- LOGIN
- MemberShip
- 2026-05-02 20:06:06
- Company
- 98% relapse-free Soliris is reimbursed for NMOSD in Korea
- by Son, Hyung-Min May 10, 2024 05:47am
- Ho Jin Kim, Head of Department of Neurology & Division of Clinical Research, National Cancer Center A new treatment option has been introduced for patients with neuromyelitis optica spectrum disorder (NMOSD), a disease that has over a 90% relapse rate. Soliris became fully reimbursable for NMOSD last month, recording a 98% relapse-free rate. However, there are concerns that the stringent reimbursement requirements for Soliris may reduce patient access to the treatment. On May 7, AstraZeneca Korea held a press conference at the Conrad Hotel in Seoul to celebrate the reimbursement of Soliris for NMOSD in Korea. Soliris is a C5 complement inhibitor developed by AstraZeneca. It binds to the C5 protein and prevents the activation of a complement terminal complex, which has been shown to be effective for several autoimmune diseases. Soliris is currently approved for paroxysmal nocturnal hemoglobinuria (PNH), atypical hemolytic uremic syndrome (aHUS), and neuromyelitis optica spectrum disorder (NMOSD). In Korea, its approval for the NMOSD indication was extended in 2021. AstraZeneca Korea succeeded in securing reimbursement for the indication in Korea approximately 3 years after expanding its indication. NMOSD is a rare autoimmune disease characterized by unpredictable, recurrent episodes of optic neuritis and myelitis. It can cause severe disability, including optic neuritis resulting in blindness and paraplegia. 7 out of 10 NMOSD patients are positive for anti-quantiporin-4 (AQP-4) antibodies, which can activate the complement cascade and cause necrosis of the optic nerve and spinal cord. 90% of the patients suffer relapses, and each relapse is characterized by worsening symptoms, increasing the likelihood of blindness, paralysis, and premature death. Soliris has gained attention for showing effects in patients with relapsing NMOSD. In the PREVENT study, at 48 weeks of treatment, 98% of patients in the Soliris treatment arm were relapse-free, compared to 63% in the placebo arm. These relapse prevention benefits were sustained over the 144-week treatment period. The PREVENT extension study confirmed Soliris’s relapse-free rate of 94.4% 197 weeks after treatment. The study also confirmed the drug’s long-term sustained relapse prevention effect. However, some have expressed concerns on how the reimbursement requirements may limit patient access. The drug is currently reimbursed for adult patients with NMOSD who are 18 years of age or older who are AQP-4 antibody-positive and have an Expanded Disability Status Scale (EDSS) scores of 7 or below at the time of Soliris administration and at least 2 symptomatic relapses within the past year or at least 3 symptomatic relapses within the past 2 years (including one within the past year). The issue is that patients have to have multiple relapses to qualify for Soliris’ use. Specifically, patients who are receiving rituximab, an existing treatment for NMOSD, off-label, are ineligible for Soliris’ reimbursement. Patients would have to experience worsening of their disease to receive reimbursement. Enspryng, which was approved for NMOSD prior to Soliris, also has strict reimbursement requirements that prevent many patients from receiving its benefits. Professor Ho-Jin Kim from the Department of Neurology at the National Cancer Center said, “Even a single relapse of NMOSD can cause permanent disability. Active relapse prevention treatment is essential in the acute phase to prevent permanent disability in patients.” He added, “New high-priced treatments that are highly effective in preventing relapses have emerged, but the current reimbursement conditions limit their use in Korea. The expensive drugs should be used when they are most beneficial, rather than in a predetermined order. There is still a lot of work to be done to ensure the most efficient use of these good drugs for our patients." Chul Woong Kim, Director of the Rare Disease Business Unit at AstraZeneca, said, “The reimbursement of Soliris will allow patients with NMOSD to receive treatment for rapid relapse prevention. We will work to improve patient access so that more patients can enjoy Soliris' benefits."
- Company
- Korean AI-driven drug discovery speeds up
- by Nho, Byung Chul May 10, 2024 05:46am
- Ministry of Health and Welfare (MOHW) and the Ministry of Science and ICT will allocate KRW 34.8 billion in AI-driven new drug discovery. AI-driven new drug discovery through data collaboration among the biopharmaceutical industry, medical institutions, research institutions, universities, and public institutions has accelerated and gained significant attention. Previously, individual biopharmaceutical companies in South Korea conducted AI-driven new drug discovery through their in-house protocol, and the process was kept confidential. Starting with the recently organized business group called K-MELLODDY (Machine Learning Orchestration for Drug Discovery), a new system is expected to be implemented within the first half of the year to build upon a system to utilize sources and information related to various clinical and substance owned by 20 institutions. An individual company’s AI-driven new drug discovery could predict a new drug candidate’s ADMET (Absorption, Distribution, Metabolism, Excretion, Toxicity) value via in-vitro studies. However, these test results were insufficient to ensure the drug would pass in-vivo (pre-clinical) or clinical trials. Furthermore, ADMET prediction for clinical trial outcomes requires learning from clinical trial (in-human) data, but not enough clinical or in vivo data makes implementation extremely difficult. Furthermore, beyond predicting ADMET for a single drug molecule, solutions were needed that could expand to include interactions with specific targets, drug-drug interactions, responses based on user types, and various toxicity predictions. Therefore, optimized models based on federated learning have been considered for AI-driven drug development. K-MELLODDY aims to utilize new drug development data without leaking internal data to the outside world. In other words, federated learning and blockchain, which securely maintain companies' intellectual property data while leveraging AI, are evaluated as the core technologies of this project. The project’s characteristics and advantage are differentiated by 'Obtaining reliability of AI-driven new drug discovery,' 'High expandability of platform,' 'Securing ownership of data utilizing technology,' and 'Reducing new drug development cost.' Upon completion of the platform, utilizing AI predictions for the candidate product's metabolism and toxicity experiment results could reduce the number of experiments by half, leading to more than 50% cost savings. “K-MELLODDY project aims to build a federated learning-based AI drug development platform by utilizing the power of data held by Korea's academia, industry, and research institutes related to drug development. It aims to derive successful AI case studies in the drug discovery stage,” Hong Sungeun, Senior Researcher at the Convergence AI Institute for Drug Discovery (CAIID), stated. He added, “A budget of KRW 34.8 billion will be allocated over the next five years to complete the development of a Korean federated learning-based AI new drug discovery model.” Meanwhile, K-MELLODDY is described as a Korean AI new drug platform project that has been improved to suit the needs of the Korean situation while benchmarking the EU-MELLODDY project. EU-MELLODDY is an initiative for data collaboration among 10 pharmaceutical companies, first implemented in the EU. The initial business goal was to prove that data-based cooperation is possible between competing pharmaceutical companies and spread technological safety. Based on the results of EU-MELLODDY, which was conducted in 2019 for three years with KRW 25.6 billion put into, it was observed that the machine learning-based cooperative ADME/Tox prediction model was learned without leaking pharmaceutical companies’ confidential information. In addition, it outperformed the single model formulated by individual institutions without data leakage.
- Company
- 'Lixiana' dominates the DOAC market in KOR
- by Kim, Jin-Gu May 09, 2024 05:53am
- (clockwise from top-left) Lixiana, Eliquis, Pradaxa, and Xarelto. Daiichi Sankyo Korea’s 'Lixiana (edoxaban)' continues to dominate the market for Direct Oral Anti-Coagulant (DOAC). BMS’s 'Eliquis (apixaban)' sales have recently experienced a slowdown, and Bayer’s 'Xarelto (rivaroxaban)' prescription performance significantly dropped after the generic launch. The sales of Xarelto generic nearly chased the original product three years after its launch. The pharmaceutical industry eyes on the substance expiry of Eliquis this September. Eliquis generics that withdrew from the market after the Supreme Court’s ruling last 2021 may re-enter the market around the time of patent expiry, and a significant shift in the market is expected. Lixiana, 9% up in one year… Eliquis growth has been stalled According to medical market research firm UBIST, the market of DOAC treatment outpatient prescriptions in Q1 in South Korea amounted to KRW 62.9 billion, up 6% over a year compared to KRW 59.3 billion in Q1 last year. The mechanism of action of DOAC treatment is based on directly affecting the blood coagulation factor, thereby preventing blood clots. It is expanding its usage as it replaces warfarin, which prevents vitamin K metabolism. In South Korea, Xarelto was approved in 2009, followed by Pradaxa and Eliquis in 2011. Pradaxa and Eliquis were also approved in 2015. When the product was launched, it was called New Oral Anti-Coagulant (NOAC). Ten years after its launch, the term was changed to Direct Oral Anti-Coagulant (DOAC) to indicate that the product directly affects the coagulation factor. Four original products have seen a shift in the market. The top-selling product, Lixiana, is strengthening its position as the market leader. Prescription sales of Lixiana in Q1 of last year were KRW 27.7 billion, up 5% over a year compared to KRW 25.5 billion in Q1 of last year. Although it was launched late, its prescription performance grew fast, reaching the top after 2019. Last year, Lixiana became the first DOAC product to reach annual prescription sales of KRW 100 billion. Quarterly DOAC prescription performance (unit: KRW 100 million, source: UBIST). The second-ranked Eliquis has seen a stall in the growth recently. Eliquis prescription sales have seen a 2% growth, increasing from KRW 19 billion in Q1 of last year to KRW 19.3 billion in Q1 of this year. After Q3 of 2022, Eliquis prescription sales remain around KRW 19 billion. As Lixiana sales grew fast while Eliquis sales stalled, the sales gap between the two products is widening. The difference in the prescription sales between these two products in Q1 of this year amounted to KRW 8.5 billion. It continues to widen every year, with a difference of KRW 4.4 billion in Q1 of 2022 and KRW 6.5 billion in Q1 of 2023. Low Xarelto generics sales… Riroxban>Riroxia>Rivoxaban Over the last two years, Xarelto has experienced a significant decrease in prescription sales among the major DOAC products. Xarelto’s prescription sales in Q1 this year amounted to KRW 7.6 billion, down 5% compared to KRW 8 billion in Q1 last year. Its prescription sales declined by almost half over the past two years compared to KRW 14.8 billion in Q1 of 2022. Xarelto’s prescription sales decline is likely due to its patent expiration and generic release that followed. Xarelto generic was launched first in Q2 of 2021. Xarelto’s prescription sales decline is likely due to its patent expiration and generic release that followed. Xarelto generic was launched first in Q2 of 2021. In Q3 of 2022, the stoppage of reducing drug prices for Xarelto due to an administrative suit was lifted. When the pricing of Xarelto decreased, prescription sales started to decline. Eventually, the presence of Xarelto generics increased while the sales of the original prescription gradually decreased. Xarelto generics have gradually increased their prescription sales, nearly catching up with those of the original products. As of Q1, the net prescription sales of generic amounted to KRW 5.9 billion, up 61% over a year compared to KRW 3.7 billion in Q1 of last year. According to Q1 prescription performance report, Hanmi Pharm’s 'Riroxban' generated KRW 1.6 billion, CKD Pharm’s 'Riroxia' generated KRW 1.2 billion, and Samjin Pharmaceutical’s 'Rivoxaban' generated KRW 1.2 billion. Trends in prescription performance of rivaroxaban-containing original product and major generics (unit: KRW 100 million, source: UBIST). Pradaxa, another original DOAC product, is also facing a significant sales slowdown. In Q1, Pradaxa prescription sales amounted to KRW 2.4 billion, down 25% year over year. Eliquis’s substance patent set to expire this September…generic re-entry predicted The future variable in this market is the expiration of Eliquis' patent. Eliquis' substance patent expires on September 9th of this year. Generic manufacturers are waiting for the expiry of the substance patent, with Eliquis' formulation patent set to expire in 2031 after being invalidated. Generic companies won both the first and second trials related to substance patents in 2018 and 2019, respectively. After June 2019, Eliquis generic products were subsequently launched and have since been steadily growing in prescription sales. However, the circumstances changed when the Supreme Court ruled on the reversal of the original decision in April 2021. Generic companies have retrieved their products that could potentially infringe on the patent. On the contrary, Eliquis successfully regained its dominant position in the market for DOAC treatments containing the apixaban ingredient. With the substance patent set to expire this September, a flood of Eliquis generics is possible. Major generic manufacturers have experience releasing their products and are expected to expand their influence in the market quickly. On the other hand, it is expected that Eliquis will experience a sharp decline in prescription performance following the release of generics. From BMS's perspective, there is no justification for delaying the government's price reduction decision following the generic reimbursement listing. Due to this, the pharmaceutical industry anticipates that Xarelto will further strengthen its dominant position in the DOAC market.
- Company
- Novel targeted therapy for gastric cancer 'Vyloy' may launch
- by Eo, Yun-Ho May 09, 2024 05:52am
- 'Vyloy,' a targeted therapy for gastric cancer with a novel mechanism of action, is expected to be launched in South Korea. According to industry sources, Astellas Korea has submitted an application for approval of Vyloy (zolbetuximab), a treatment for claudin 18.2 (CLDN18.2) positive, HER2-negative, unresectable, advanced, or recurrent gastric cancer. In March, it acquired the final approval in Japan. Vyloy is a monoclonal antibody targeting CLDN18.2, which is expressed in the stomach. Its mechanism of action involves binding to CLDN18.2, a protein expressed on the cancer cell membrane of gastric epithelial cells. The efficacy of Vyloy was proven through the Phase 3 SPOTLIGHT and GLOW clinical trials. The SPOTLIGHT trial, which enrolled 557 adult patients with metastatic gastric or gastroesophageal junction (GEJ) adenocarcinoma, compared the efficacy and safety of zolbetuximab plus mFOLFOX6 combination therapy (fluorouracil, leucovorin, and oxaliplatin) to placebo plus mFOLFOX6. In the clinical trial, zolbetuximab combination therapy recorded progression-free survival (PFS), the primary endpoint, of 10.6 months, 8.7 months longer than the comparator. Additional clinical results showed that zolbetuximab combination therapy had an overall survival (OS) of 18.2 months, which was improved by 2.7 months compared to the comparator. In the GLOW study, zolbetuximab combination therapy improved PFS and OS. The study compared zolbetuximab plus CAPOX therapy (capecitabine and oxaliplatin) to placebo plus CAPOX therapy. The clinical results demonstrated that zolbetuximab combination therapy had a PFS of 8.2 months, 1.4 months longer than the comparator. The OS of zolbetuximab combination therapy was 14.4 months, compared to 12.2 months for the comparator. The common side effects related to safety were nausea and vomiting. Meanwhile, Vyloy is in progress for the discussion with the U.S. FDA, Europe, and China approvals. In the United States, its approval was rejected once over manufacturing issues.
- Company
- Novo Nordisk and Lilly post strong performance
- by Son, Hyung-Min May 08, 2024 05:48am
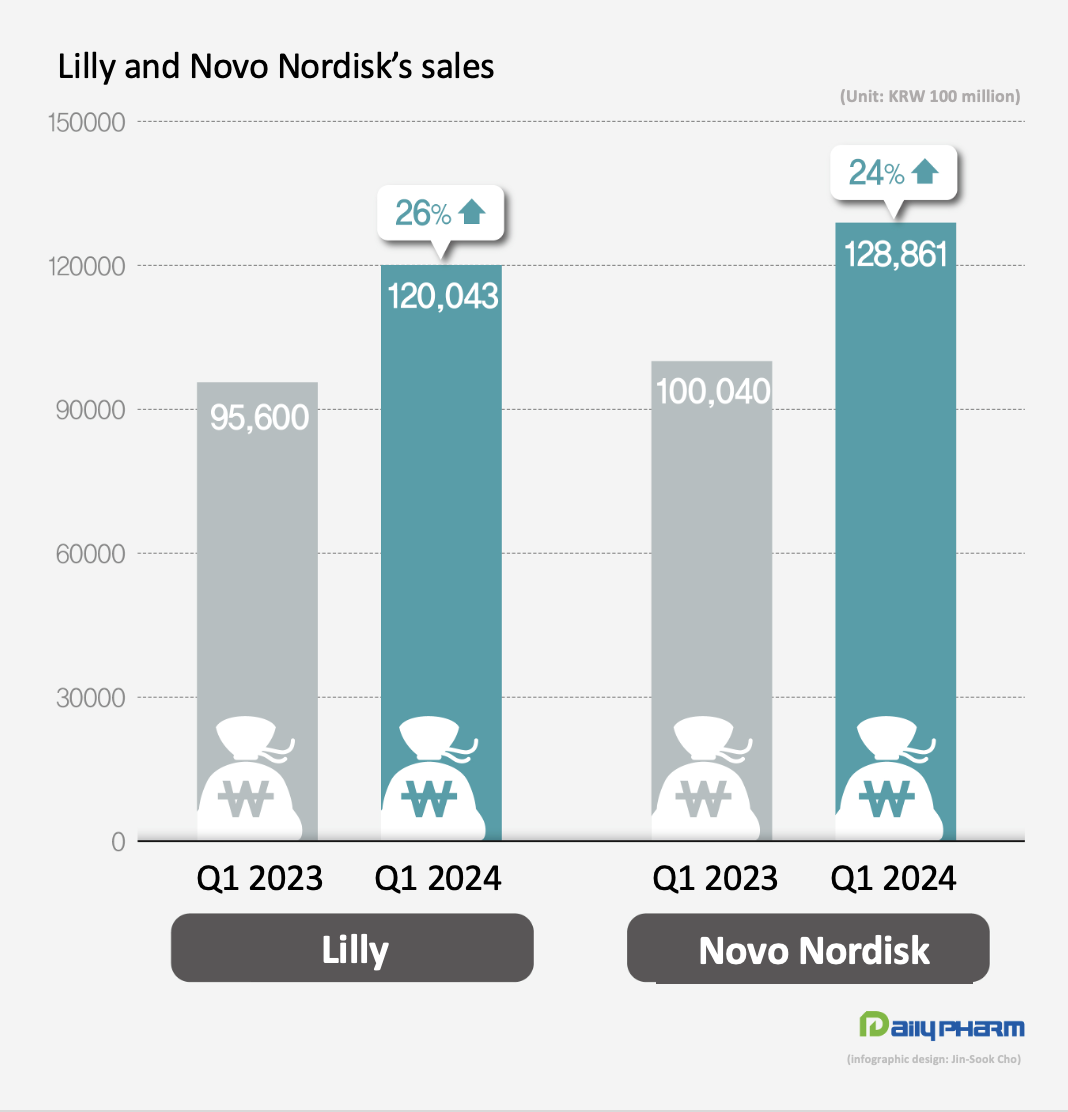
- The performance of Novo Nordisk and Eli Lilly, which released blockbuster diabetes and obesity drugs, have improved greatly in Q1 this year. Novo Nordisk's GLP-1 class diabetes drugs together generated KRW 10 trillion in quarterly sales. Lilly's obesity drug Zepbound posted sales of KRW 700 billion in Q1 this year, reaching nearly 1 trillion won in only half a year since its launch. According to KoreaBio data on the 3rd, Novo Nordisk's first-quarter sales rose 24% YoY to KRW 12.886 trillion (DKK 65.349 billion). The exchange rate was based on the basic rate of exchange as of May 2. Novo Nordisk's diabetes medicines generated sales of KRW 9.8456 trillion, up 24% YoY. In particular, sales of GLP-1 diabetes medicines surged 32% YoY to KRW 6.8981 trillion. Novo Nordisk's GLP-1 class diabetes drugs include Ozempic, Rybelsus, and Victoza, which are semaglutide and liraglutide drugs. Sales of obesity treatments totaled at KRW 2.1759 trillion, up 42% from Q1 2022. Specifically, Wegovy’s sales rose 107% to KRW 1.849 trillion. As of February 2024, the global market share of Novo Nordisk's diabetes treatments was 34%, including 35% in North America, 30% in Europe and 32% in China. Its global share of obesity medicines amounts to 85%. Lilly sees strong sales of diabetes and obesity drugs Lilly also reported first-quarter sales of KRW 12.43 trillion (USD 8.77 billion), up 26% YoY, driven by new diabetes and obesity drugs. The increase was driven by the company’s new products. Sales of its new products launched in 2022 and thereafter jumped 495%, bringing in KRW 3.27 trillion in Q1, up from KRW 550 billion in Q1 last year. Mounjaro’s revenue increased 218% from KRW 780 billion in Q1 last year to KRW 2.48 trillion in Q1 this year, recording a revenue of KRW 2.08 trillion in the U.S. alone. Mounjaro is a type 2 diabetes treatment developed by Eli Lilly that has a dual effect on glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). By acting on GLP-1 and GIP, Mounjaro provides further blood glucose control. Mounjaro has also been shown to have dramatic weight loss benefits. Last year, Lilly also launched Zepbound, an obesity drug that contains the same substance as Mounjaro, after confirming its weight-loss benefits in clinical trials. The drug has become a global blockbuster only half a year after its launch, posting sales of KRW 71 billion in Q1. Demand for GLP-1 class drugs surge...company will address supply issue Novo Nordisk and Lilly are working to ensure smooth production of their GLP-1s, which are experiencing supply shortages. Currently, GLP-1 medicines are experiencing global stock-outs due to a surge in demand. Novo Nordisk plans to accelerate production by acquiring Catalent’s plant from Novo Holdings. Since Novo Holdings acquired Catalent last year, it has handed over 3 plants to its subsidiary, Novo Nordisk. Lilly has recently signed contract development and manufacturing organizations (CDMOs) with National Resilience in the U.S. and BSP Pharmaceuticals in Italy to increase its production capacity. To further increase its production facility in the mid-to long-term, Lilly recently acquired Nexus Pharmacutical's manufacturing facility in the U.S. and broke ground on a parenteral (injectable) manufacturing plant in Germany, both of which are expected to be operational by the end of 2025 and 2027, respectively.
- Company
- Prescriptions for cephalosporin antibiotics double in 3yrs
- by Chon, Seung-Hyun May 08, 2024 05:48am
- The popularity of antibiotics in the outpatient prescription market continues. The prescription market for cephalosporin-class antibiotics has nearly doubled in the past 3 years. This ‘heyday’ in the cephalosporin prescription market is continuing, fueled by the steady increase in flu and cold cases since the end of the pandemic. According to the market research institution UBIST, the outpatient prescription market for cephalosporin antibiotics was KRW 80.4 billion in Q1 last year, up 11.3% year-on-year. Compared to the KRW 64.1 billion it posted in Q1 2022, the market has expanded 24.4% in 2 years. Cephalosporin antibiotics, which are also called ‘Cepha antibiotics,’ are antibiotics used to manage a wide range of conditions including pneumonia, sore throat, tonsillitis, and bronchitis. The prescription market for cephalosporin antibiotics has undergone significant changes during the pandemic. The prescription market for cephalosporin antibiotics shrank by 29.3% from KRW 60.2 billion in Q1 2020 to KRW 42.6 billion in Q1 2021. In the early days of the COVID-19 pandemic, the prescription market for cephalosporin antibiotics shrank significantly, with the number of patients with infectious diseases such as flu and colds plummeting due to increased personal hygiene measures such as hand washing and mask-wearing. However, the demand for cephalosporin antibiotics surged with the explosive rise in the number of COVID-19 cases in late 2021. In Q1 2022, the prescription value of cephalosporin antibiotics began to rebound, reaching KRW 64.1 billion, up 50.6% YoY. Even after the end of the pandemic last year, the demand for cephalosporin antibiotics increased due to a steady number of confirmed daily COVID-19 cases and the rising number of flu and cold cases that arose after the lifted mask mandate. In Q1, the prescription market for cephalosporin antibiotics soared 88.8% in 3 years compared to Q1 2021. Cefaclor, the leading cephalosporin antibiotic, has recently grown significantly after the prescription market contracted in the early days of the COVID-19 outbreak. The outpatient prescription market for Cefaclor shrank by 23.2% from KRW 31.8 billion in Q1 2020 to KRW 24.4 billion in Q1 2021. However, from the end of 2021, the prescription market expanded at a rapid pace. In Q1, Cefaclor’s prescription size was KRW 40.6 billion, which is a 66.4% rise in 3 years from Q1 2021. Daewoong Bio was the dominant player in the Cefaclor market. Prescriptions of Daewoong Bio's Ceclor in Q1 was KRW 3.4 billion, up 20.1% YoY. Prescriptions of Ceclor have nearly doubled in 3 years from 1.8 billion won in Q1 2021. Prescription sales of Withus Pharamceutical’s Withus Cefaclor have increased sixfold from KRW 500 million in Q1 last year to KRW 2.8 billion in a single year. Other companies in the cephalosporin market, such as HLB Pharma, Mothers Pharmaceutical, Celltrion Pharm, Samsung Pharm, and Theragen Etex, generated over KRW 1 billion in prescriptions in Q1. Cephalosporin antibiotics have recently emerged as a new cash cow for pharmaceutical companies due to such a significant increase in demand. Many pharmaceutical companies had been considering withdrawing from the cephalosporin antibiotics business due to the declining market rising raw material costs and low drug prices. However, with the recent increase in demand, pharmaceutical companies are rather facing an urgent need to secure more supply. Daewoong Bios, which has been leading the market for cephalosporin antibiotics, announced last year that it would invest KRW 50 billion to build a new plant dedicated to the production of cephalosporin antibiotics. The current cephalosporin antibiotics plant in Seongnam, South Korea, will cease operation after this year due to outdated facilities. Once the new plant is completed, the company plans to further expand its antibiotic business. Separation of factories has become mandatory for the production of cephalosporin antibiotics since 2011. Since separate manufacturing facilities are required for its production, not many companies own dedicated facilities. One pharmaceutical company official said, "Cephalosporin antibiotics are a representative drug supplied through active consignment agreement. With the recent surge in demand, we have been struggling to secure sufficient supplies from our consignees.”
- Company
- Another petition for reimb listing of TNBC drug 'Trodelvy'
- by Eo, Yun-Ho May 07, 2024 05:50am
- Gilead Science Korea’s Trodelvy (Sacituzumab govitecan). There is now yet another petition requesting reimbursement listing of the new breast cancer drug 'Trodelvy'. There was already a previous posting with 50,000 votes in January of last year. It appears that another petition was posted because it has been three months without any updates. A petitioner, who introduced themselves on the national petition platform on May 2nd as a stage 4 triple-negative breast cancer (TNBC) patient, pleaded for support in need of Trodelvy reimbursement, stating, "The tumor is growing too fast. I earnestly request the community to participate to extend the treatment option to me as a stage 4 patient." There has not been any updates regarding Gilead Science Korea’s Trodelvy (Sacituzumab govitecan), a treatment of triple-negative breast cancer (TNBC), being considered for the Drug Reimbursement Evaluation Committee (DREC) after the clearance of the Cancer Disease Review Committee of the Health Insurance Review and Assessment Service (HIRA) in November of last year. Trodelvy is the first Antibody-Drug Conjugate (ADC) composed of a monoclonal antibody that binds to the cell surface antigen Trop-2 and a DNA TOP1 inhibitor payload called 'SN-38,' which destroys cancer cells. It received approval as a treatment for adult patients with unresectable locally advanced or metastatic triple-negative breast cancer (mTNBC) who have received two or more prior systemic therapies, including at least one for metastatic disease, from the Ministry of Food and Drug Safety (MFDS) in May last year. Although Trodelvy is the only drug to have demonstrated improvements in overall survival period among those used for second-line treatment or more of metastatic TNBC, it is stalled at getting the reimbursement listing due to cost-effectiveness evaluation. One factor that can anticipate reimbursement coverage is recognition for innovativeness, as seen in the case of Enhertu (trastuzumab deruxtecan), which was listed in April and received a benefit from the government based on its ICER value. Whether Trodelvy will be considered for the upcoming DREC review is to be watched. Meanwhile, Trodelvy demonstrated its clinical effectiveness through the Phase 3 ASCENT clinical study. It has shown a 49% reduction in the risk of death and a 57% improvement in progression-free survival (PFS) in adult patients with unresectable locally advanced or metastatic triple-negative breast cancer (mTNBC) who have received two or more prior systemic therapies, including at least one for metastatic disease, compared to patients who received single-agent chemotherapy (TPC, Treatment of Physician’s Choice). These effects were observed regardless of the presence of brain metastasis.
- Company
- Hyperparathyroidism drug Orkedia may be reimb in Q3
- by Eo, Yun-Ho May 07, 2024 05:50am
- ‘Orkedia,’ a drug for secondary hyperparathyroidism, has entered its final gateway to insurance reimbursement in Korea. According to industry sources, Kyowa Kirin Korea recently entered into pricing negotiations with the National Health Insurance Service for Orkedia (evocalcet), a new drug for secondary hyperparathyroidism that can develop in patients with chronic kidney disease on dialysis. If the company and the authorities reach an agreement within the 60-day negotiation deadline, the drug could be launched with reimbursement in Q3 this year. The drug passed the Health Insurance Review and Assessment Service's Drug Reimbursement Evaluation Committee last month after receiving domestic approval in November last year. Orkedia is a calcimimetic agent and acts on calcium receptors on the surface of parathyroid cells to inhibit excessive parathyroid hormone (PTH) secretion, thereby reducing serum parathyroid hormone levels Secondary hyperparathyroidism is a condition in which excessive secretion of parathyroid hormone persists due to hypocalcemia caused by decreased kidney function, leading to complications such as bone disease. Orkedia is expected to help expand treatment options for patients who are taking existing medications for secondary hyperparathyroidism. Meanwhile, Kyowa Kirin acquired the rights to cooperative research, develop, market, and manufacture Orkedia in Japan and some parts of Asia with Mitsubishi Tanabe in March 2008 and has since been developing the drug.
- Company
- Fate of Retevmo’s reimb in Korea receives attention
- by Eo, Yun-Ho May 03, 2024 06:00am
- No news has been heard of ever since its drug pricing negotiations broke down in August last year. However, RET inhibitors are a hard-to-forego treatment option for a small number of lung cancer patients. There are only 2 RET-targeted therapies – ‘Gavreto (pralsetinib),’ which Roche Korea introduced from Blueprint Medicines, and ‘Retevmo (selpercatinib’ by Lilly Korea – currently approved in Korea. Both drugs have attempted insurance reimbursement in Korea but were denied reimbursement last year. Adding Roche's market withdrawal for Gavreto, it would be hard for Gavreto to be listed for reimbursement in Korea. The case is not much better for Retevmo either. Retevmo, which was approved in Korea in March 2022, failed to pass the Health Insurance Review and Assessment Service Cancer Disease Review Committee (CDDC) review in May of the same year, but then passed the CDDC review in November and finally passed the Drug Reimbursement Evaluation Committee in May last year. After passing the DREC review, the company entered into drug price negotiations with the National Health Insurance Service in June, raising expectations on Retevmo’s reimbursement. However, the two ultimately failed to reach an agreement. In fact, it was the only news of a drug pricing negotiation failure reported in the past year. As a result, it has become unclear whether or not anticancer drugs targeting RET mutations will ever be covered in Korea. However, there has been one glimmer of hope - Phase III results of Retevmo that were released in October last year. Results from Phase III trials on Retevmo - LIBRETTO-431 and LIBRETTO-531 - were presented at the European Society for Medical Oncology Annual Congress (ESMO 2023) last year. The results were published in the internationally recognized New England Journal of Medicine (NEJM) along with the congress presentation. The LIBRETTO-431 trial presented at the meeting compared Retevmo with platinum-based chemotherapy±pembrolizumab as a first-line treatment in patients with advanced or metastatic RET fusion-positive NSCLC. Key findings in the trial showed that in the ITT-pembrolizumab population, the median progression-free survival (PFS) by an independent centralized review committee (BICR) was 24.8 months in the Retevmo arm, and 11.2 months in the control arm, with a hazard ratio of 0.465. The overall response rate (ORR) by BICR was 83.7% in the Retevmo arm, which was statistically significantly higher than the 65.1% in the control arm. Retevmo was granted conditional authorization for conducting a Phase III trial and had trouble during the registration process as the authorities requested the company for data equivalent to a Phase III trial due to its lack of a Phase III trial. This led to criticism about the evaluation criteria for conditionally approved drugs that were granted fast-track review at the time. Retevmo, which applied for fast-track status under the accelerated review system, had been in reimbursement discussions for about a year and a half to no avail. This time, however, the company has secured solid Phase III trial results. Now it remains to be seen whether Lilly will be willing to try again and whether the government will be willing to reimburse the RET inhibitor. In 2020, Retevmo was approved as the first treatment option for cancer patients with RET alternations in the US after the FDA reviewed the drug through the Accelerated Approval and Priority Review pathway and granted the Breakthrough Therapy & Orphan Drug Designation.
- Company
- Roche Korea appoints Ezat Azem as new general manager
- by Son, Hyung-Min May 03, 2024 05:52am
- Ezat Azem, new General Manager, Roche Korea Roche Korea announced that it had appointed Ezat Azem as the new general manager, effective as of May 1. The new GM first joined Roche’s Israel subsidiary in 1997, after which he served nearly 27 years in the group as a marketing division leader in Romania, Slovenia, and other countries around the world, contributing to the launch and growth of the company’s key products. From July 2019 to most recently, Azem has served as General Manager of Roche Greece. Azem was recognized for his excellent people leadership and expertise, particularly for successfully leading the strategic portfolio expansion in the Oncology, Hematology, and Specialty Care business units. In addition, during his service as GM of Roche Greece, 8 drugs were approved for reimbursement, improving patient access to new drugs in the area. Azem placed great emphasis on partnerships between the private, public, and academic sectors, and helped Roche fulfill its role as a trusted partner in building Greece's healthcare system. This has resulted in significant achievements, including bringing patients, the medical community, and companies together to ensure that patients receive healthcare closer to home. Azem graduated from the Hebrew University, where he majored in Medical Sciences. He later obtained an MBA from Tel Aviv University and completed executive education programs at INSEAD (Institut Européen d'Administration des Affaires) Business School in France and London Business School (LBS) in the U.K. The new GM said, “I am delighted to be joining Roche Korea, a country known for its excellent healthcare infrastructure. I look forward to contributing to improving the health of patients in Korea by introducing Roche’s innovative new drugs and personalized treatments, drawing on my various expertise.”