- LOGIN
- MemberShip
- 2026-06-06 16:21:52
- Facing the era of low-priced generics…diabetes drug competition
- by Chon, Seung-Hyun | translator Hong, Ji Yeon | 2026-03-16 09:25:29
Dong-A ST is adding two more combination therapies that utilize its in-house-developed diabetes drug, Suganon. Having completed the development of these new combinations, the company has entered the regulatory approval stage. Once these two combination drugs are approved, six types of Suganon lineups will be secured.
Chong Kun Dang has also expanded its Duvie line, new diabetes drugs, to six products. While competing drugs are seeing stagnant growth due to overheated market competition from hundreds of generics, Chong Kun Dang has strengthened its competitiveness by securing new growth engines through the concentration of its research and development (R&D) capabilities. Analysis suggests that establishing new, incrementally modified drugs (IMDs) will be a powerful driving force for profitability in preparation for the upcoming era of low-priced generics.
Dong-A ST files for two Suganon combination drugs...establishing new growth engines amid slow growth in over-saturated market

According to the Financial Supervisory Service (FSS) on the 16th, Dong-A ST submitted applications for the marketing authorizations of “Sugaempa” and “Suganova SR” to the Ministry of Food and Drug Safety on the 13th. Both “Sugaempa” and “Suganova” are combination drugs made using Dong-A ST's diabetes drug, Suganon.
Suganon, approved in October 2015 as the 26th Korea-developed new drug, is a DPP-4 inhibitor diabetes treatment. Suganon has an outstanding blood glucose-lowering effect even at low doses due to its high selectivity for the DPP-4 enzyme. It has a low impact on the metabolism of other drugs, resulting in high medication convenience and compliance for patients with chronic diseases who must take multiple medications. Furthermore, it can be used without dose adjustment, even in patients with decreased renal function.
"Sugaempa" is a combination drug that joins Suganon with empagliflozin, an SGLT-2 class diabetes drug. The original drug for the empagliflozin component is Jardiance. "Suganova" is a triple combination drug consisting of Suganon, empagliflozin, and metformin.
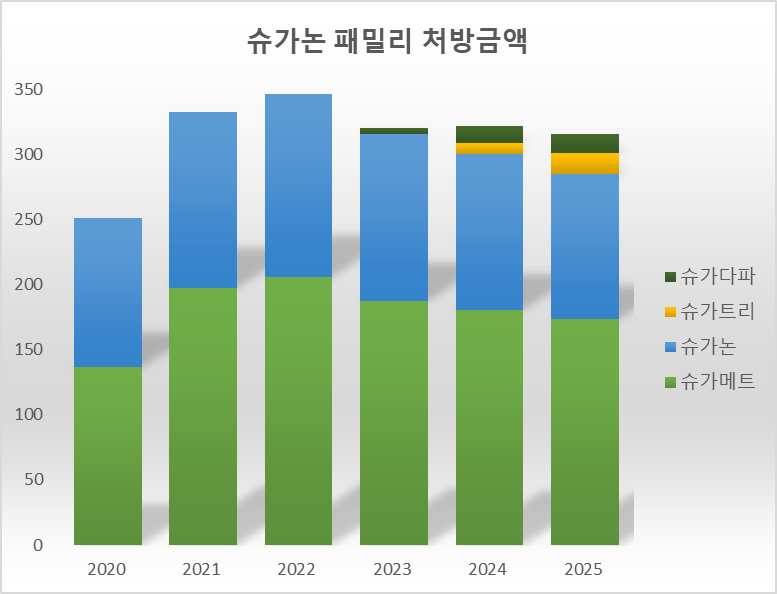
If Dong-A ST receives approval for "Sugaempa" and "Suganova," the Suganon family lineup will expand to six types.
In July 2016, Dong-A ST launched Sugamet, a combination containing Suganon and metformin. In May 2023, it released Sugadapa, combining Suganon with the SGLT-2 inhibitor dapagliflozin, and in January 2024, it added a triple combination drug comprising Suganon, dapagliflozin, and metformin.
By combining two different SGLT-2 inhibitor components, Dong-A ST will offer two types of Suganon + SGLT-2 inhibitor combinations and two types of Suganon + SGLT-2 inhibitor + metformin combinations. This strategy aims to equip the maximum number of combinations possible so that patients taking individual component drugs separately can utilize a Suganon combination drug.
The Suganon family is evaluated to have successfully settled into the prescription market, recording annual sales of approximately KRW 30 billion.
According to the pharmaceutical research organization UBIST, the four types in the Suganon family recorded KRW 31.5 billion last year. Sugamet and Suganon recorded prescription amounts of KRW 17.4 billion and KRW 11.1 billion, respectively. Sugatree and Sugadapa recorded KRW 1.6 billion and KRW 1.4 billion, respectively.
However, recent growth has been slow. Last year's prescription amount for Sugamet decreased by 3.5% compared to the previous year, and Suganon decreased by 7.7%. Sugamet recorded prescription sales of KRW 20.6 billion in 2022, but they have decreased by 15.6% over the last three years. Suganon has shown a downward trend for three consecutive years, following KRW 14.1 billion in 2022, a 20.9% decline. While Suganon and Sugamet combined for KRW 34.7 billion in 2022, the prescription amount for the four Suganon family products, including Sugatree and Sugadapa, decreased by 9.0% compared to three years ago.
With both the DPP-4 inhibitor and SGLT-2 inhibitor markets in Korea entering a state of oversaturation due to the entry of generics, it is a difficult environment to sustain growth. While most domestic pharmaceutical companies compete by offering generics, Dong-A ST's move is to strengthen its competitiveness in the oversaturated market by investing in its self-developed new drugs and additional R&D capabilities. 174 subjects participated in the clinical trials for Dong-A ST's "Sugaempa" and "Suganova."
Chong Kun Dang establishes 6 Duvie lineup... Securing profitability weapon against low-priced generic era
Chong Kun Dang is also strengthening its competitiveness in a stagnant market by steadily adding to its self-developed diabetes drug Duvie lineup.
On the 11th, Chong Kun Dang received marketing authorization approval from the Ministry of Food and Drug Safety for Duviempol XR. Duviempol XR is a triple combination drug comprising lobeglitazone, empagliflozin, and metformin. Lobeglitazone is the main component of Duvie, the diabetes drug independently developed by Chong Kun Dang.
Chong Kun Dang stated, "We expect to increase administration convenience by providing a new treatment therapy with the fixed-dose combination of lobeglitazone, empagliflozin, and metformin for type 2 diabetes patients whose blood sugar is not appropriately controlled by the dual therapy of empagliflozin and metformin."
Duviempol XR is the sixth lineup developed based on Duvie. Approved in 2013 as the 20th Korea-developed new drug, Duvie is a thiazolidinedione (TZD) class diabetes treatment.
Starting with Duvie, Chong Kun Dang currently sells four Duvie lineups: Duviemet SR, Duviemet S, and Duet S. Duviemet SR, approved in 2016, is a combination drug combining Duvie and metformin.
Duviemet S, approved in May 2023, is a combination drug containing Duviemet and the DPP-4 inhibitor sitagliptin. The original drug for sitagliptin is Januvia. In June 2023, Chong Kun Dang received additional approval for Duet S, a dual combination drug joining lobeglitazone and sitagliptin.
In January, it equipped its fifth lineup by receiving approval for Duviempa, which joins Duvie and empagliflozin. Duviempa can be used for adult type 2 diabetes patients for whom the concomitant administration of lobeglitazone and empagliflozin is appropriate.
For Chong Kun Dang, the strategy is to maximize synergy in the prescription market by presenting new treatment alternatives to medical staff and patients through the introduction of various combination drugs centered on Duvie.
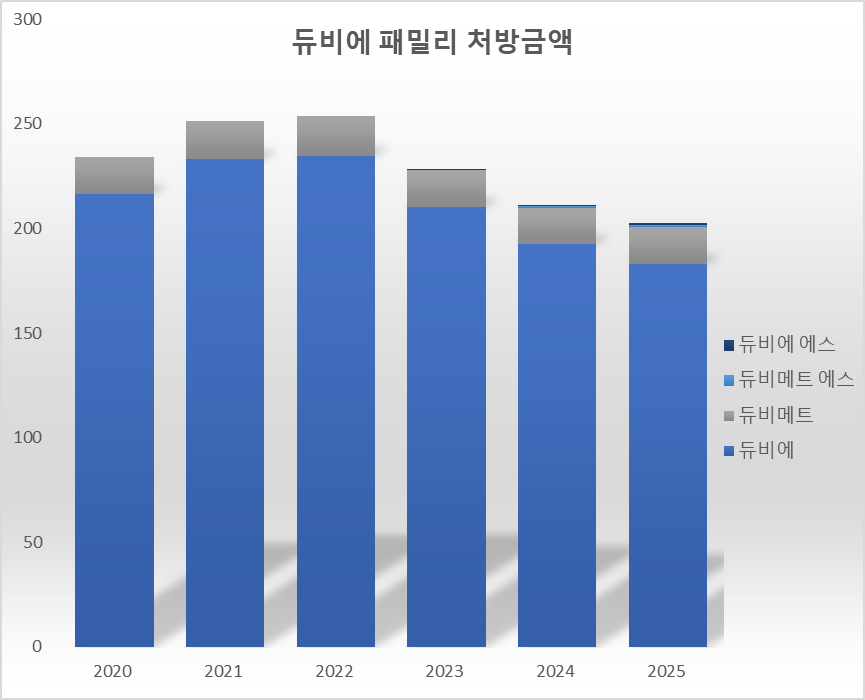
The market situation for Duvie is also not easy. Last year, outpatient prescription costs for the four types in the Duvie family totaled KRW 20.3 billion, a 3.9% decrease from the previous year. Duvie's prescription volume was KRW 18.3 billion, accounting for about 90%, while the remaining products were not significantly prominent. In 2022, Duvie and Duviemet combined for KRW 25.4 billion. Last year, the combined prescription amount of the four products decreased by 20.2% compared to three years ago.
As with Suganon, it is a difficult environment for Duvie to sustain growth as pharmaceutical companies indiscriminately release generics for diabetes drugs like SGLT-2 inhibitors. However, the company is moving to equip itself with additional growth engines by steadily releasing combination drugs developed through its R&D capabilities.
It is viewed that combination drugs developed by a pharmaceutical company's proprietary new drugs can become a driving force for future profitability, especially as the government continues its attempts to lower generic drug prices.
In November last year, the Ministry of Health and Welfare (MOHW) reported to the Health Insurance Policy Deliberation Committee a plan to improve the drug pricing system, which includes lowering the price calculation rate for generics and off-patent drugs from 53.55% to the 40% range. It is reported that on the 11th, the MOHW held a sub-committee of the Health Insurance Policy Deliberation Committee and suggested a generic drug price calculation rate in the low-to-mid 40% range.
If the generic drug price standard is lowered from 53.55% to 43%, the calculation shows the maximum generic price will be reduced by 19.7%.
In the reorganization plan reported by the MOHW last November, it was specified that, while maintaining the maximum price requirements applied since 2020, the reduction rate for unmet requirements would be expanded from 15% to 20%. This means that the prices of generics that do not meet the maximum price requirements will drop even further.
Under the drug pricing system reform since July 2020, a generic product can receive the maximum price only if it meets both requirements: performing a bioequivalence study and using registered drug master file ingredients. Every time one requirement is not met, the upper limit price drops by 15%. If both requirements are not met, the structure results in a 27.75% reduction. Applying a 15% reduction rate, the generic maximum price calculation standard of 53.55% drops to 45.52% if one requirement is unmet, and to 38.69% if two requirements are unmet.
If the generic calculation standard is set at 43%, a generic that fails to meet one maximum price requirement will be lowered to 34.40%, and a generic that fails to meet two requirements will be lowered to 27.52%. In this case, the price of a generic failing to meet one requirement is reduced by 24.4% compared to the current level, and the reduction rate for failing two requirements is 28.9%. It is mathematically possible to calculate that the reduction in generic drug prices will approach 30%.
This is the background for which pharmaceutical companies are complaining that performance pressure could intensify due to the generic drug price cuts. An industry official stated, "Due to the government's policy on lowering drug prices, the market may find it difficult to expect profits from generics in the future. Equipping new drugs or IMDs that can receive high drug prices will greatly help future performance strength."
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.








