricting its period of administration. JAK inhibitors that receive attention as oral rheumatoid arthritis treatments are facing a switching issue. The JAK inhibitors approved in Korea are ' Xeljanz (tofacitinib)', ' Olumiant (baricitinib)', ' Rinvoq (upadacitinib),’ etc. However, if patients switch to a different drug while receiving another, the reimbursement for their first drug is not recognized, In other words, if the patient who had switched to a different drug expecting better treatment results wishes to reuse the previous drug after seeing poor results, they may not receive reimbursement. The anti-TNF agents that had first entered the autoimmune disease treatment market had also undergone the same process. With the issue continuously raised in the field, switching of drugs such as ‘Humira (adalimumab),’‘Remicade (infliximab),’ and ‘Enbrel (etanercept)’ were finally allowed reimbursement in 2013. In Korea, the reimbursement of drugs significantly affects prescription in the field. Most HCPs in Korea give up prescribing a drug even if it is deemed necessary for a patient if the drug is non-reimbursed. Restricting fiscal spending in areas where prescriptions are needed may be toxic. In this sense, a little more trust on the health authorities’ part in the judgment made in the field would be mutually beneficial.
- Policy
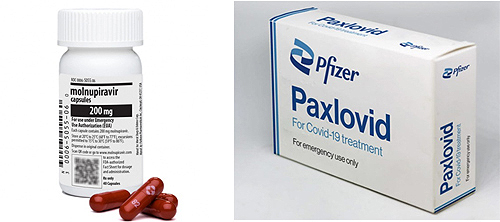
- New guidelines for Lagevrio & Paxlovid have been released
- by Kim, Jung-Ju Jun 02, 2022 05:58am
- (from left) Lagevrio and PaxlovidNew clinical guidelines for Lagevrio and Paxlovid's administration of COVID-19 patients have been released. Lagevrio considers medication if it is difficult to use other treatments among confirmed patients with mild and severe symptoms over the age of 18, and Paxlovid considers patients aged 12 or older who weigh more than 40kg. Eight member societies of the National Evidence-based Healthcare Collaborating Agency (Director Han Kwang-hyeop) and the Korean Academy of Medical Sciences (Chairman Jung Ji-tae) announced additional clinical treatment guidelines for the diagnosis and treatment of COVID-19. This guideline reflects the results of a comprehensive review of papers published around the world with the participation of the National Evidence-based Healthcare Collaborating Agency, a specialized research institute under the MOHW, and the COVID-19 diagnosis and treatment. The eight member societies are the Korean Academy of Medical Sciences, the KSID, the Korean Academy of Tuberculosis and Respiratory Diseases, the KSPID, the Korean Society of Radiology, the KSEM, the Korean Society of Critical Care Medicine, the KSLM, and the KSCM. The National Evidence-based Healthcare Collaborating Agency explained that COVID-19 new mutations are continuously occurring and that the administration of oral treatments has been clinically safe and effective in preparation for the re-pandemic this fall. Looking at the details, first of all, Lagevrio has a risk factor for severity among mild or moderate COVID-19 confirmed patients aged 18 or older, but if it is difficult to use other COVID-19 treatments, medication is considered and recommended to be used within 5 days after symptoms occur. The risk factor refers to immunodeficiency or underlying disease patients known to have a high risk of severe COVID-19 progression or low vaccination effect, and if it is difficult to use other treatments, it refers to cases where monoclonal antibodies, including Paxlovid and Remdesivir, are difficult to use. In the case of Paxlovid, medication is considered for COVID-19 infections aged 12 or older with a mild or moderate weight of 40Kg or more, and it is recommended to use it within 5 days after symptoms occur. This clinical treatment guideline was developed by applying the GRADE methodology. The National Evidence-based Healthcare Collaborating Agency searches and reviews the latest evidence every month, and if it is deemed necessary to revise the recommendations quickly, it is revised and announced every three to four months after discussion by the Committee of Medical Guidelines composed of representatives of the Association. Anyone can download this recommendation from www.neca.re.kr, Research Information >> COVID-19 Living Guideline. Han Kwang-hyeop said, "The number of COVID-19 confirmed cases is gradually decreasing, but COVID-19 new mutations continue to appear, so it is important to prepare through scientific evidence." He said, "In the future, the National Evidence-based Healthcare Collaborating Agency plans to continue to develop clinical care guidelines based on the latest evidence in cooperation with the government and the medical community."
- Policy
- The GMP reinforcement bill passed the plenary session
- by Lee, Jeong-Hwan Jun 02, 2022 05:58am
- Sustainable Support for New Drug Development Pharmaceutical Companies Increasing Punishment for Violations of GMP A bill to extend special cases for innovative pharmaceutical companies and a bill to significantly strengthen the level of GMP regulatory management compared to the previous one passed the plenary session of the National Assembly on the night of the 29th. With the revision of the Special Act on the Promotion and Support of the Pharmaceutical Industry, the deadline for special provisions on the construction of research facilities for innovative pharmaceutical companies and exemption from various levies will be extended to March 30, 2032. It is expected that it will continue to support innovative pharmaceutical companies that lead the development of new drugs in the future and boost investment in the biohealth industry. The level of regulation will increase as the National Assembly passes the revision of the Pharmaceutical Affairs Act, which promotes the improvement of the quality of domestic distributed drugs by strengthening the standards for drug manufacturing and quality management. The standards for drug manufacturing and quality management are strengthened to secure the effectiveness of the system and strengthen the international competitiveness of products, ▲ upgraded the basis for determining compliance with drug manufacturing and quality control standards (GMPs) to the law, ▲ establishment of sanctions standards for serious violations, such as being judged to be suitable for GMP by false or illegal means, ▲ appointment of GMP investigators and grounds for entry, and ▲preparation of the basis for publication of the details of disposition to drug-related business operators whose administrative disposition has been confirmed. Currently, in order to manufacture and sell medicines, the MFDS stipulates that they are suitable for GMP by formulation or manufacturing method, but it was raised to the law to strengthen the management of serious violations related to GMP. Sanctions standards have been established for serious violations related to GMP-related violations, punitive fines, and penalties of up to 5 years or 50 million won. In order to increase the administrative efficiency of GMP investigation and evaluation work, GMP education and training were appointed as manufacturing and quality control investigators for medicines, etc., and provided a basis for entry and investigation. The head of the MFDS has prepared a basis for informing the public of the violation and the details of the disposition to those whose administrative disposition has been confirmed.
- Company
- Commercialization of ADC Enhertu is imminent in Korea
- by Eo, Yun-Ho Jun 02, 2022 05:58am
- The HER2-directed antibody-drug conjugate (ADC) ‘Enhertu’ is expected to be commercialized in Korea soon. According to inducts sources, Daiichi Sankyo Korea and AstraZeneca Korea’s human epidermal growth factor receptor 2 (HER2)-directed antibody-drug conjugate (ADC) ‘Enhertu (trastuzumab deruxtecan),’ is expected to be approved within this year. Enhertu, which has indications for breast and gastric cancer, was jointly developed by AstraZeneca and Daiichi Sankyo and was first approved for the treatment of recurrent metastatic HER-positive breast cancer in the U.S. in 2019. Afterward, Enhertu was additionally approved in the US for the treatment of locally advanced or metastatic HER2-positive gastric or gastroesophageal junction (GEJ) adenocarcinoma who have received prior standard treatment with ‘Herceptin(trastuzumab).’ The drug was also granted the Breakthrough Therapy Designation (BTD) in the US, and is referred to as a second-generation ADC. As Roche’s ADC ‘Kadcyla (trastuzumab emtansine)’ failed to demonstrate its efficacy in gastric cancer, the expectations in the field have been rising on the possibility of prescribing Enhertu in gastric cancer. Although HER2 protein is generally expressed in breast cancer, it is also found in some other types of cancer. One out of five gastric cancer patients in the U.S. is diagnosed with advanced HER2-positive gastric cancer. The efficacy of Enhertu in gastric cancer was demonstrated through the Phase II DESTINY-Gastric01 trial. In the study, 26 patients were randomly assigned to receive Enhertu or the investigator’s choice of chemotherapy (paclitaxel or irinotecan) that was administered by intravenous infusion every 3 weeks. The clinical trial on gastric cancers was conducted on patients with HER2-positive locally advanced or metastatic gastric or gastroesophageal junction cancer whose disease had progressed after he/she had received at least two previous regimens including Enhertu and fluoropyrimidine-based chemotherapy. Results showed that the median overall survival (OS) in the Enhertu group was 12.5 months, which was superior to 8.4 months in patients that received the investigator’s choice of therapy. The Objective Response Rate (ORR) was 40.5% in the Enhertu group and 11.3% in the paclitaxel or irinotecan group. The median duration of confirmed objective response was 11.3 months in the Enhertu group, demonstrating its efficacy compared to the 3.9 months in the group that received the investigator’s choice of therapy. Meanwhile, the Ministry of Drug and Food Safety is reviewing the results of the DESTINY-Breast01 trial that evaluated the drug’s efficacy in the third or later line treatment of metastatic stage patients, and expansion of Enhertu’s approval in the second line will take place in the future.
- Policy
- 22nd new homegrown drug Acelex adds side effect after PMS
- by Lee, Hye-Kyung May 31, 2022 06:06am
- Crystal Genomics’ ‘Acelex Tab (polmacoxib)’ that was approved as the 22nd homegrown new drug in Korea will be adding ‘strokes, etc’ to its list of adverse events. The Ministry of Food and Drug Safety announced that it had completed the opinion review for the Change in Acelex's Indications based on Reevaluation Results (draft) and will be reflecting the changes in the indication from August 26th. Acelex was approved on February 5th, 2015 as an anti-inflammatory analgesic to relieve the symptoms of osteoarthritis patients. The drug was also designated the 22nd new homegrown drug to receive marketing authorization in Korea. The drug has been supplied to large general hospitals and university hospitals through Dong-A ST since September 2015, and Crystal Genomics also signed a joint marketing agreement for its sale with Daewoong Pharmaceuticals in March 2018. The post-marketing surveillance on Acelex was conducted for 6 years on 3,289 patients for the MFDS reevaluation in Korea. The PMS results showed that the incidence of adverse events regardless of their causality was 7.66% (252 of 2289 patients, 285 cases). Among these adverse events, one serious adverse drug reaction whose causal relationship with the drug could not be ruled out, a stroke, was reported with an incidence rate of 0.03%. The incidence rate of unexpected drug adverse events whose causal relationship with the drug could not be ruled out was 0.43% (14 of 3289 patients, 14 cases), where rare cases of stroke, paresthesia, hypoesthesia, rash, drug rash, skin erosion, tachycardia, conjunctival congestion, gastrointestinal disorders, abdominal distension, decreased appetite, daydreams, and difficulty urinating were reported. The MFDS said “We ask relevant groups to be aware of the change and to disseminate the news to their member and non-member companies. Also, institutions involved in pre-or post-management of pharmaceuticals should use the changed order for the review of civil documents such as marketing authorization of pharmaceuticals as well as for pharmaceutical surveillance monitoring-related tasks such as post-management of labels.
- Company
- Evusheld to be introduced in Korea
- by Eo, Yun-Ho May 31, 2022 06:06am
- The introduction of the COVID-19 prevention drug Evusheld in Korea has been confirmed. The KDCA supplementary budget of 4.9083 trillion won was confirmed yesterday (29th), which includes securing a shortage of living support, paid vacation expenses, quarantine hospitalization, and home treatment expenses due to a surge in confirmed cases. The government has decided to spend 39.6 billion won of the budget to introduce a new 20,000 doses of Evusheld by AstraZeneca, an antibody treatment for the purpose of preventing COVID-19 infection and severe immunity. As a result, Evusheld, the first antibody complex for preventing the COVID-19 in Korea, is expected to be prescribed to immunodeficiency patients. However, it is believed that the subject of administration will be limited. The government limited Evusheld to patients undergoing severe immunosuppressive treatment who necessarily needed preventive antibody treatments. Those with a history of COVID-19 confirmation were also excluded. As a result of the report, it was confirmed that the health authorities expressed their intention to discuss increasing the use of reserve funds in consideration of the future epidemic situation and the need to expand the number of subjects and re-administration. The U.S. FDA approved Evusheld last December. European EMA confirmed its introduction in March. In addition, France, Australia, and Singapore have completed pre-purchase contracts for Evusheld for the purpose of preventing COVID-19. An official from AstraZeneca said, "We hope it will help immunodeficiency patients such as blood cancer and organ transplant patients who were in the blind spot of COVID-19 prevention, and we will continue to do our best to provide smooth domestic supplies." Through various studies published in Nature Medicine, it has been confirmed that Evusheld is the only anti-antibiotics that maintain neutralization activity against omicron mutations. As a result of phase 3 PROVID study, Evusheld reduced the risk of symptomatic COVID-19 infection by 77% compared to placebo in the first analysis, and the risk was reduced by 83% in 6-month follow-up. During the 6-month follow-up period, there were no severe or fatal cases in the Evusheld medication group. More than 75% of the subjects who participated in the study had comorbid diseases that are likely to develop into severe diseases in the event of COVID-19, such as weakened immune systems at the time of baseline or did not show sufficient immune response to vaccination.