- LOGIN
- MemberShip
- 2026-05-02 00:25:46
- InterView
- Approval called for drug switching in atopic dermatitis
- by Whang, byung-woo Jan 24, 2025 05:51am
- Atopic dermatitis treatment settings are changing quickly. New treatment options are available as new drugs with fewer side effects and superior treatment effectiveness than existing therapies emerge. There is a growing interest in how atopic dermatitis can be treated rather than just treating it, as changes have been brought to the treatment paradigm. According to health experts, considering many factors contibute to the nature of atopic dermatitis, even if the same medication is used, the treatment effects may vary by patient. Dr. Yang Won Lee, Professor in the Department of Dermatology at Konkuk University Medical Center, who has the latest expertise in this field stresses, the importance of treatment choice based on potential treatment effects and side effects and the need for improvement to the system. "New drugs for atopic dermatitis have shifted the treatment paradigm" Dr. Lee says the most significant change he has experienced following the introduction of new drugs for atopic dermatitis is the treatment effects and patient awareness. Dr. Yang Won Lee, Professor in the Department of Dermatology at Konkuk University Medical Center"In the past, patients were reluctant to receive therapy or even avoid getting one because of the notion that atopic dermatitis treatment is not efficacious and long-term steroid therapy may result in side effects. However, the launch of biological agents and targeted therapies such as JAK inhibitors have changed the care settings," Dr. Lee says. New drugs for atopic dermatitis are good news because treatment effects vary greatly depending on the patient's sensitivity. "Atopic dermatitis is a multifactorial disease where it is not affected by just a single factor but caused by various contributing factors, such as genetic factors, skin barrier issues, and dysfunctional immune responses," Dr. Lee stated. "Due to varying patient sensitivity, even if the same medication is used, patients may experience varying treatment effects." For instance, it means that even if biological agents or JAK inhibitors of the same class are used, patient treatment can differ depending on the mechanism. More treatment options became available this year and will likely change the treatment setting. On January 9, Lily Korea's Ebglyss (ingredient name: lebrikizumab), used to treat moderate to severe atopic dermatitis, was launched. Ebglyss was launched six months after obtaining approval from the Ministry of Food and Drug Safety (MFDS) in August 2024. It is a new biologic treatment that selectively blocks interleukin (IL)-13. The efficacy and safety profile of Ebglyss have been confirmed in the Phase 3 clinical trials. Once a patient meets the clinical response after 16 weeks treatment, the drug can be administered every 4 weeks with a maintenance dose (250 mg). Dr. Lee focused on fewer side effects associated with Ebglyss compared to conventional therapies. "While long-term use of cyclosporine or steroids can lead to various side effects, Ebglyss, a biological treatment, is relatively free from such concerns in terms of side effects. It has the advantage of long-term prescriptions and greater efficacy than existing treatments," Dr. Lee says. The basis of approval for Ebglyss was ADvocate-1 and ADvocate-2 Phase 3 studies. According to the results, the most common adverse reactions are conjunctivitis (6.9%), injection site reactions (2.6%), allergic conjunctivitis (1.8%), and dry eye (1.4%). Although dupilumab, a representative treatment for atopic dermatitis, is effective, some patients may experience side effects, including worsening conjunctivitis or facial and neck dermatitis. Therefore, therapies like Ebglyss are seen as potential alternatives. "Compared to dupilumab, Ebglyss shows a relatively lower frequency and severity of side effects such as conjunctivitis or facial and neck dermatitis," Dr. Lee said. "Additionally, if clinical response is achieved, Ebglyss can be administered monthly after 16 weeks. If the efficacy and side effects are comparable, drug adherence can be considered to reduce patient burden." Switching drugs for atopic dermatitis treatment has limitations…"We must provide broader range of treatment options to provide patient-customized treatments" However, there are challenges to overcome to achieve this. The remaining issue is a 'drug switching' between treatments. Last year, the Korean Atopic Dermatitis Association (KADA) submitted a statement to the health authority illustrating that drug switching should be allowed in the field of atopic dermatitis. The government is reviewing this matter, but it is expected to take time. The reimbursement criteria for special cases of atopic dermatitis patients switching treatments are complicated. For example, if patients transition from a biological therapy to a JAK inhibitor, they must meet complicated eligibility requirements from the beginning. Similarly, the same applies when switching from a JAK inhibitor to a biological therapy. "Patients often feel discomfort from the side effects of their current treatments, but due to the reimbursement requirements, which mandate waiting periods of three to four months, they often give up on switching therapies," Dr. Lee explained. "Meeting these conditions is burdensome, leaving patients in a situation where they must continue treatments despite experiencing side effects." "From the patient's perspective, having a broader range of treatment options is essential, and from the doctor's perspective, it is crucial to have multiple tools to combat a disease like atopic dermatitis. Therefore, the drug switching is vital," Dr. Lee emphasized. "If drug switching becomes more accessible, doctors will have a wider range of options for treatment, and patients can look forward to achieving better therapeutic outcomes." Dr. Lee particularly mentioned that if drug switching for atopic dermatitis is approved, the treatment options among drug types and the number of limitations must be discussed. "Patients who do not respond to existing treatments should be able to switch medications immediately. They should be able to choose from the same drug type, such as a biological agent or JAK inhibitor," Dr. Lee says. "In my opinion, not limiting the number of possible drug switching will broaden the range of treatments." "Drug switching is permitted for psoriasis, which is the same inflammatory skin disease. However, unlike psoriasis, atopic dermatitis is significantly limited. Like psoriasis, the treatment setting for atopic dermatitis should improve quickly," Dr. Lee said. Ultimately, Dr. Lee advised actively treating atopic dermatitis with a wider range of treatment options. "In the past, patients treated for atopic dermatitis often experienced several side effects or did not fully benefit from their treatment. However, new drugs provide opportunities for patients. Because many opportunities are open for patients with severe disease who need treatment, we hope patients will participate in getting treatment," Dr. Lee said.
- Policy
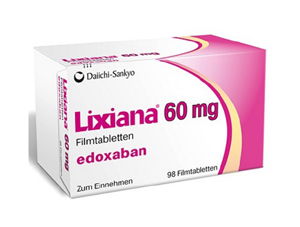
- 'Lixiana' patent expires next year, good news for
- by Lee, Hye-Kyung Jan 24, 2025 05:51am
- Product photo of As the patent of 'Lixiana (edoxaban),' a Direct Oral Anti-Coagulant (DOAC), is set to expire, generic companies are entering the competition. On January 21, the Ministry of Food and Drug Satefy (MFDS) approved bioequivalence tests, which are open-label, randomized, two groups, single time-point, cross-over Phase 2 trials involving healthy adult study participants oral administration on an empty stomach, for Hanlim Pharm's 'Edoxaban' and Daiichi Sankyo Korea's 'Lixiana Tab 60 mg.' The Lixiana patents listed in the Ministry of Food and Drug Safety's (MFDS) patent registry are two types: the substance patent, which is set to expire on November 2026, and the composition patent, which is set to expire on August 2028. For the Lixiana composition patent, 10 companies, including Boryung, Dong-A ST, HK inno.N, Samjin Pharm, Shinil Pharma, Chong Kun Dang, Kolmar Pharma, Kolmar Korea, Hutex, and Hanmi Pharmaceutical, have successfully avoided the patent through passive rights scope confirmation trials. Korean companies that won patent avoidance can launch their generics when the substance patent expires in November 2026. To meet this schedule, these companies have been applying for approvals of generic Lixiana and bioequivalent tests since 2023. Dong-A ST was the first to obtain approval for its generic version of Lixiana in 2021. However, despite winning patent avoidance, the company did not meet the requirement for being the first request for trial, so it failed to obtain a right to priority sale. Without a right to priority of sale, 12 companies, including Genuone Sciences, Genu Pharma, Hutex Korea Pharmaceutical, Handok, Shinil Pharma, Ahngook Pharm, Shinpoong Pharm, Nexpharm Korea, Samsung Biologics, Korea Prime Pharm, and Union Korea Pharm, have received approvals for 29 generic products. Additionally, as companies such as CMG Pharmaceutical, Hanlim Pharm, Theragen Etex, and Vivozon conduct bioequivalence tests, competition intensifies in the generic market even before product launches. Lixiana has been ranked top for a long time in the DOAC market, with yearly prescription sales of KRW 100 billion. According to a pharmaceutical market research firm, UBIST, Lixiana's prescript sales for 2023 amounted to KRW 105.3 billion, up 9% from KRW 96.7 billion in 2022. It recorded KRW 55.7 billion in the first half of last year. Lixiana has maintained a leading position in the market since it ranked first in 2019. Analysis suggests that joint sales with Daewoon have contributed positively to the increase in prescription sales.
- Policy
- Hokunalin Patch has been removed from Korea’s reimbursement
- by Lee, Tak-Sun Jan 24, 2025 05:51am
- Hokunalin Patch, asthma and bronchitis deodorant based on tulobuterol, will be removed from the reimbursement list. The move was made upon the expiry of the item’s marketing authorization last month. As a result, only domestic generic versions of the drug are available in South Korea. According to industry sources on Jan. 22, Abbott's 3 versions of Hokunalin Patch (0.5 mg, 1 mg, and 2 mg) will be removed from the reimbursement list on Jan. 1 next month. The drug's domestic license expired on Jan. 1. The medication is an adhesive strip that is applied to the skin. It is characterized by its 24-hour effectiveness after a single application. In particular, it is used for infants and children suffering from asthma as well as acute and chronic bronchitis, as it has fewer side effects than oral medications and can be used from the age of 6 months. Abbott has already announced that it will discontinue the domestic supply of Hokunalin in 2022. The reasons included the end of the contract with the manufacturer and the rising cost of raw materials. However, the company had maintained the domestic marketing authorization for the drug, and with the expiration of the license and the removal from the reimbursement list, Hokunalin Patch will disappear into history. However, there is a six-month grace period before August 1 to apply for insurance reimbursement benefits. The withdrawal of Hokunalin from the domestic market was also due to the emergence of generic drugs. Generic versions of the drug have been available since 2010, and 19 pharmaceutical companies currently maintain licenses. The use of Tulobuterol Patch has been increasing due to the recent surge in respiratory diseases. In response, the government designated the transdermal tulobuterol patch as a national essential medicine in November 2023. In addition, in March last year, the upper insurance price limit was raised by up to 27.2% to address the supply-demand imbalance. However, the field reports that there is still a shortage of tulobuterol deodorant. The number of products being withdrawn is also increasing. From August 2023 to January this year, 21 products from nine pharmaceutical companies did not renew their licenses. “Tulobuterol patch is a product with a high production cost and not much profit,” said an industry official. ”Recently, the price of raw materials has risen, and due to intensified competition between generic companies, more and more companies are leaving.”
- Policy
- Govt plans to expand CDMO capacity by 2.5 times
- by Lee, Hye-Kyung Jan 24, 2025 05:51am
- The government plans to expand Korea’s CDMO manufacturing capacity 2.5 times by 2032 to become the No. 1 global CDMO by 2032. It will also support domestic companies with a 'megafund' by expanding the size of the K-Bio-Vaccine Fund, currently at KRW 600 billion, to over KRW 1 trillion. The government launched the National Bio Commission in anticipation of the global bio market growing to KRW 3.3 trillion by 2027. According to the 'Vision and Mission of the National Bio Commission (draft)’ released at the first meeting, the government will quickly create mega funds worth more than KRW 1 trillion, such as the K-Bio-Vaccine Fund, for early investment and scale-up of bio companies, and build a growth ladder by expanding policy finance and trade insurance support, including preferential interest rates and expanded loan limits. To promote R&D activities of companies, the government will activate M&As and expand tax credits by adding bio-related technologies such as bio buffer and bio jet fuel to the list of national strategic technologies. Also, it will support corporate growth by expanding support bases such as K-Biodesk and Boston CIC (Cambridge Innovation Center) in major overseas countries, along with management consulting and overseas certification support. Sung-hyuk Yoon, Director General of the Ministry of Trade, Industry and Energy “The KRW 1 trillion megafund will be based on the KRW 50 billion Korea Fund for startups in the bio sector, the KRW 300 billion fund created by the Ministry of Health and Welfare, and the KRW 100 billion fund dedicated to green bio and food tech by the Ministry of Agriculture, Food and Rural Affairs,” said Sung-hyuk Yoon, Director General of the Ministry of Trade, Industry and Energy's Advanced Industry Policy Bureau. The K-Bio-Vaccine Fund, which the government has been funding since 2023, is worth KRW 306.6 billion. The third and fourth funds are currently being established, and if the fifth fund is added this year, it will total KRW 600 billion. To expand private investment, the government will reactivate M&A by abolishing the obligation to sell VC stakes and providing loan guarantees for acquisition funds, and expand tax credit benefits by adding bio-related technologies such as bio buffers and bio aviation oil to the list of national strategic technologies. In addition, by 2032, domestic CDMOs plan to expand their production capacity to 2.5 times the current level. “The production capacity of CDMOs in Songdo, Incheon is about 870,000 liters,” said Yoon, adding, ”Domestic CDMOs such as Samsung Biologics are planning to invest KRW 21 trillion, and if this amount is executed, the production capacity will increase to 2.16 million liters by 2032, making it possible to rank first in the world in terms of sales and capacity.” To support the manufacturing base of domestic biotech companies that have the technology to discover new drug candidates but do not have production facilities, the government plans to utilize five public CDMOs already established to help them commercialize their technologies, including cell line manufacturing, sample, and finished product production. In addition, the government will build a Korean bio cluster. It plans to promote convergence between various fields such as red, green, white, and blue bio by linking high-tech medical complexes, R&D zones, and industrial complexes nationwide, and create an ecosystem that runs from R&D to commercialization by attracting key institutions (universities, research institutes, companies, and hospitals). In addition to establishing a 'Bio Cluster Commission’ under the National Bio Committee, it will jointly utilize regional equipment, experts, and startup support programs through a 'virtual platform' connecting 20 clusters, and expand exchanges with leading clusters overseas. “The health and medical sector has been investing and industrializing for so long that there are currently about 18 clusters across the country,” said an MOHW official. ”We cannot physically gather all of them like the Boston cluster in the United States, so we will create a system to share resources based on the virtual platform, such as the facilities, equipment, and incubation centers that each cluster has.”
- Company
- The era of compound management for obesity
- by Whang, byung-woo Jan 23, 2025 05:54am
- Wegovy (semaglutide), an obesity drug hailed as a game-changer, is seeking synergy with its cardiovascular benefits in addition to weight loss. Experts say that the obesity treatment paradigm is already evolving beyond weight loss to overall health care. In the future, it will be a challenge to improve access through expert review and reimbursement. (from the left) Soo Lim, Professor of endocrinology and metabolism at Seoul National University Bundang Hospital, Jong Chan Yoon, Professor of Cardiology at Seoul St. Mary Novo Nordisk held a media session on the 21st of this month to highlight the weight loss and cardiovascular disease risk reduction effects of Wegovy. Wegovy is an obesity treatment that is administered once a week. It is licensed in Korea as an adjunct to weight loss and weight management in patients with weight-related comorbidities. In July last year, the indication was expanded to include the risk reduction of major cardiovascular events (cardiovascular death, nonfatal myocardial infarction, or nonfatal stroke) in overweight and obese patients with established cardiovascular disease. “The 20% reduction in major adverse cardiovascular events (MACE) seen with semaglutide in adult obese patients with cardiovascular disease is a significant step forward in the treatment of obesity,” said Julie Broe Honore, Senior CMR (Clinical, Medical, and Regulatory) Director at Novo Nordisk Korea. “From a clinical perspective, semaglutide is a treatment option that can be used in conjunction with diet and increased physical activity to help manage weight and reduce the risk of major cardiovascular events.” Korean experts also emphasize the importance of active treatment interventions as obesity is a chronic disease that goes beyond simple weight gain and leads to multiple metabolic and cardiovascular complications. “The comorbidities of diabetic patients in Korea include hypertension (60%), kidney disease (40%), hyperlipidemia (70%), and obesity (50%),” said Dr. Soo Lim, Professor of endocrinology and metabolism at Seoul National University Bundang Hospital. ”It is time to control multiple diseases together, and I think GLP-1 will be the best medicine to manage them all at once.” Jong Chan Yoon, Professor of Cardiology at Seoul St. Mary Jong Chan Yoon, MD, Professor of Cardiology at Seoul St. Mary's Hospital, noted that obesity significantly increases the risk of cardiovascular disease. “Cardiovascular disease is one of the leading causes of death globally, and after analyzing data from 195 countries around the world from 1990 to 2015, we found that about two-thirds of obesity-related deaths were associated with cardiovascular disease,” said Professor Yoon. In the SELECT study, Wegovy demonstrated a significant 20% reduction in the risk of major cardiovascular events in the Wegovy arm compared to the placebo arm over a median follow-up of 39.8 months. “The results of the SELECT study demonstrate that the treatment goals for obesity can be expanded beyond simple weight loss to include reducing the risk of major cardiovascular events,” said Professor Yoon. In particular, Professor Lim emphasized that the results of the study showed that GLP-1 class drugs have greater cardiovascular benefits than other diabetes medications. “When we directly compared the cardiovascular disease reduction effects of GLP-1 and SGLT2, GLP-1 showed better benefits, and in stroke, SGLT2 had no effect, but GLP-1 showed good effect,” Lim analyzed. However, Wegovy, which was launched in Korea in October last year, is currently non-reimbursed and has a cost hurdle. Apart from its clinical benefits, it is unclear how much it can be utilized in practice considering its cost-effectiveness. Soo Lim, Professor of endocrinology and metabolism at Seoul National University Bundang Hospital. In response to this, Professor Lim said, “It is good to use a lot of good drugs, but there is a limit to their use because they are not reimbursed. The government needs to also take the view that severe obesity is a disease,” adding, ”If a person with a BMI of 35 or 40 or more is socially disadvantaged due to obesity, it is necessary to consider providing some support through insurance.” “Although the treatments are also expensive in the U.S., if you consider the cost of procedures and hospitalization due to cardiovascular diseases, papers that show that treatments like Wegovy are rather cost-effective,” said Professor Yoon. ”I expect prescriptions to gradually increase in Korea, and if there is no cost hurdle, it will likely be used without limitation within the given indications.” In the end, the will of pharmaceutical companies in attempting reimbursement will be important to resolve the cost issue. In this regard, Novo Nordisk is considering mid- to long-term plans to strengthen access. “We understand that it is necessary to expand access to therapeutics, but this is something that needs to be discussed extensively,” said Ju Ok Lim, head of Medical Affairs at Novo Nordisk. ”It requires mid- to long-term discussion, but we are in the beginning stages of the process and are exploring various ways to expand access.”
- Policy
- If atrial fibrillation develops, Rx 'Omega-3' API
- by Lee, Hye-Kyung Jan 23, 2025 05:54am
- Product photo of Kuhnil The administration of the active ingredient 'omega-3-acid ethyl esters90,' which is used for treating dyslipidemia, will be permanently discontinued if a patient develops atrial fibrillation. Starting February 10, the Ministry of Food and Drug Safety (MFDS) will modify the approval specification based on the results of the European Medicines Agency (EMA)'s safety assessment and monitoring of 'omega-3-acid ethyl esters90.' Last year, EMA's Pharmacovigilance Risk Assessment Committee (PRAC) agreed to add atrial fibrillation (irregular, rapid contraction of the heart) as a common side effect to the product information for medicines containing omega-3. The review result of a Periodic Safety Update Single Assessment (PSUSA) procedure conducted by the PRAC highlighted a dose-dependent increased risk of atrial fibrillation in patients with established cardiovascular disease or cardiovascular risk factors treated with omega-3 compared to placebo. The atrial fibrillation risk is highest with a dose of 4g per day. EMA concluded that if atrial fibrillation develops, omega-3 treatment should be permanently discontinued. The MFDS also concluded to reflect such change on the approval specification for omega-3-acid ethyl esters (monotherapy) and will include the clause, 'atrial fibrillation has been reported to be a common side effect' in the adverse reaction section. Additionally, the MFDS will add a clause that 'a dose-dependent increased risk of atrial fibrillation in patients with established cardiovascular disease or cardiovascular risk factors is observed. If atrial fibrillation develops, omega-3 treatment should be permanently discontinued' in the general safety section. Omega-3-acid ethyl esters90 is the only prescription medicine available containing omega-3 fatty acids and Kuhnil's 'Omacor' in the original drug. 56 Omacor generics have been approved. Omacor is a medicine made of omega-3 fatty acids extracted from fish oil. It is used for the treatment of hypertriglyceridemia. According to a pharmaceutical market research firm, UBIST, Omacor's outpatient prescription sales were reported to be KRW 34.6 billion.
- Policy
- MFDS approves immune thrombocytopenia drug Tavalisse
- by Lee, Hye-Kyung Jan 23, 2025 05:54am
- The Ministry of Food and Drug Safety (MFDS) announced on the 20th that it has approved Tavalisse Tab 100 mg (fostamatinib sodium hydrate), an orphan drug used to treat immune thrombocytopenia in adults. Adult immune thrombocytopenia is an autoimmune disease in which antibodies against platelets are produced and platelets are destroyed in the spleen, resulting in petechiae and purpuric spots on the skin. Tavalisse inhibits the activation of spleen tyrosine kinase (Syk), which inhibits the production of antibodies against platelets, thereby preventing platelet destruction while also interfering with platelet ingestion by macrophage. Spleen tyrosine kinase is a non-receptor tyrosine kinase that is widely present in blood cells and plays an important role in activated signal transduction within beta cells of the pancreas. The MFDS said, “We expect the treatment to provide a new treatment opportunity for patients with adult immune thrombocytopenia who have had an inadequate response to previous treatments,” the agency said. In July 2023, Tavalisse was designated as a priority review target and fast-tracked for rapid introduction into Korea. In particular, JW Pharmaceutical applied for health insurance reimbursement benefits to the Health Insurance Review and Assessment Service last year using the approval-evaluation linkage system. JW Pharmaceutical signed a contract with Japan's Kissei Pharmaceutical in 2021 for the domestic distribution right to Tavalisse. The original developer is the U.S. company Rigel Pharmaceutical, and Kissei acquired the development and exclusive commercialization rights for the Korean, Japanese, Chinese, and Taiwanese markets in 2018. “We will continue to make the best efforts to quickly supply new treatments to patients with rare and intractable diseases based on our regulatory science expertise, to expand treatment opportunities for the patients,” said the MFDS.
- Company
- MET inhibitor Tepmetko enters last stage to reimb in KOR
- by Eo, Yun-Ho Jan 23, 2025 05:54am
- MET-targeted anticancer drug ‘Tepmetko’ has entered the final gateway to insurance reimbursement nearly 3 years after its domestic approval. According to industry sources, Merck Korea has recently started negotiating drug prices with the National Health Insurance Service for its Tepmetko (tepotinib), a treatment for locally advanced or metastatic non-small cell lung cancer with a confirmed MET exon 14 skipping mutation. The drug passed the Health Insurance Review and Assessment Service’s Drug Reimbursement Evaluation Committee review in December last year. Tepmetko obtained domestic approval and went through the reimbursement process in 2021 at the same time as the same mechanism drug Tabrecta (capmatinib). However, no MET cancer drug has been listed for reimbursement in Korea yet. Therefore, it remains to be seen whether Tepmetko will complete the reimbursement process. The drug has failed to meet the criteria for insurance reimbursement twice, including failing review by the Health Insurance Review and Assessment Service's Cancer Disease Deliberation Committee in March. It then voluntarily suspended the coverage process and submitted another application for coverage in July, and this time, it passed the committee. This achievement was made 3 years after its domestic approval. Non-small cell lung cancer accounts for 80% of all lung cancer diagnoses. MET exon 14 skipping mutation is a rare type of cancer that is present in approximately 3-4% of these patients. In Korea, of the 1,020 NSCLC patients in Korea, 1.9% of the NSCLC patients were confirmed to have MET exon 14 deletion. Tepmetko’s efficacy was demonstrated through the VISION study, which enrolled the largest number of NSCLC patients with MET exon 14 skipping mutations. The results showed a significant life extension effect, with a median progression-free survival (PFS) of 15.3 months and an objective response rate (ORR) of 56.8 percent. The median duration of response (DoR) was 46.4 months, and the median overall survival (OS) was 25.9 months, showing continuous antitumor activity in the long term. Also, according to a presentation by Ji-Youn Han, Professor of Oncology at the Center for Lung Cancer at the National Cancer Center, which was at the Korean Association for Lung Cancer International Conference last year after analyzing 79 Asian patients that participated in the VISION study, the ORR was quite high at 66.7%, and 48.1% in the second-line treatment group. Meanwhile, Tepmetko also showed significant results in a follow-up analysis of Asian patients enrolled in the pivotal Phase III VISION study. In this analysis, Tepmetko demonstrated an objective response rate of 56.6%, a median duration of response of 18.5 months, a median progression-free survival of 13.8 months, and a median overall survival of 25.5 months. The objective response rate was 64.0% in treatment-naïve Asian patients, confirming previous findings that the first dose was more effective. No new safety information was identified, with 39.6% of patients experiencing grade 3 or higher adverse events. In addition, Tepmetko has now passed the Drug Committees (DCs) of more than 30 medical institutions nationwide, including the Big 5 tertiary hospitals - Samsung Medical Center, Seoul National University Hospital, and Seoul St. Mary's Hospital, Seoul Asan Medical Center, and Sinchon Severance Hospital.
- Company
- "C-Trelin proven effective for SCD treatment…reimb needed"
- by Lee, Seok-Jun Jan 22, 2025 05:55am
- Product photo of There is currently no standard therapy available for 'spinocerebellar degeneration (SCD).' Doctors write prescriptions based on a patient's condition, but there are still unmet needs in 'SCD treatment.' Patients affected by disease likewise. SCD is a degenerative disease affecting the cerebellum or spinal cord due to various underlying causes. Ataxia and dysarthria commonly occur at an early stage, but the disease progression accompanies complications, including dysfunction of the heart, lungs, spine, and bones, causing the risk of death. Other diseases have specific medications; for instance, 'Levodopa' is used for Parkinson's disease and 'aspirin' is used for cerebral thrombosis. However, there is currently no major medication available for SCD. This is because no medication has been proven to improve symptoms or slow down disease progression. The efficacy and safety of C-Trelin Orally Disintegrated Tab (taltirelin hydrate) was demonstrated in large-scale Phase 4 clinical trials in patients with SCD. Dr. Seong Beom Koh, Professor at the Korea University Guro Hospital's Department of Neurology who led the Phase 4 clinical trial, stated, "None of the medications used in patients with SCD are better than C-Trelin Orally Disintegrated Tab." "C-Trelin Orally Disintegrated Tab has proven effective and safe in patients with SCD. Its oral formulation also yields favorable patient compliance. It means that it is a valuable treatment for patients. We should widely use this medication through reimbursement." The efficacy and safety of the drug has been demonstrated in the large-scale Phase 4 trial The clinical trial was published in the Journal of Movement Disorder under the title, 'The Efficacy and Safety of C-Trelin Orally Disintegrated Tab in Patients with ataxia induced by SCD.' The clinical trial involved 160 study participants, including randomly assigned 79 experimental group and 81 control group, and the results showed a significant reduction in the K-SARA (Korean version of Scale for the Assessment and Rating of Ataxia), objective evaluation index for ataxia at 24 weeks treatment, thus confirming the statistical significance of the drug. The standing up and impairment in language entries showed that the average difference in K-SARA of the experimental group was statistically lower than that of the control group. While no suitable medication is available for ataxia in SCD, publication of the Phase 4 trial results in the international journal is significant. Doctors are stating the importance of providing reimbursement for C-Trelin Orally Disintegrated Tab. "C-Trelin Orally Disintegrated Tab is an oral tablet required twice daily. This drug had no significant issue based on the clinical trial evaluating the efficacy and safety. Patient tolerance was favorable, and there were no side effects after administration. To date, no other treatments administered to patients with SCD are said to be efficacious than this drug. Therefore, this drug provides a valuable treatment option for patients with SCD," Dr. Koh remarked. "C-Trelin Orally Disintegrated Tab may require long-term use, which can be costly. This drug should be considered for patients with good mobility and could benefit from it. If a drug is as effective as this, we should pursue active treatment options through reimbursement," Koh added. 'C-Trelin Orally Disintegrated Tab 5 mg' obtained the Ministry of Food and Drug Safety (MFDS) approval in 2015. It costs KRW 4,900 per tablet as a non-reimbursable drug. Two tablets per day cost KRW 9,800 daily and cost KRW 3.5 million yearly. Doctors say that without National Health Insurance reimbursement coverage, it is practically impossible for patients, about 80% of those who do not have jobs, to continue taking the drug that requires long-term administration. Based on these clinical trial results, HLB Pharmaceutical has recently applied for reimbursement again.
- Company
- "Foreign drug price re-evaluation, unfair direct comparison"
- by Kim, Jin-Gu Jan 22, 2025 05:54am
- The Q&A session at the New Year press conference hosted by the KPBMA on January 21. Yunhong Noh, President of the Korea Pharmaceutical and Bio-Pharma Manufacturers Association (KPBMA), has criticized the government's re-evaluation of foreign drug price comparison. Noh points out that making a simple drug price comparison poses problem despite differences in socioeconomic circumstances and healthcare systems between South Korea and the countries being compared to. "South Korea faces an important turning point for leaping to the advanced country in the global new drug development field," Noh said. "The government should establish a policy by assessing comprehensive effects on the pharmaceutical and biotech industry." Noh made this statement during the Q&A session at the New Year press conference hosted by the KPBMA on January 21. The government is pursuing a measure to lower domestic drug prices compared to those from 8 countries (U.S., Japan, Germany, U.K., France, Switzerland, Italy, and Canada). The government plans to reference the average adjusted price of six countries, excluding the highest and the lowest, for lowering domestic drug prices. The government and the pharmaceutical industry established a public-private consultative body last year and discussed the specifics of this measure. The previous meeting was held in July 2024, and the government is set to announce the final draft and implement the measure soon. Even after the meeting concluded, the pharmaceutical industry continued criticizing this matter. Despite the upcoming implementation of this system, it is still being criticized. Noh has a similar opinion. "During the ten meetings, I have suggested concerns on this matter," Noh said. "Each country has a different socioeconomic situation. healthcare system, and drug price system with foundation. However, it is worrisome to compare drug prices without considering the difference in the systems." "In advanced pharmaceutical countries, generics are priced high when initially registered, then later lowered due to various mechanisms. However, in South Korea, these drugs are priced low and maintained for a long time," Noh said. "Drug prices differ by time, so it is problematic to compare drug prices at a particular time point superficially." Additionally, Noh suggested to the government to adequately consider the consequences to promote the pharmaceutical industry. "South Korea faces an important turning point for leaping to the advanced country in the global new drug development field," Noh said. "The government should establish a policy by assessing comprehensive effects on the pharmaceutical and biotech industry."