- LOGIN
- MemberShip
- 2026-06-06 12:33:14
- MOHW agrees to relax convenience store medicine policy
- by Lee, Jeong-Hwan | translator Hong, Ji Yeon | 2026-03-11 08:28:12
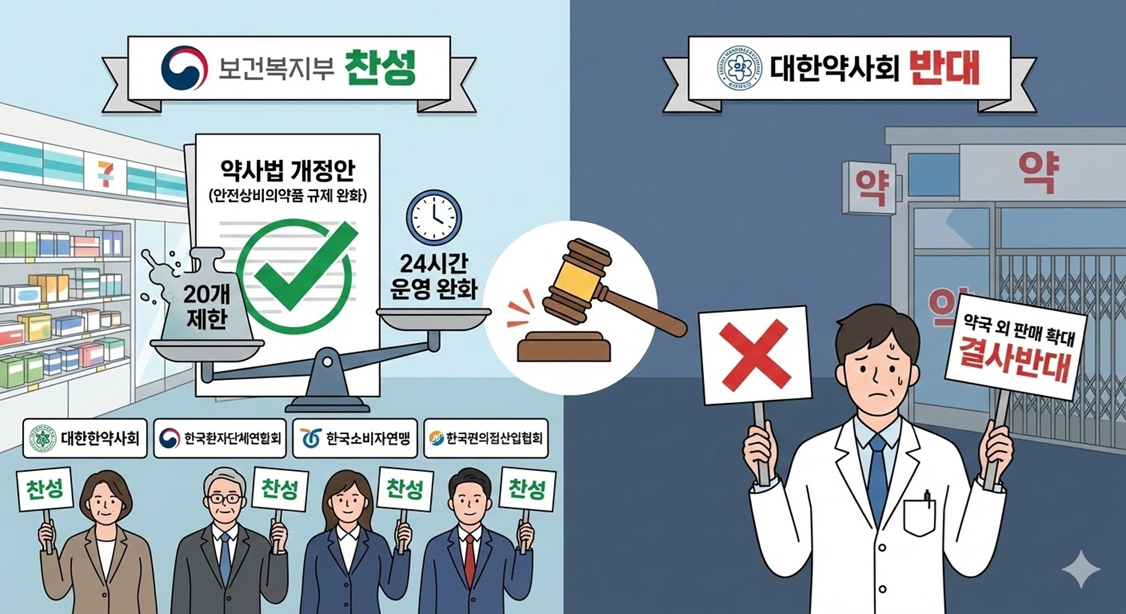
The Ministry of Health and Welfare (MOHW) has cast a vote in favor of an amendment to the Pharmaceutical Affairs Act that legislates the '20-item limit' on the number of safe household medicine items, currently prescribed by the Pharmaceutical Affairs Act, to a Presidential Decree (Enforcement Decree) to allow for lower-level legislation.
The Ministry also expressed support for a provision that eases the '24-hour operation' requirement, which is a mandatory registration criterion for sales outlets, in eup, myeon, and dong where there are no pharmacies or safe household medicine sales outlets.
In addition, the Ministry presented a specific proposal to delay enforcement by one year from implementation date to prepare for various matters related to institutional improvement, such as the establishment of subordinate statutes.
The bill currently under discussion is facing significant backlash from the pharmacist community, as it could lead to the complete removal of regulations on the number of safe household medicine items in convenience stores and to the expansion of the sale of general medicines outside pharmacies. However, with the competent MOHW expressing support, the likelihood of its passage has increased.
In addition to the MOHW, the Korean Society of Oriental Pharmacy, the Korea Alliance of Patients Organizations, the Consumers Union of Korea, and the Korea Association of Convenience Store Industry also supported the legislation. The Korean Pharmaceutical Association was the only organization to oppose the bill.
On the 10th, the amendment to the Pharmaceutical Affairs Act, proposed by Representative Han Ji-ah of the People Power Party, is planned to be tabled at the plenary session of the National Assembly's Health and Welfare Committee.
Rep. Han Ji-ah's stands that fixing the number of safe household medicine items in the Pharmaceutical Affairs Act is hindering the ability to respond flexibly and administratively to changes in the pharmaceutical market and environment, as well as to public demand.
This is the reason for introducing a bill that provides a delegation provision, allowing the number of safe household medicine items to be determined by Presidential Decree.
Rep. Han's proposal also includes a provision to establish the basis for the installation and operation of a Pharmaceutical Policy Deliberation Committee under the Minister of Health and Welfare.
Easing the 20-item upper limit on safe household medicine items... Ministry supports, Pharmaceutical Association opposes
The Ministry supported the provision delegating the regulation of the 20-item limit for convenience store medicines, which is currently fixed in the Pharmaceutical Affairs Act, to a Presidential Decree.
The MOHW also submitted a supportive opinion on relaxing the 24-hour operation requirement and the registration criterion for sales outlets on a limited basis in eup, myeon, and dong areas where there are no pharmacies or safe household medicine sales outlets, to improve accessibility to safe household medicines.
However, the MOHW stated that the enforcement date should be adjusted to 1 year after promulgation to prepare for various matters related to institutional improvement, such as the establishment of subordinate statutes.
The Korean Society of Oriental Pharmacy also agreed with the amendment to reorganize the safe household medicine system.
The Korea Alliance of Patients Organizations and the Consumers Union also supported it. The Consumers Union also expressed the view that it is appropriate to determine the regulation on the number of items through an Enforcement Rule, an Ordinance of the Ministry of Health and Welfare, rather than a Presidential Decree.
The Korea Association of Convenience Store Industry supported the bill while suggesting a modification that would require the MOHW to designate the number of household medicine items after receiving advice from a committee.
The Korean Pharmaceutical Association strongly opposed. They pointed out that although various systems to supplement medically underserved areas, such as public late-night pharmacies, health clinics, and the designation of special locations, are already in place, the management and operation of safe household medicine sales outlets and special locations remain insufficient.
The Korean Pharmaceutical Association argues that, rather than indiscriminately expanding the convenience store medicine system, the priority should be to evaluate whether the management systems of existing programs are functioning properly and to reorganize the system to ensure safe use.
The Korean Pharmaceutical Association stated, "To resolve the pharmacy accessibility of residents in medically underserved areas such as farming and fishing villages, special locations can already be designated according to the Pharmaceutical Affairs Act, and a system is in place to allow the purchase of medicines through designated special locations. Currently, medicine is also accessible through 1,895 health clinics nationwide," and emphasized, "In particular, considering the increase in cases of acetaminophen poisoning, the lack of management of sales outlets and the increase in cases of non-compliance with requirements, and the trend of strengthening regulations abroad, we actively oppose the expansion of safe household medicine items at this point as it is a policy that directly contradicts national health."
Establishment of Pharmaceutical Policy Deliberation Committee...Ministry of Health and Welfare, Ministry of the Interior and Safety, etc., all opposed
The MOHW and the Ministry of the Interior and Safety opposed the provision establishing a Pharmaceutical Policy Deliberation Committee, arguing that it would create an environment in which pharmaceutical-related matters, such as medicines, could be discussed regularly.
The logic is that, since the Central Pharmaceutical Affairs Council exists and is established, installed, and operated by the MFDS, the establishment of an additional body is unnecessary.
The Ministry of the Interior and Safety stated, "The Pharmaceutical Policy Deliberation Committee is an advisory committee under the MOHW, but considering the purpose of the Act on the Establishment and Operation of Committees Under Administrative Agencies, it is necessary to consider plans to expand the functions of the Central Pharmaceutical Affairs Council or utilize the policy advisory committees within the MOHW rather than establishing a separate committee."
The Korean Pharmaceutical Association and the Korean Medical Association also submitted opposing views.
The Korean Pharmaceutical Association opposed it, stating, "The role of the Pharmaceutical Policy Deliberation Committee in the bill is managed by the Pharmaceutical Policy Division of the MOHW," adding, "The policy planning and drafting are performed by the Ministry's Division of Pharmaceutical Policy, while the Pharmaceutical Policy Deliberation Committee performs policy deliberation and advice. In practice, this would lead to discussing the same matter twice, causing inefficiency and making the responsibility and subject of policy decisions unclear."
The Korea Medical Association said, "Since the Central Pharmaceutical Affairs Council (CPAC) is already running, establishing a separate Pharmaceutical Policy Deliberation Committee would be a redundant installation of a committee performing similar functions, which is a waste of administrative power," adding, "There are concerns that it could lead to role conflicts between committees and inefficiency in the policy-making process."
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.








