- LOGIN
- MemberShip
- 2026-04-29 13:25:05
- Company
- Hanmi's new bio drug 'Rolvedon' sales 88%↑ in the US mkt
- by Son, Hyung Min Nov 15, 2024 05:49am
- The U.S. sales of Rolvedon (Korean product name: Rolontis) have substantially increased. Hanmi Pharmaceutical's U.S. partnering company, Assertio, plans to expand market share by securing Rolvedon's indication for the same-day administration. According to Assertio on Novermber 14, Rolveron's sales in the U.S. market in Q3 were US$ 15 million (about KRW 21 billion), up 87.5% Year-over-Year (YoY). Rolvedon has recorded cumulative sales of US$110.30 million (about KRW 155 billion) since its launch in Q4 of 2022. Rolvedon is a treatment for neutropenia developed by Hanmi Pharmaceutical. It is Korea's 33rd new drug, approved in March 2021. After that, Hanmi Pharmaceutical and its U.S. partnering company Spectrum (now Assertio) obtained U.S. Food and Drug Administration (FDA) approval in September of the same year. Rolvedon was outlicensed to the U.S. Spectrum in 2012. After Assertio acquired Spectrum in April last year, it secured the licensing of sales and development of Rolvedon and lung cancer therapy Poziotinib. Rolvedon Since its release in the United States in October 2022, Rolvedon has recorded sales of $10 million within three months. After the product launch, 70 distributors purchased Rolvedon. It was then utilized by the top three community oncology networks, accounting for 22% of the clinic market share. It continued to show strong sales up to Q2 last year. Rolvedon generated sales of $15.6 million in Q1 last year, and in Q2, it recorded $21 million (approx. KRW 28 billion), an increase of 34.6% from the previous quarter. Rolvedon’s sales in Q3 last year slowed down since its launch. It recorded $8 million in Q3 last year, a decrease of 62% from the previous quarter. Regarding sales reduction, Assertio explained that the demand for Rolvedon after applying the reimbursement system was below expectations. Rolvedon successfully rebounded in Q4 last year, generating US$11 million in sales. Rolvedon's sales in Q1 amounted to US$14.50 million and maintained recovery in Q2, recording sales of US$15.10 million. Assertio has high hopes for Rolvedon's same-day administration clinical trial. Currently, Neulasta, jointly developed by Amgen and Kyowa Kirin, is recording over half of the market share in the U.S. market for neutropenia treatment. However, conventional treatments for neutropenia, such as Neulasta, can only be administered 24 hours after cancer therapy, thereby prolonging hospitalization. Assertio plans to gain a competitive edge with its strategy of Rolvedon's same-day administration method. Assertio has recently completed the Phase 1 clinical trial for Rolvedon's same-day administration. The company plans to present the clinical results at the 2024 San Antonio Breast Cancer Symposium (SABCS 2024), held for four days, starting December 10.
- Company
- Lee Hyun-ju named as the representative of ZP Therapeutics
- by Eo, Yun-Ho Nov 14, 2024 05:52am
- Lee Hyun-ju, new representative of ZP Therapeutics Korea Lee Hyun-ju (48), ex-Daiichi Sankyo Korea's Oncology Business Franchise Head, was appointed as the new representative of ZP Therapeutics Korea. Industry sources said Zuellig Pharma has recruited Lee Hyun-ju as the new head of ZP Therapeutics Korea, Zuellig Pharma's commercial services corporation. Lee holds a degree from Sungkyunkwan University's College of Pharmacy, and she has years of experience in the Korean pharmaceutical market with expertise in the anticancer business. Lee started her career in 1999, undertook roles in Handok's marketing and Sanofi's marketing, and worked as Roche Korea's Oncology Cluster Lead and Novartis Korea's Hematology Cluster Lead. Lee moved to Daiichi Sankyo's Oncology Cluster last year and served her role until the new appointment. Meanwhile, ZP Therapeutics Korea is committed to providing a comprehensive solution, encompassing marketing, sales promotion, product launch medical e-detailing, registration & approval, market access, digitalization & data analysis-based sales excellence, adapting needs and changes of the pharmaceutical market. ZP Therapeutics Korea established itself as a commercial solution partner sought by pharmaceutical clients. The company is involved in in-licensing of many prescription drugs and over-the-counter (OTC) drug brands. ZP Therapeutics Korea also provides sales & marketing services, supporting major pharmaceutical companies.
- Company
- Celltrion anticipates Trump admin will bring positive shift
- by Whang, byung-woo Nov 14, 2024 05:51am
- Celltrion anticipates that President-elect Donald Trump's second administration in the United States will positively affect the company's biosimilars and Contract Development and Management Operations (CDMO) services. On November 12, Celltrion presented stockholders with the potential impact on the business under the title 'The Business Impact and Outlook upon President-elect Donald Trump's second administration in the United States launches.' Celltrion cited a report from the Korea Institute for Industrial Economics and Trade, 'The potential impacts of the U.S. presidential election on Korean industry and outlook,' and analyzed that the Trump administration will be friendly towards using generics and biosimilars. Currently, healthcare expenditures in the United States accounted for 17.6% of the country's GDP in 2023. As a solution, Trump's first administration implemented initiatives such as the 'Lowering Drug Prices by Putting America First' administrative orders and the 'American Patients First' plan to lower drug prices. These policies include details regarding biosimilars, including 'Improve Competition' and 'Lowering List Prices.' "When President-elect Trump's second administration launches, the administration is expected to take over the policy during the first administration and pursue healthcare policy," Celltrion said. "We expect the administration will favor expanding biosimilar prescriptions, which is Celltrion's main business." Trump's first administration pursued a policy of regulating drug pricing, proposing a bill to Congress to stop pharmaceutical companies from paying rebates to the top 3 pharmacy benefit managers (PBMs) in the United States. Such a move was said to be friendly to the biosimilar market. Additionally, as the administration pursues the PVM reform to alleviate financial loss, a by-product of economic stimulation, by reducing taxes, implementing a policy that expands the use of biosimilars is expected. Such a move is expected to be favorable to Celltrion. In particular, Celltrion anticipated these policies would positively impact its CDMO service, which will be Celltrion's growth momentum. The 'Biosecure Act' bill, the U.S. Congress initiative, 'prohibits entities that receive federal funds from using biotechnology from a biotechnology company of concern may not contract with any entities that do so.' The Biosecure Act is expected to support establishing a new supply network in US-friendly countries with business competitiveness, including South Korea, Japan, and India. "In line with the U.S. industry trend, we will complete establishing the CDMO corporate entity within this year to seize the opportunity to secure demands from Chinese companies," Celltrion added. "Celltrion will secure a new manufacturing plant, as Celltrion's 100% owned subsidiary, in South Korea or overseas to expand production capacity." Additionally, as Trump's second administration's policy priority is the 'America First' agenda, Celltrion anticipates US dollar strength will likely result in trade disputes and interest rate rises. During the process, Celltrion anticipates its products will remain unaffected by tariff increases, as tariffs on drugs are exempt under the WTO's Pharma Agreement. "Upon Trump's second administration launches, we expect the company will have an opportunity to focus mainly on business aspects, marketing expansion and sales growth, compared to other type of business," Celltrion said. "Celltrion will focus on changes to the U.S. biopharmaceutical industry and generate outcomes by maximizing business opportunity," Celltrion emphasized.
- Company
- ‘Access to bispecific antibody Columvi should be improved’
- by Son, Hyung Min Nov 14, 2024 05:51am
- Dr. Chris Fox, Professor of Haematology at the University of Nottingham, U.K. “Diffuse large B-cell lymphoma (DLBCL) is a disease in which one in four patients experience relapse even after treatment. The bispecific antibody Columvi has demonstrated efficacy in relapsed patients at up to 18 months of follow-up. The clinical performance of Columvi is not just an incremental improvement over existing therapies, but a paradigm shift in the DLBCL treatment environment.” At a recent meeting with Dailypharm, Dr. Chris Fox, Professor of Haematology at the University of Nottingham, U.K., recently described so about Columbo, a bispecific antibody approved for diffuse large B-cell lymphoma in Korea. DLBCL is a disease in which the body's protective “B cells” grow or multiply uncontrollably and is the most common form of non-Hodgkin's lymphoma that accounts for about 40% of all non-Hodgkin's lymphomas. The disease is characterized by aggressive, rapidly progressive staging. The number of DLBCL patients in Korea was 14,183 as of last year, a 36% increase from the 10,428 in 2018. Up to 15% of DLBCL patients fail first-line standard therapy, and 25% of patients experience relapse within 18 months despite achieving a complete response (CR). Patients with relapsed or refractory DLBCL show a characteristically rapidly worsening prognosis as the number of treatment cycles increases. Columvi, the first bispecific antibody targeting CD20XCD3 enters the market...offers the advantage of a fixed dosing period The good news is that a variety of new drugs have emerged for this disease. Roche's Polivy, a representative DLBCL drug, is said to be effective in about two-thirds of patients when used as a first-line treatment. However, this means that about one-third of patients who do not respond to first-line treatment remain in need of further options. Bispecific antibodies and chimeric antigen receptor T-cell (CAR-T) therapy, such as Columvi, are used in such cases of relapse. Columvi, the first bispecific antibody to target CD20xCD3 in DLBCL, was launched without reimbursement in Korea in May and is now on the formulary of more than 10 general hospitals. The drug has a 2:1 structure that binds to two CD20 regions on the surface of malignant B cells and one CD3 region on immune T cells, resulting in a stronger binding. Bispecific antibodies have two targets, each targeting a different cell: one that draws immune T cells closer to malignant B cells and the other that activates the T cells to kill the malignant B cells. Based on this mechanism of action, bispecific antibodies have been shown to be effective in patients who are resistant to conventional antibody therapies or chemotherapy. “Bispecific antibodies and CAR-T therapies have been explored as treatment options for DLBCL, but without head-to-head trials, it is difficult to say which is better. The choice of treatment should be based on the individual patient's state of disease progression. However, one of the side effects of CAR-T in elderly patients, immune effector cell-associated neurotoxicity syndrome (ICANS), is considered when selecting a treatment,” said Dr. Fox. He added, “Columvi has a fixed dosing period. It is designed to be administered for up to 12 cycles (8.3 months), so there is a clear end date for the treatment. It also has the advantage of being an off-the-shelf treatment that can be administered to patients immediately.” Columvi achieves 39% CR rate - still effective after 18 months...“justifies the need for its reimbursement” Columvi demonstrated efficacy in the multicenter, open-label Phase I/II NP30179 trial in patients with relapsed or refractory DLBCL after two or more prior systemic therapies. Trial results showed that Columvi achieved a complete response (CR) of 40% and an overall response rate (ORR) of 52%. Among patients who achieved CR, the median duration of response was 26.9 months, with 67% of patients maintaining CR at 18 months. The study also included about one-third of patients who had received prior CAR-T therapy. “Columvi demonstrated a 40% CR rate in the trial, even in patients who are difficult to treat,” said Dr. Fox. This data alone confirms the efficacy of Columvi, as such data cannot be expected with existing standard treatment options, and Columvi is showing similar results in the real world to the clinical trial,” said Dr. Fox. “In DLBCL, relapse typically occurs within 12 to 18 months, and staging progresses rapidly in relapsed patients. We already have data on Columvi’s use in these patients up to 18 months of follow-up. So we can be confident about Columvi’s efficacy data and maintenance of its effect.” However, Columvi’s reimbursement was rejected in July by the Cancer Disease Review Committee, the first gateway to reimbursement in Korea, due to the lack of long-term data. Roche Korea is aiming to reapply for Columvi’s CDDC review later this year. “Patient access to Columvi has been secured in the UK with reimbursement approval,” said Dr. Fox. “This is because the health authorities have recognized Columvi as an effective treatment in DLBCL.” “Columvi is not just an improvement over existing therapies, but a paradigm-shifting treatment for DLBCL. I want to emphasize that this is a treatment that could have an impact on prolonging the survival of patients with relapsed or refractory DLBCL.”
- Company
- Global CDMOs compete to expand ADC capacities
- by Kim, Jin-Gu Nov 14, 2024 05:51am
- Global competition is heating up in the contract development manufacturing organization (CDMO) market for antibody-drug conjugates (ADCs). Major players include Switzerland's Lonza and Samsung Biologics, the world's top two CDMOs, which are competitively expanding their manufacturing facilities. Lonza recently announced the expansion of a 1,200-liter ADC manufacturing facility, while Samsung Biologics announced the start-up of a 500-liter ADC manufacturing facility within the year. According to KoreaBIO, Lonza announced on Dec. 13 (local time) that it plans to add 2 manufacturing facilities in Visp, Switzerland, to expand its 'bioconjugation' service. An additional 1,200-liter manufacturing facility will be built to produce commercial bioconjugates, including ADCs, in high volumes. At the same time, the company will expand the infrastructure of the existing facility. Construction is expected to be completed and the facility fully operational by 2028. The new manufacturing facility will provide comprehensive end-to-end lifecycle support. This includes drug manufacturing for early-stage clinical development, large-scale manufacturing for commercial supply, and finished product filling services. Lonza has been in the bioconjugate CDMO business since 2006. To date, it has produced more than 1,-00 cGMP batches for more than 70 programs. Christian Morello, Vice President, Head of Bioconjugates, Lonza, said, “We continue to see strong growth in the bioconjugates space as ADCs and other bioconjugated drugs increasingly progress towards commercialization. This investment in our multipurpose commercial bioconjugation capacity addresses the growing market demand, enables us to support the growth of our customers, and offers a flexible and integrated service for manufacturing bioconjugates.” The global CDMO market, including Lonza, has recently been intensely competing to expand capacities around ADC drugs. Samsung Biologics is building a dedicated 500-liter ADC manufacturing facility at its Songdo Biocampus in Incheon, South Korea. The company plans to finalize the construction this year and begin full-scale operation after receiving GMP approval. Lotte Biologics is expanding its ADC manufacturing facility at its Syracuse, USA plant. This is an investment of USD 80 million (approximately KRW 100 billion). The ADC manufacturing facility is currently being expanded and is targeting GMP approval in the first quarter of next year. The company is also in the process of building a related plant in Songdo, Incheon. In addition, Celltrion plans to establish a separate CDMO subsidiary while pursuing ADC drug development. Kyongbo Pharmaceutical is investing KRW 85.5 billion to build an ADC plant. The reason why domestic and foreign CDMOs are rushing to expand production capacity for ADC drugs is due to their marketability and high barriers to entry. ADC is a type of antibody conjugated with a cytotoxic drug (payload) as a linker. They have a high structural complexity compared to conventional antibody drugs, which makes the development and manufacturing process difficult, but they have emerged as the next generation of biopharmaceuticals due to their relatively high therapeutic efficacy and low side effects. Following the success of Daiichi Sankyo's breast cancer drug Enhertu (trastuzumab deruxtecan), research on ADC drugs has increased explosively worldwide. However, facilities for the development and mass manufacture of ADC drugs have not been able to keep pace. This is why an imbalance between ADC-related research and manufacturing is expected in the field. Unlike conventional antibody drug CDMOs, ADC-specific manufacturing facilities require more particular technologies. Unlike antibody drug production facilities, ADC production facilities must incorporate additional design principles because they handle cytotoxic drugs (payloads) and organic solvents. Additional design details include negative pressure design, differential pressure between cleanrooms, and airlocks to prevent the spread of cytotoxic drugs and protect operators.
- Company
- Imfinzi combo drug Imjudo can be prescribed at hospitals
- by Eo, Yun-Ho Nov 13, 2024 05:54am
- Immuno-oncology drug Imfinzi's combination partner Imjudo may now be prescribed in general hospitals in Korea. According to industry sources, AstraZeneca Korea's CTLA-4 inhibitor Imjudo (tremelimumab) has passed the drug committees (DCs) of tertiary hospitals in Korea including Seoul National University Hospital and Seoul Asan Medical Center. For now, however, Imjudo is a non-reimbursed drug. AstraZeneca submitted an application for the reimbursement of the PD-L1 inhibitor Imfinzi (durvalumab) and Imjudo combination for liver cancer in June and is currently awaiting a review by the Health and Insurance Review and Assessment Service’s Cancer Disease Review Committee. Imjudo was approved by the Ministry of Food and Drug Safety in combination with Imfinzi in June last year. The first target indication for the combination is liver cancer and can be prescribed as a first-line treatment for adult patients with advanced or unresectable hepatocellular carcinoma (HCC). The specific regimen is the STRIDE (Single Tremelimumab Regular Interval Durvalumab) regimen, which consists of a single dose of Impinj 1,500 mg plus 300 mg of Imfinzi, followed by an additional dose of Impinj at regular intervals every 4 weeks. At the recent European Society for Medical Oncology (ESMO) Congress 2024, the 5-year overall survival data from the Phase III HIMALAYA trial that demonstrated the efficacy of the Imfinzi and Imjudo combination in hepatocellular carcinoma was presented. In the HIMALAYA trial, patients with inoperable HCC were treated with STRIDE (single dose of Imjudo followed by Imfinzi maintenance therapy), Imfinzi monotherapy, and sorafenib monotherapy. When comparing the results of the Imfinzi and Imjudo combination with sorafenib combination therapy in patients with unresectable HCC, patients who received the STRIDE regimen had a 5-year overall survival (OS) rate of 19.6%, compared with the 9.4% for patients who received sorafenib. The median overall survival was 16.43 months and 13.77 months, respectively, showing a 24% lower risk of death in the Imfinzi-Imjudo combination arm. “ The Imfinzi-Imjudo combination therapy has significant advantages in that it has a much lower risk of bleeding than conventional therapies and does not worsen liver function," said Hong Jae Chon, Professor of Hemato-Oncology at CHA Bundang Medical Center. “In particular, the combination shows potential for longer survival than existing therapies."
- Company
- Treatment-refractory Dravet syndrome calls for new options
- by Whang, byung-woo Nov 13, 2024 05:54am
- Despite increased treatment options for the ultra-rare Dravet syndrome, there are still gaps in care that require attention. Even with the introduction of medical cannabis, cannabidiol, there are patients who do not respond to the drug, which is why improving access to new options should be considered. Dravet syndrome is a rare neurological disorder that begins with fever and convulsions within the first year of life, persists into adulthood, and leaves nearly all young patients moderately to severely disabled after each attack. Although it is known to be a rare disease with an estimated prevalence of 1 to 2 per 10,000 people worldwide, there is no officially investigated prevalence in Korea and was designated as an ultra-rare disease in 2022. Dravet syndrome is characterized by the onset of the first seizure, which is similar to a febrile convulsion that usually occurs with fever at 6 months. The biggest risk factor is “Sudden Unexpected Death in Epilepsy” (SUDEP). While the rate of sudden death in intractable epilepsy is 20-25%, in Dravet syndrome, up to 59% of all deaths are associated with SUDEP. The goal of treatment for Dravet syndrome is to control seizure frequency and non-seizure symptoms to reduce the patient's risk of sudden death and improve quality of life. Initial treatment includes anti-seizure medications and add-on treatments such as the anti-seizure medications stiripentol and cannabidiol are used to treat the “drug refractory” nature of Dravet syndrome. Cannabidiol is a medical cannabis preparation that was previously supplied without reimbursement through the Korea Orphan & Essential Drug Center for urgent use but then has been reimbursed since April 2021. Hoon-Chul Kang, professor of pediatric neurology at Severance Hospital, emphasized that the government's approval of medical cannabis has contributed to improving the treatment environment for Dravet syndrome. Kang said, “The government's decision was based on the desperate voices of parents and caregivers of children with Dravet syndrome, as well as objective data reported in the literature,” he explains. Limitations remain for drug-refractory Dravet syndrome...a fundamental solution is needed However, stiripentol and cannabidiol are only available through the Korea Orphan & Essential Drug Center, and the treatment process from applying for the drugs to meeting the criteria for reimbursement coverage is rather complicated. Hoon-Chul Kang, professor of pediatric neurology at Severance Hospital Hoon-Chul Kang, professor of pediatric neurology at Severance HospitalIn particular, there are still many patients with Dravet syndrome who are refractory to existing medications, leaving a blind spot in terms of seizure management. Unlike Korea, where treatment options are limited, options are increasing overseas with the emergence of new options. In the long run, experts agree that Korea also needs a fundamental treatment for seizures that reduces the quality of life for people with Dravet syndrome and their caregivers. If a new treatment option can significantly improve seizure control while also managing additional comorbidities and disabilities, it would substantially improve the treatment landscape. In addition, despite the limitations of being an ultra-rare disease, there are expectations that Dravet syndrome will benefit from the government's 'fast-track program for serious and rare diseases to reduce the burden of medical expenses’ plan. The industry predicts that the government's interest in pediatric rare and intractable diseases will continue for the time being, given the revised pediatric drug pharmacoeconomic evaluation exemption system last year and the selection of drugs for the first pilot project for the approval-evaluation-negotiation linkage system. A professor of pediatrics at a tertiary hospital said, "The government needs to make another timely decision to improve the treatment environment for Dravet syndrome, an extremely rare disease that is even more neglected than others."
- Company
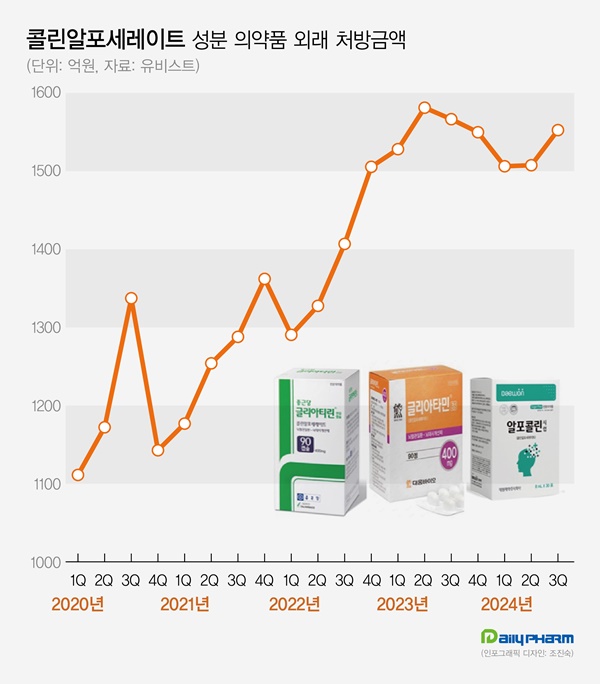
- 'Choline alfoscerate' prescription market continues to grow
- by Chon, Seung-Hyun Nov 13, 2024 05:54am
- The cognitive enhancer 'choline alfoscerate (choline products)' has expanded its presence in the prescription market. Choline products' growth slowed earlier this year but rebounded in Q3, further expanding the market size. Although a few companies withdrew from the market due to the risk of failing clinical re-evaluation, the prescription market continued sales boom. According to the pharmaceutical market research firm UBIST on November 11, the outpatient prescription market for choline products totaled KRW 155.3 billion in Q3. It decreased by 0.9% compared to Q3 of last year but increased by 3.0% compared to the previous quarter. Choline products' prescription sales recorded KRW 158.1 billion in Q2 of 2023. Then they went on a downward slide for three consecutive quarters until Q1 this year. In Q2, the sales slightly increased compared to the previous quarter. In Q3, they showed a strong rebound. Choline products' prescription size for Q3 was recorded as the third highest in history. The slowing growth of choline products in the first half of this year is likely due to steep growth in the past few years. The prescription market for choline products recorded KRW 308.8 billion in 2018. Then, it continued to renew the best record every year. Last year, the market amounted to KRW 622.6 billion, expanding more than twice in five years. Quarterly prescription sales indicate a 46.4% increase over five years from KRW 106.1 billion in Q3 of 2019. Outpatient prescription sales of pharmaceuticals containing choline alfoscerate (unit: KRW 100 million, source: UBIST). Even though choline products are under clinical re-evaluation for efficacy evaluation, clinical practices have continued to have a high demand for choline products. In June 2020, the Ministry of Food and Drug Safety (MFDS) requested companies with choline products to submit their clinical trial documents, and 57 pharmaceutical companies began clinical trials for reassessment. Previously, three indications for choline products had been approved, including ▲secondary symptoms caused by cerebrovascular deficit or degenerative organic brain syndrome ▲emotional and behavioral changes ▲senile pseudodementia. In the re-evaluation process, two out of three indications for choline products were deleted, excluding 'secondary symptoms caused by cerebrovascular deficit or degenerative organic brain syndrome.' Choline products are facing the possibility of reduced reimbursement in addition to the issue of their efficacy. In August 2022, the MOHW issued revised regulations on 'The Criteria and Scope of National Health Insurance,' indicating that patients without prior dementia diagnosis will have a copayment increased from 30% to 80%. After that, two groups of pharmaceutical companies, led by Daewoong Bio and Chong Kun Dang, filed an administrative suit to cancel the MOHW's notification. However, they all lost in the first trial in 2022. Chong Kun Dang also lost in the second trial in May. However, as the suspension of execution filed by pharmaceutical companies has been accepted, reimbursement reductions are on hold. Despite many products being withdrawn from the market after the clinical re-evaluation of choline products, the prescription market continued to grow. According to the MFDS, choline products that received Korean approval total 278 items. Among these, 134 items have been withdrawn from the market due to approval withdrawal or cancelation. Previously, the MFDS ordered the clinical re-evaluation of choline products from 134 companies. 77 companies withdrew from undergoing re-evaluation, resulting in a significant number of withdrawals from the market. More companies that commenced clinical re-evaluation of choline products are withdrawing. In two months from September, Guju Pharmaceutical, Kyongbo Pharmaceutical, PharmGen Science, YooYoung Pharmaceutical, and Medix Pharm voluntarily withdrew choline product approval. Increasing number of companies are exiting the market due to potential retrieval amounts that arise when they fail the re-evaluation for choline products. In 2020, the MOHW issued a national health insurance contract to companies with choline products, entailing 'companies failing clinical trials must return the prescription sales.' Within eight months of the negotiation order, pharmaceutical companies agreed to the term that they would return 20% of the prescription sales from the time they received IND approval to the date of deletion when the indication for the product was deleted due to failed clinical re-evaluation. The retrieval negotiations for choline products are determined by agreements between the MOHW and each pharmaceutical company, resulting in different contract details for each company. While a 20% retrieval rate from prescription sales is commonly applied, the timing of the retrieval rate varies among companies. Sources said that most companies have agreed to increase retrieval rate. For instance, companies may have agreed to set a 10% retrieval rate for this year when they fail the clinical re-evaluation of choline products, then increase to 30% after five years. As the prescription market for choline products continues to grow, companies that agreed to a gradual increase of retrieval rate would end up exponentially expanding retrieval amount due to the market growth. Pharmaceutical companies may have to face increased retrieval amounts as the market for choline products continues to grow if they fail clinical re-evaluation. For these reasons, sources said that more companies are considering exiting the market before completing the clinical re-evaluation. However, analysis suggests that the entire market for choline products continues to grow as other products replace the withdrawn products. Prescription sales by major products indicate that Daewoong Bio's Gliatamin recorded KRW 41.2 billion in Q3, a 4.4% reduction from the previous year. Chong Kun Dang's Chongkundang Gliatirin generated prescription sales of KRW 31.1 billion in Q3, up 10.9% from last year. Arlico's Choliatin recorded prescription sales of KRW 5.1 billion in Q3, a 28.7% reduction from the previous year. Daewon Pharmaceutical's Alfocholine generated prescription sales of KRW 4.9 billion in Q3, down 10.3%. Yuhan's Alfoatilin recorded KRW 3.7 billion, down 16.7% from the previous year. Dongkoo Bio's Glifos' sales increased from KRW 2.7 billion in Q3 of last year to KRW 3.7 billion in a year, up 33.9%. Mother's Pharm's Memoem recorded prescription sales at KRW 1.1 billion in Q3 of last year, then increased to KRW 3.3 billion in a year, an increase of over threefold.
- Company
- Asthma drug 'Monterizine' sales rise despite generic entries
- by Kim, Jin-Gu Nov 12, 2024 05:51am
- Hanmi's asthma treatment, 'Monterizine,' successfully expanded its prescription sales over 10% Year-over-Year (YoY), despite the release of generics. Generic drugs had been listed for reimbursement in October 2023. The analysis suggests that Monterizine's continued sales expansion is because generic prices have not been reduced and, it maintains a strong market presence with its broader scope of use. Prescription sales 14%↑despite generic releases… Monterizine Chewable Tab is the only available treatment for young children According to the pharmaceutical market research firm UBIST on November 8, Hanmi's Monterizine recorded prescription sales of KRW 4.3 billion in Q3, which is an increase of 14% over a year compared to KRW 3.7 billion in Q3 2023. Monterizine is a combination drug containing the asthma treatment, 'Montelukast,' and the third-generation antihistamine, 'Levocetrizine.' Hanmi received approval for 'Monterizine Cap' in May 2017. The following April, Hanmi changed the formulation and received approval for 'Monterizine Chewable Tab' designed to be taken by chewing. After the launch of Monterizine Chewable Tab, prescription sales quickly expanded for the Monterizine series. The combined prescription sales of Monterizine Cap and Monterizine Chewable Tab have increased by 8%, from KRW 7.9 billion in 2019 to KRW 8.6 billion in 2020. It recorded KRW 9.7 billion in 2021, surpassing KRW 10 billion when the sales amounted to KRW 12.4 billion in 2022. Last year, it recorded KRW 15.6 billion, a 21% increase from the previous year. This year, Monterizine is maintaining solid sales. In Q4 2023, it surpassed KRW 4 billion for the first time, generating KRW 4.3 billion to KRW 4.4 billion in sales each quarter. Quaterly prescription sales of the original drug Monterizine (blue) and generics (ligt blue) (unit: KRW 100 million, source: UBSIT). Interestingly, generics of Monterizine were launched in October 2023. Typically, sales of original drug slow down after the generic launches, but Monterizine is following a different trend. The analysis suggests that Monterizine Chewable Tab is showing a strong market presence with its broader scope of use. The scope of use for the original Monterizine Cap and generics to Monterizine are limited to 'adults and adolescents at the age of 15 or older.' Monterizine Chewable Tab is prescribed to 'young children at the age of 6-14.' The analysis is that Monterizine Chewable Tab is widely used in the prescription market, especially because young children require frequent prescriptions in the case of asthma drugs. Drug price has been maintained despite generic releases…the generics formulation is different from the original Additionally, one of the reasons for continued sales of Monterizine is that it remains the same price following the generics' releases. Product photo of HanmiIn September 2021, generics companies challenged four patent cases of Monterizine. In September of the following year, they successfully avoided patents one after another. After losing the first trial, Hanmi appealed to the patent court, but the company soon withdrew. Since October 2023, Monterizine generics have launched. Six products that met the requirement for the highest price by conducting bioequivalence tests became listed at KRW 886 per tablet. 14 products that did not meet the bioequivalence test because they were produced by contract manufacturing organization (CMO) became listed at KRW 753 per tablet. The drug price of Monterizine was not reduced following the generic release. The original drug price is automatically reduced by 30% when generics are released. Monterizine drug price should have been reduced from KRW 886 to KRW 620, but it was not. This is because the generics have been developed in a formulation different from the original. The Health Insurance Review and Assessment Service (HIRA) reduces the original drug price by 30% in the first year when a product with the same formulation is launched. In the following year, the HIRA further reduces the drug price of the original by 53.55%. In this case, the 'same formulation' means the active ingredient, administration route, dosage, administration method, formulation, and efficacy·effects are matched. The original Monterizine is available in capsules and chewable tablets. Generic products received approval as tablets. The government analyzed that the generics are not in the same formulation as the original Monterizine. Therefore, the drug price of the original was maintained at KRW 886.
- Company
- 'Vabysmo' for macular deg associated RVO indication expected
- by Eo, Yun-Ho Nov 12, 2024 05:51am
- Product photo of Vabysmo.The first bispecific antibody for eye diseases, 'Vabysmo,' is soon to be approved in South Korea for its indication of treating macular degeneration associated retinal vein occlusion (RVO). According to industry sources, Roche Korea's Vabysmo (faricimab) is being reviewed by the Ministry of Food and Drug Safety (MFDS) for its indication expansion. It is expected to be approved within the year. For RVO indication, it received the U.S. FDA approval in October 2023. Attention has been drawn to Vabysmo, a treatment for macular degeneration because it significantly extended the administration interval compared to Bayer's 'Eylea (aflibercept), which has been the standard therapy. In South Korea, Vabysmo's prescription became available after it was approved for reimbursement listing for neovascular age-related macular degeneration (nAMD) and diabetes-related macular edema (DME) in October last year. Existing macular degeneration drugs used in South Korea are vascular endothelial growth factor-A (VEGF-A) drugs such as Novartis' 'Lucentis (ranibizumab),' 'Beovu (brolucizumab),' and Eylea. Unlike existing VEGF drugs, like Lucentis and Eylea, Vabysmo can also block the angiopoietin-2 (Ang-2) pathway, thus inhibiting new blood vessel formation. The analysis suggests that blocking two independent pathways can more effectively stabilize blood vessels and reduce inflammation, abnormal vessel growth, and fluid leakage than the VEGF-A pathway alone. RVO is Vabysmo's third indication. Its efficacy has been confirmed through the Phase 3 BALATON and COMOINO studies. In these two clinical trials, Vabysmo achieved non-inferiority in the patient's vision improvement compared to Eylea. When treated with Eylea, the patients had continual vision improvements from the early stage. The safety profile of the trials was similar to previous study reports. Meanwhile, Vabysmo has seen an increase in sales this year. Its sales for Q1 amounted to 847 million francs, up 88.6% from the previous year. It recorded sales of 947 million francs (about 1.51 trillion won) in Q2, up 86.4% from last year. In Q2, Vabysmo surpassed the sales of the market leader Eylea (about 1.26 trillion won) for the first time.