- LOGIN
- MemberShip
- 2026-06-06 15:07:48
- Company
- NMOSD drug ‘Uplizna’ fails reimbursement again in KOR
- by Eo, Yun-Ho Apr 01, 2026 08:15am
- Uplizna, a new drug for neuromyelitis optica spectrum disorder (NMOSD), has failed to secure reimbursement listing in its second attempt.According to Dailypharm coverage, Mitsubishi Tanabe Pharma Korea’s pricing negotiations with the National Health Insurance Service (NHIS) for Uplizna (inebilizumab), a treatment used to treat adult patients with neuromyelitis optica spectrum disorder (NMOSD) who are positive for anti-Aquaporin-4 (AQP4) antibodies, have ultimately collapsed.Although both sides made efforts to continue discussions by extending the negotiation period, it is understood that disagreements arose over adjustments to the expenditure cap.Consequently, it is expected that Uplizna, which had been gaining expectation as a new treatment option for NMOSD, will face significant challenges in establishing a practical prescription environment in the domestic market for the time being.Uplizna had also previously halted its listing process last October at the pricing negotiation stage due to supply-related issues.At the time, the company had accepted the “below the evaluated amount” condition set by the Health Insurance Review and Assessment Service's Drug Reimbursement Evaluation Committee and entered price negotiations. However, the parties failed to reach an agreement within the 60-day negotiation period. Subsequently, HIRA attempted to enter into extended negotiations, but the talks could not begin as the pharmaceutical company was unable to ensure domestic supply.Supply-related issues for Uplizna remain unresolved. The drug was originally developed by Amgen, while Tanabe holds commercialization rights for Korea and other Asian countries through a licensing agreement.It remains to be seen what steps Uplizna will take next after failing its second attempt to secure reimbursement coverage in Korea.NMOSD occurs when AQP4 autoantibodies, a disease-specific biomarker produced by B cells, bind to AQP4, a target antigen present on glial cells in the central nervous system, and activate the immune responses, causing nerve damage.Uplizna is an anti-CD19 human monoclonal antibody that selectively binds to CD19, a B-cell-specific surface antigen, depleting B cells that produce AQP4 antibodies, thereby preventing disease relapse.The safety and efficacy of Uplizna were demonstrated in the N-MOmentum study, which evaluated the use of Uplizna monthly in 230 patients without the use of concomitant immunosuppressive agents.Study results showed that 89% of patients treated with Uplizna did not experience a relapse during 197 days of follow-up, resulting in a 77.3% reduction in the risk of relapse compared to placebo. Safety evaluations of Uplizna also showed comparable rates of adverse events to the placebo group.
- Company
- Patient & Consumer Alliance says "Structural reform needed for drug pricing reform"
- by Son, Hyung Min Mar 31, 2026 08:45am
- Patient & Consumer Alliance for Healthcare Rights (PCA) provided both positive evaluations and concerns over structural limitations regarding the recent drug pricing system reform. The drug price cut is a significant change; However, they argue that a breakthrough reform remains difficult as long as the market remains centered on rebate-driven structures.On the 27th, the PCA stated as such after the Health Insurance Policy Review Committee approved the "Measures to Improve the National Health Insurance Drug Pricing System" the previous day.The Patient & Consumer Alliance for Healthcare Rights (PCA) was officially launched on the 24th joined by four public citizens and patient organizations, including the Consumers Union of Korea, the Korea Alliance of Patient Organizations, Citizens' Movement for Consumers, and the Korean Organization for Rare Diseases, with the goal of shifting the current "government-led and provider-centric" medical structure toward a patient- and consumer-centered one (photo= Korean Organization for Rare Diseases).Previously, the Ministry of Health and Welfare (MOHW) finalized a plan to lower the drug price calculation rate for generics and off-patent drugs from the current 53.55% to 45%. The PCA evaluated this measure as "meaningful progress, marking the first reform of the generic drug pricing structure in 14 years."However, the PCA stated that price reductions alone are insufficient to eradicate the practice of rebates.The PCA stated, "Drug prices are not commensurate with rebates and that rebate practices will persist as long as the unfair competitive structure across licensing and distribution remains intact."They also raised concerns about the tiered price adjustment structure and the newly introduced "New Innovative Pharmaceutical Company" system included in the reform.According to the PCA, "The grace period of up to 10 years delays market restructuring," adding, "New Innovative Pharmaceutical Company" system lacks clear criteria and evaluation methods, potentially allowing companies without innovative capabilities to be incorporated into the support system.Furthermore, the PCA identified potential issues with the government's policies supporting the pharmaceutical and biotech industries. They argued that if this continues, with savings from price cuts being reinvested in industrial support, it could result in using taxpayer money to sustain companies with uncertain competitiveness.The PCA further stated, "The scale and performance of investments into pharmaceutical R&D over the 26 years since the separation of prescribing and dispensing have never been fully disclosed to the public," adding, "Discussions on additional support should be provided only after independent performance evaluations are made public."Issues were raised regarding the sales structures of certain companies within the Korean pharmaceutical market.The PCA pointed out, "There may be so-called bogus pharmaceutical companies that maintain market presence through rebate-driven sales via Contract Sales Organizations (CSOs) without possessing production facilities or research capabilities," and argued that "These companies undermine the competitive foundation for legitimate companies."The PCA stated, "If this structure persists even after lowering drug prices, companies relying on rebates, rather than those with true competitiveness, will maintain their market status," and adding, "The key is the normalization of the competitive order rather than just price reduction."The PCA stressed that government support is necessary to improve the pharmaceutical market structure. They called for the immediate launch of a 'Pharmaceutical Market Fair Trade Task Force (TF)' involving the Ministry of Health and Welfare, the Ministry of Food and Drug Safety, and the Fair Trade Commission to promote: ▲Eradicating rebates and strengthening CSO management ▲Establishing exit criteria for pharmaceutical companies without production capabilities ▲Disclosing performance results of financial support for pharmaceutical R&D.The PCA stated, "Drug price reduction is only the beginning, and as long as the rebate structure remains, patients may not receive benefits," adding, "The government must prioritize creating a foundation for fair competition before returning financial savings to the industry."The PCA concluded, "We hope this pricing reform serves as a starting point for reform leading to a fair pharmaceutical market," and stated, "The alliance would continuously monitor the implementation process." The PCA emphasized that government support is necessary to improve the pharmaceutical market structure.
- Company
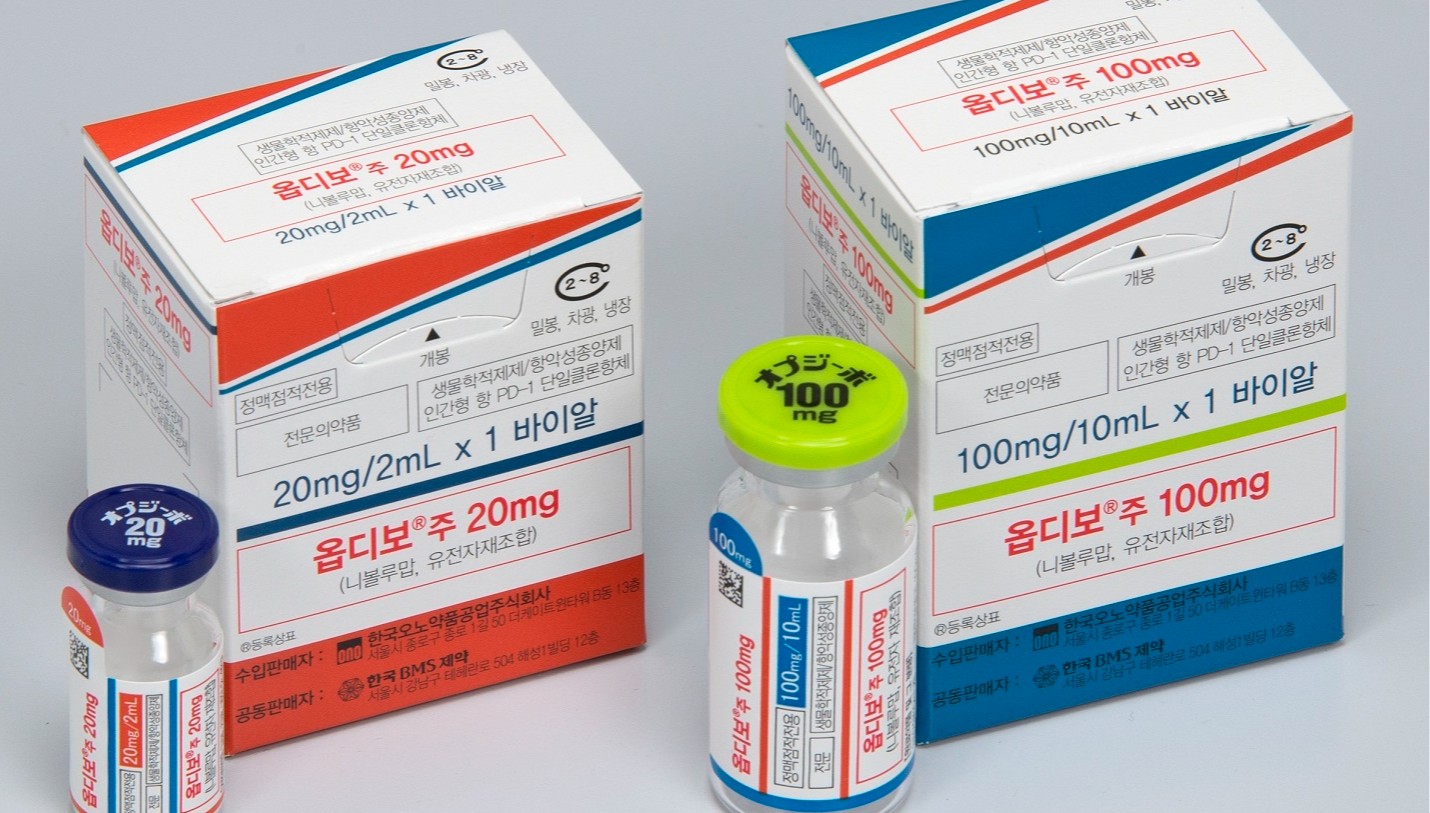
- Opdivo reattempts reimb expansion for first-line liver·lung cancer
- by Eo, Yun-Ho Mar 31, 2026 08:45am
- Immunotherapy Opdivo is set to reattempt reimbursement listing for liver and lung cancer in Korea.According to industry sources, Ono Pharmaceutical Korea’s PD-1 inhibitor Opdivo (nivolumab) is expected to be submitted to the Health Insurance Review and Assessment Service (HIRA) Cancer Disease Deliberation Committee in April.Last October, Opdivo failed to secure coverage criteria from the Cancer Disease Deliberation Committee for first-line treatment of hepatocellular carcinoma and non-small cell lung cancer. At that time, reimbursement criteria were only established for pleural mesothelioma.Ono immediately resubmitted applications for reimbursement expansion for the two indications, which will be reviewed again next month.Opdivo is essentially one of the earliest immuno-oncology drugs introduced, alongside Keytruda (pembrolizumab). However, it has remained a non-reimbursed indication for first-line NSCLC for an extended period. Discussions on reimbursement for first-line NSCLC have been ongoing since 2021.In hepatocellular carcinoma, Opdivo is indicated in combination with the CTLA-4 inhibitor Yervoy (ipilimumab). This combination regimen has demonstrated the longest survival data among first-line treatment options for hepatocellular carcinoma.The Opdivo + Yervoy combination showed a median overall survival (OS) of 23.7 months in the phase III CheckMate-9DW trial, which included patients with unresectable or advanced hepatocellular carcinoma who had not received prior systemic therapy. This represents a 21% reduction in the risk of death compared to the control group treated with ‘Lenvima (lenvatinib)’ or ‘Nexavar (sorafenib),’ which showed a median OS of 20.6 months.It remains to be seen whether Opdivo, which has faced a rocky road from its initial listing to the reimbursement expansion, will be able to expand its prescription scope this time.
- Company
- "Do we really need BE testing for already-listed generics?"
- by Chon, Seung-Hyun Mar 31, 2026 08:45am
- The pharmaceutical industry is assessing potential losses from price cuts for already-listed generic drugs. Substantial losses are expected as the government has officially announced that the new, lower price calculation rates will apply to already-listed generic drugs.With the standardized price calculation rate lowered and the highest price requirements expanded, generics without direct bioequivalence (BE) testing are expected to see their prices drop by more than 20%. There may be instances where companies rush to conduct bioequivalence studies on already approved products to avoid price reductions.On the 26th, the Ministry of Health and Welfare (MOHW) finalized the "Measures to Improve the National Health Insurance Drug Pricing System" during a meeting of the Health Insurance Policy Review Committee, confirming that existing drugs will be adjusted based on the revised calculation standards.Under the reformed system, the price for both off-patent original drugs and generics will decrease from 53.55% to 45% of the new drug's pre-patent-expiry price. The MOHW plans to categorize already-listed drugs into groups based on whether they were listed before or after 2012 and gradually adjust them to the 45% level. Both generics and the off-patent originals with listed generics are subject to these cuts.To maintain drive for new drug development, the MOHW will grant temporary exceptions for "Innovative" and "New Innovative" pharmaceutical companies. Under this scenario, Innovative companies will have their generic price calculation rate set at 49% for four years, while "New Innovative" companies will receive a rate of 47% for three years before eventually reaching the 45% criteria. Companies that do not fall into these categories will also face price cuts over a four-year period, likely dropping to 49% next year, 47% in 2028, and finally 45% in 2029.Under the reformed system, the price for both off-patent original drugs and generics will decrease from 53.55% to 45% of the new drug's pre-patent-expiry price; a generic failing one requirement will drop to 36%, and one failing both will drop to 28.8%.Pharmaceutical companies are primarily concerned about the loss resulting from these price adjustments on existing products. For instance, if a product with annual sales of KRW 10 billion has its price reduced from 53.55% to 45%, it mathematically results in an annual revenue decrease of KRW 1.6 billion. Effectively, KRW 1.6 billion in operating profit per product would evaporate. The price cut range is even greater if the top-tier price requirements, such as performing direct BE studies and using registered drug substances (DMF), are applied to these already-listed generics.Under the reformed system, the penalty for failing to meet top-tier price requirements will expand from 15% to 20%. Since July 2020, a system was introduced where generics could only receive the 53.55% maximum price if they met both the direct BE study and DMF requirements. For every requirement not met, the ceiling price dropped by 15%; failing both resulted in a 27.75% reduction. Currently, under the 15% penalty rule, a generic failing one requirement drops to 45.52%, and failing both drops to 38.69%.However, applying the new 45% requirement and the increased 20% penalty means that a generic failing one requirement will drop to 36%, and one failing both will drop to 28.8%. The price for a generic failing one requirement will be 20.9% lower than current levels, while those failing both will see a 25.6% decrease. Pharmaceutical companies with generics that have not undergone BE studies would have to endure a 20.9% price cut.An industry official stated, "For consigned generics, the price cut could be mitigated by performing a BE study, forcing companies to calculate whether the cost of the study outweighs the benefit of maintaining a higher price."Consequently, companies have begun reviewing the profitability of their generic portfolios. For products where profitability would be significantly damaged by the 20.9% cut, they may seek strategies to minimize losses, such as initiating late-stage BE studies.The industry is concerned that the confusion seen during the two rounds of price re-evaluations for approximately 8,000 generic items in September 2023 and March last year may recur.On September 5, 2023, the prices of 7,355 generic items were reduced by up to 28.6%, which was the first result of the generic price re-evaluation project launched in 2020. At that time, the MOHW announced that generics failing top-tier requirements could maintain their previous prices if they submitted proof of BE studies and DMF use by the end of February 2023. This policy was intended to apply the new 2020 pricing system to previously listed generics.At that time, most of the 7,355 items were hit with a 15% reduction, largely because they lacked BE studies. A total of 145 items saw cuts exceeding 20%, and 125 items saw cuts exceeding 27% because they failed both requirements, resulting in price drops approaching 30%. A total of 179 companies suffered losses from the first round of price cut. Korea Huons had 154 items affected, while Hana Pharm and Daewoong Bio saw cuts to 122 and 104 items, respectively.In March 2024, the second round of re-evaluations resulted in price cuts of up to 27.9% for 948 items. These additional cuts targeted sterile preparations like injections that were newly classified as subjects for equivalence testing.At that time, 125 items containing Artemisia ethanol soft extract saw prices drop by an average of 14.5% and a maximum of 27.4%. Artemisia extract is a natural product-based medicine used for gastric lesions. Stillen is the original product. Because it is difficult to prove equivalence for herbal medicines via traditional blood concentration levels, most of these generics could not fulfill the BE study requirement and were forced to accept the cuts.Drug prices were reduced for 94 generic items of Stillen and 31 generic Stillen 2X. These generic products of Stillen and Stillen 2X had been authorized based on comparative dissolution and comparative disintegration tests rather than bioequivalence (BE) studies. Because they failed to conduct BE studies (one of the requirements for the highest generic drug price) the prices of all these generic products were lowered. Among the 125 items subject to the price reduction, 108 saw their prices decrease by 15% due to failure to meet the BE study requirementPharmaceutical companies gave up conducting BE studies and were forced to accept price cuts, arguing that it is difficult to prove equivalence through BE studies, which compare blood concentrations of active ingredients, because of the specific nature of herbal preparations.There are concerns among pharmaceutical companies that efforts to conduct BE studies for price maintenance may resurface, as generic drug prices will drop even further following the reform of the drug pricing system.While companies previously gave up on BE studies for low-volume products and accepted the 15% cut, the higher 20.9% penalty and lower base price may trigger a vicious cycle of wasteful spending to protect revenue.In fact, during the previous re-evaluation, the rush to conduct BE studies for the sake of price maintenance led to significant social costs.According to the Ministry of Food and Drug Safety (MFDS), BE study approvals rose from 178 in 2018 to 323 in 2020, an 81.4% increase in two years, and reached 505 in 2021, nearly triple the number from three years prior. This phenomenon involved companies conducting new BE studies on products already on the market, then switching from "consigned manufacturing" to "in-house manufacturing" via permit changes to satisfy the "direct BE" requirement and evade price cuts.Once the re-evaluation ended, BE approvals returned to a downward trend, dropping to 296 in 2022, 229 in 2023, and 197 in 2024, returning to levels seen six years ago.Pharmaceutical companies have criticized these mandatory BE studies for already-listed drugs as a "waste of money."They argued that it is exhausting to spend upwards of KRW 500 million per BE study on drugs whose safety and efficacy have already been proven to meet a pricing requirement. Some companies have collectively spent billions of won on these efforts.An industry representative commented, "We are currently calculating the revenue impact and price reduction rates for products undergone price cuts during the last re-evaluation because BE studies were not conducted. We are devising strategies to minimize losses as the new pricing system is implemented."
- Company
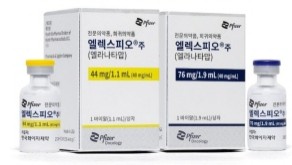
- Bispecific antibody Elrexfio lands in Big 5 Hospitals
- by Eo, Yun-Ho Mar 30, 2026 09:12am
- The multiple myeloma drug Elrexfio has secured access to prescribing at major tertiary hospitals in Korea.According to industry sources, Pfizer Korea’s bispecific antibody therapy Elrexfio (elranatamab) has passed the Drug Committees (DC) of Korea’s ‘Big 5’ hospitals, including Samsung Medical Center, Seoul National University Hospital, Seoul St. Mary’s Hospital, Asan Medical Center, and Severance Hospital.However, Elrexfio remains a non-reimbursed drug. Whether it leads to actual prescribing will depend on future reimbursement listing.Although Elrexfio previously passed the Health Insurance Review and Assessment Service (HIRA) Cancer Disease Deliberation Committee review after a second attempt last year, the reimbursement process is currently on hold. Pfizer is expected to pursue reimbursement listing again in the future.Elrexfio, a fourth-line therapy, is an immune cell–engaging treatment composed of two monoclonal antibodies that recognize the target antigen of multiple myeloma and T cells.Elrexfio is a bispecific IgG2 kappa antibody composed of two monoclonal antibodies that respectively recognize BCMA (B-cell maturation antigen), a target antigen of multiple myeloma, and the CD3 antigen. As such, it represents a novel therapy that enables cytotoxic T cells to directly target BCMA-expressing multiple myeloma cells.Multiple myeloma, a cancer of plasma cells in the bone marrow, is a hematologic malignancy that primarily occurs in the elderly. It is a disease where life expectancy can be extended through sustained treatment. While various new drugs are being developed, monoclonal antibodies and bispecific antibody therapies are currently being used in clinical practice.In particular, the bispecific antibody mechanism is considered a safe and effective treatment for relapsed or refractory multiple myeloma, where resistance increases with each treatment cycle, leading to shorter remission periods and fewer available treatment options.Since life expectancy can be extended through continuous treatment, various options must be available for each treatment stage, and securing reimbursement coverage for fourth-line or later treatments is an urgent priority.Currently, bispecific antibody therapies approved in Korea include Elrexfio, as well as Tecvayli (teclistamab) and Talvey (talquetamab), but all remain non-reimbursed. Amid the failed discussions over coverage of a series of bispecific antibody drugs in the early stages, whether any drug will be granted reimbursement and improve patient access is gaining attention.Meanwhile, Elrexfio was designated by the Ministry of Food and Drug Safety as a GIFT item and was approved as a monotherapy for adult patients who have received more than three lines of treatment, including proteasome inhibitors, immunomodulators, and anti-CD38 monoclonal antibodies, in May last year. The US FDA has also designated it as a breakthrough therapy and granted accelerated approval for the drug.Elrexfio’s efficacy was demonstrated through the Phase II MagnetisMM-3 trial, which was conducted on 123 patients who had not received prior BCMA-directed therapy (i.e., BCMA-naïve patients). Results of Cohort A showed that the drug recorded an objective response rate (ORR) of 61.0% and a complete response (CR) of 37.4%.The progression-free survival (PFS) period was 17.2 months, and the overall survival (OS) period was 24.6 months, demonstrating an unprecedented long-term treatment effect. The data demonstrated that Elrexfio provided long-term survival benefits and slowed down disease progression to improve the quality of life of patients who had no other treatment options.
- Company
- Price cuts trigger ‘100:100’ promotion comeback
- by Kim, Jin-Gu Mar 27, 2026 08:32am
- Amid the government’s announcement of sweeping generic drug price cuts, the so-called “100:100” promotions, where pharmaceutical companies return the full prescription amount as commission to CSOs (contract sales organizations), have re-emerged in the field.The government’s drug pricing system reform is cited as the reason behind the revival of this distorted sales model, which involves accepting heavy financial losses. With price cuts expected as early as July, most pharmaceutical companies are facing a direct hit to profitability. As a result, the industry appears to be resorting to desperate measures to offset losses before the price cuts take effect. The core strategy behind the spread of the 100:100 promotions is to quickly clear inventory before the price cuts while using high commission rates as bait to retain prescribers and encourage them to switch to the company’s products.“100% commission on new prescriptions”…Spread of 100:100 promotionsAccording to the industry sources on the 26th, Company A—a mid-sized firm with annual sales of around KRW 200 billion—recently announced on the 24th that it would implement a 100:100 promotion. The program offers 100% commission on new prescription sales for about 20 products, including its flagship dementia and hypertension combination drugs. The promotion runs from April to June, and any new prescriptions during this period will receive 100% commission for the following 3 months.The notice also includes a condition requiring sales to be maintained for 6 months after the promotion ends. It also states that commissions will be reclaimed if sales fall below the average during the promotion period. With price cuts expected to take effect in July, the move is interpreted as an attempt to secure continued prescriptions for the company’s products even under the new, reduced pricing structure.The so-called “100:100” promotion is now spreading across the industry. Company B, a small firm with annual sales under KRW 50 billion, is reportedly offering “100% commission for securing new clients” for highly competitive products such as lipid-lowering drugs. Company C, with annual sales of KRW 70 billion, also implemented a 100:100 promotion earlier this year for its new products. Industry sources indicate that an additional 2–3 mid-sized or small firms are currently running similar promotions.Government pressure on generic price cuts fueling distorted sales practices?The 100:100 model refers to a structure in which pharmaceutical companies pay CSOs commissions equal to the actual prescription volume. For example, if a CSO secures prescriptions worth KRW 10,000, the company pays the same amount (KRW 10,000) as commission.From the pharmaceutical company’s perspective, this means that every sale results in a loss when considering manufacturing costs, labor, and logistics. While short-term losses are inevitable, this method was often employed to facilitate initial market entry and secure prescribers. However, some have pointed to it as a backdoor for providing illegal rebates.This distorted sales model reportedly disappeared from the front lines for a while following the successive introduction of government policies aimed at improving distribution transparency, including the CSO reporting system and mandatory expenditure reports. A pharmaceutical industry insider stated, “Except for cases where a pharmaceutical company temporarily adopts it ahead of a new product launch, ‘100: 100’ promotions have been virtually nonexistent recently.”However, as fear of drastic price cuts spreads across the industry, the model has resurfaced in the sales field.Last November, the government announced a reform plan to lower the generic pricing rate from 53.55% to the low-40% range. Since then, a sense of crisis has intensified, particularly among small and medium-sized pharmaceutical companies, who feel that “it is impossible to maintain prescriptions through normal sales methods,” ultimately leading to the revival of the ‘100:100’ promotions.For smaller companies, these promotions allow them to push out inventory at the highest possible price before the expected July price cut, thereby preserving revenue. At the same time, high commissions help secure prescription channels, minimizing the impact after the price reduction. This has led to criticism that aggressive government price cuts are inadvertently encouraging irregular sales practices.Another key factor behind the resurgence is the pharmaceutical companies’ “aggressive switching” strategy that exploits the structural vulnerability of CSOs. For CSOs, which receive commissions at a fixed rate, drug price cuts mean a sharp drop in actual revenue. For example, if a CSO earns a 50% commission (KRW 500) on a product priced at KRW 1,000, a price drop to KRW 800 immediately reduces their income to KRW 400. This represents a significant blow comparable to that faced by pharmaceutical companies.The 100:100 promotion exploits this weakness. By offering exceptionally high commission rates, companies entice CSOs to switch prescriptions from competing products to their own. By guaranteeing commissions that more than offset reduced margins, the strategy creates strong incentives for CSOs to engage in product switching.A pharmaceutical industry insider commented, “As the government pushes aggressive price cuts targeting generics, companies have resorted to gambling, spending money to buy tomorrow’s prescriptions instead of investing in R&D. Mid-sized and small firms, which are heavily dependent on generics and face significant losses, are particularly vulnerable to the temptation of 100:100 promotions.”Concerns are also emerging within the CSO sector. A CSO executive stated, “While high commissions may promise immediate gains, they effectively push us toward illegal rebate practices. In a situation where it’s difficult to refuse pharmaceutical companies’ offers, there is growing concern that both pharma sales organizations and CSOs could face mutual destruction, or that market order itself could be disrupted after the price cuts.”Further spread of irregular practices expected during the “gap period” until price cuts take effectThe problem is that these sales practices are likely to intensify in the near term. The government plans to finalize generic drug price cuts through the Health Insurance Policy Deliberation Committee on the 26th. The revised pricing rate is expected to fall in the low-to-mid 40% range. The implementation is expected either in July this year or January next year.This creates a “gap period” of 3 to 9 months before the actual price cuts take effect. During this period, pharmaceutical companies are likely to use aggressive strategies to push out existing inventory. Like those already implementing 100:100 promotions, many may view this as the last opportunity to secure prescriptions.This is why criticism has emerged within the industry that the government’s aggressive push for drug price cuts has paradoxically disrupted the previously stable distribution order. One industry insider criticized, “The government needs to recognize the unintended side effects that price-cut-focused regulatory policies are creating in the field.”
- Company
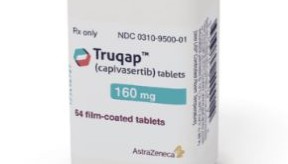
- Anticancer drug 'Truqap' likely to be tabled in CDRC
- by Eo, Yun-Ho Mar 27, 2026 08:32am
- The AKT-targeting oral anticancer drug 'Truqap' has entered a crucial stage for insurance reimbursement listing.According to sources, AstraZeneca Korea submitted a reimbursement application for Truqap (capivasertib), a treatment for hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced breast cancer. The company is currently discussing the date of tabling at the Health Insurance Review and Assessment Service (HIRA)'s Cancer Drug Review Committee (CDRC). It is expected to be considered as early as May.Attention is focused on whether Truqap will pass the CDRC and succeed in final reimbursement listing.Truqap, which was approved in South Korea in April 2024, was launched as a non-reimbursed drug in September of the same year. This drug can be prescribed in combination with fulvestrant for cases that progress during or after endocrine therapy, or recur within 12 months of completing adjuvant therapy.The significance of Truqap's introduction lies in the expanded options for second-line treatment, given the high unmet need following first-line therapy for HR-positive/HER2-negative patients. Cases of HR-positive/HER2-negative patients account for approximately 70% of all breast cancer patients.Truqap's efficacy was demonstrated through the Phase 3 CAPItello-291 study. According to the research results, patients who failed first-line therapy following endocrine therapy (ET) with or without a CDK4/6 inhibitor had a median progression-free survival (mPFS) of approximately 2.5 times longer than in the fulvestrant monotherapy group.Specifically, the mPFS for the Truqap + fulvestrant combination group was 7.3 months, more than double the 3.1 months observed in the fulvestrant monotherapy group, and it reduced the risk of disease progression or death by 50%.Professor Kyung Hwa Park of the Department of Medical Oncology and Hematology at Korea University Anam Hospital explained, "Patients with one or more PIK3CA/AKT1/PTEN mutations, who account for about 50% of HR-positive/HER2-negative patients, may experience faster disease progression. Consequently, there has been a continuous call for second-line targeted treatments for metastatic breast cancer that specifically target these mutations."Furthermore, Professor Park added, "It is very rare for patients with metastatic breast cancer to achieve a complete cure with first-line treatment. Most patients fail treatment and progress to second- or subsequent lines of therapy. Since the mutations targeted by Truqap are subtypes that frequently metastasize to the liver and various other organs, positive clinical effects from the treatment are expected."
- Company
- Takeda’s ‘Takhzyro’ listed for reimbursement
- by Son, Hyung Min Mar 26, 2026 09:28am
- Hereditary angioedema (HAE) treatment strategies are showing signs of shifting from an emergency response–centered approach to long-term prevention. With Takeda’s kallikrein inhibitor ‘Takhzyro’ entering the reimbursement list, changes are expected in the existing treatment paradigm that relied on repeated management of acute attacks.On the 25th, Takeda Pharmaceuticals Korea held a press conference at the Glad Hotel in Yeouido, Seoul, announcing the reimbursement listing and domestic launch of the HAE treatment Takhzyro (lanadelumab).Reimbursement criteria include ▲Cases where, despite receiving the androgen-suppressing agent ‘Danazol’ for six months or more, attacks requiring an average of three or more monthly doses of pirazir (acetate) have occurred in the past six months, ▲ases where Danazol administration is contraindicated or cannot be administered due to side effects, provided that emergency treatment was required an average of three or more times per month during the six months prior to administration of this drug.Professor Kyung-min Ahn, Division of Allergy and Clinical Immunology, Ewha Womans University Seoul HospitalHAE is a rare genetic disorder characterized by recurrent severe swelling of the face, hands, feet, abdomen, and especially the airways, caused by a deficiency or dysfunction of C1 esterase. It is characterized by painful swelling without urticaria or itching, and laryngeal edema can lead to fatal asphyxiation.Takhzyro is a treatment used for the routine prevention of symptoms of hereditary angioedema. This medication works by selectively inhibiting the plasma kallikrein (pKal) enzyme, which triggers bradykinin production, thereby preventing angioedema.The efficacy and safety of Takhzyro were confirmed through Globee’s Phase III HELP study. This study enrolled 125 patients with Type 1 and 2 HAE who experienced an average of 3.7 acute swelling episodes per month.In the trial, the group receiving Takhzyro 300 mg every two weeks showed an 83% reduction in moderate-to-severe acute attacks and an 87% reduction in attacks requiring acute treatment compared to placebo.Additionally, in the HELP OLE extension study, which followed 212 patients for approximately 30 months, an average 87.4% reduction in acute attacks was maintained compared to baseline. No new safety concerns were reported in long-term follow-up.Professor Kyung-min Ahn of the Department of Allergy and Clinical Immunology at Ewha Womans University Seoul Hospital stated, “The reduction in acute angioedema and long-term maintenance of efficacy confirmed in clinical studies demonstrate that Takhzyro is a treatment capable of stably managing the disease.”He added, “In major countries such as the U.S. and Europe, prevention-focused treatments have already become the standard of care. In particular, both emergency and preventive treatments are reimbursed, enabling personalized care. The inclusion of Takzyro in the national health insurance reimbursement system aligns with global treatment strategies.”“Fundamental prevention of HAE is needed”Young Joo Cho, Professor of Allergy and Clinical Immunology, Ewha Womans University Mokdong HospitalHAE treatment is divided into preventive therapy and acute treatment. Danazol, an androgen therapy, has primarily been used for prevention.However, danazol has serious adverse effects in women, including hirsutism, acne, voice changes, and amenorrhea. As a result, regular blood tests (liver function, lipid profile) and ultrasound monitoring are essential.Firazyr is used for acute treatment. Approved in 2018, Firazyr is a subcutaneous injection that blocks the action of bradykinin, which causes vasodilation, and relieves acute swelling within two hours. It has the advantage of allowing patients to self-administer the medication.However, experts agree that in order for patients to maintain a normal life, treatment should shift toward prevention-based therapy.The 2021 guidelines from the World Allergy Organization (WAO) and the European Academy of Allergy and Clinical Immunology (EAACI) clearly define the treatment goal for HAE as “complete disease control and normalization of patients’ lives,” emphasizing the need for long-term preventive therapy.Professor Young Joo Cho of the Department of Allergy and Clinical Immunology at Ewha Womans University Mokdong Hospital explained, “Acute attacks significantly affect patients’ quality of life, including work, leisure, and travel. The reimbursement of Takhzyro represents an important step toward shifting Korea’s HAE treatment strategy from acute management to prevention.”She added, “D Although danazol is inexpensive, it causes side effects that make it virtually unusable for women. Danazol is rarely used overseas. It is regrettable that the criteria for domestic reimbursement for Taczyro require the use of danazol first.”
- Company
- Drug price pressure from US…"K-similars may need to join GENEROUS model"
- by Kim, Jin-Gu Mar 25, 2026 07:21am
- As the U.S. pharmaceutical policies under the Trump administration have been rapidly shifting, suggestions have emerged that South Korean biosimilar companies should consider participating in the CMS (Centers for Medicare & Medicaid Services) new drug pricing model, 'GENEROUS,' as a strategic breakthrough.Professor Dong-chul Seo, Professor Emeritus at Chung-Ang University College of Pharmacy, advised this on the 23rd during a seminar held at the Korea Pharmaceutical and Bio-Pharma Manufacturers Association (KPBMA) titled 'Changes to Pharmaceutical Policy in the Second Trump Administration and Response Strategies for Domestic Companies in South Korea.'Professor Seo analyzed that the GENEROUS model could offer Korean firms an opportunity amid the unpredictable tariff risks of Trump's second presidency and the pressure for Most Favored Nation (MFN)-based price reductions.The GENEROUS (Generating Cost Reductions for U.S. Medicaid) model is a pilot program designed by CMS to reduce costs for U.S. Medicaid. Scheduled to run for five years starting in 2026, this model's key is the application of MFN pricing based on companies' 'voluntary participation'.Participating companies must provide additional rebates to Medicaid referencing the second-lowest price among eight reference countries (UK, France, Germany, Italy, Canada, Japan, Denmark, and Switzerland). Unlike the Inflation Reduction Act (IRA), which mandates price reductions, this is closer to a negotiation-based model in which companies accept price cuts in exchange for securing status in the nation's public insurance programs.CMS introduced this model to lower patients' out-of-pocket costs by reducing drug prices. CMS calculates the Guaranteed Net Unit Price (GNUP) based on the MFN price. Pharmaceutical companies must meet this standard by adding extra rebates to existing ones. State governments accept the prices negotiated by CMS with manufacturers, and further price-reduction negotiations are restricted.Professor Seo stated, "This system is designed to provide additional rebates to the government if foreign drug prices are lower," adding, "Since it is a voluntary participation method with applications accepted until the 30th of next month, companies can review their participation."Analysis suggests that the GENEROUS model may favor biosimilars, as it secures standing in public insurance on the premise of price reductions.For original drugs, accepting MFN pricing is difficult due to the burden of maintaining global pricing tiers. Conversely, biosimilars, which have already been competing at lower price points, are differentiated by the possibility for additional reductions. Accordingly, this is seen as an opportunity to expand market share in the Medicaid market, where the influence of original drugs is diminishing.There is also the possibility of using this as a tool to counter tariff risks. The Trump administration previously proposed 'tariff exemptions for the next three years' to 16 global pharmaceutical companies that agreed to supply at MFN prices. Other mentioned benefits include the provision of FDA's National Priority Vouchers and the application of 'PreCheck' programs. As the GENEROUS model is also a program predicated on fiscal savings, similar policy incentives may be granted.However, suggestions have been made that precise profit-and-loss calculations are needed, given the inherent unpredictability of the Trump administration's pharmaceutical policies. This means a thorough review is required to see if the volume increase from market share expansion outweighs the price cuts and potential tariff burdens.Professor Seo stated, "It would be beneficial for Korean biosimilar manufacturers to participate in this model and experience the actual price application methods," and suggested, “Since generics and biosimilars are highly price-sensitive and have relatively limited global price-linkage effects, participation in the GENEROUS model can be reviewed."Professor Seo further stated, "Companies must comprehensively consider their Medicaid market dependency and sales proportions, the risk of exposing reference country price in MFN is applied, and the impact of price linkages in other nations," and concluded, "Participation may require systematic management of global price exposure and its subsequent effects.”
- Company
- Imfinzi approved as perioperative gastric cancer therapy in KOR
- by Son, Hyung Min Mar 25, 2026 07:21am
- Immuno-oncology drug ‘Imfinzi’AstraZeneca Korea (CEO Eldana Sauran) announced on the 23rd that Imfinzi (durvalumab) has received approval for perioperative treatment of gastric cancer in Korea.The approved indication is for the treatment of patients with resectable gastric or gastroesophageal junction adenocarcinoma, as perioperative therapy in combination with 5-fluorouracil, leucovorin, oxaliplatin, and docetaxel (FLOT) chemotherapy before and after surgery, followed by Imfinzi monotherapy as adjuvant treatment.Under the approved regimen, Imfinzi is administered in combination with FLOT (5-fluorouracil, leucovorin, oxaliplatin, and docetaxel) for two cycles before surgery, followed by surgery. After surgery, two cycles of Imfinzi plus chemotherapy are administered, and then maintenance treatment with Imfinzi monotherapy is initiated. With this approval, Imfinzi becomes the first immuno-oncology drug approved in Korea for perioperative treatment of gastric cancer.While surgery is the primary treatment for gastric cancer, recurrence rates have historically been high even after surgical intervention. In fact, about 40% to 60% of gastric cancer patients die from recurrence. It is also reported that among patients who experience recurrence after surgery, 50% recur within two years and 90% within five years.This approval of Imfinzi is viewed as a new therapeutic approach that can reduce the risk of recurrence and provide survival benefits as a perioperative therapy for gastric cancer.The approval was based on results from the global Phase III MATTERHORN study in patients with resectable gastric cancer and gastroesophageal junction adenocarcinoma. The MATTERHORN study evaluated the efficacy and safety profile of Imfinzi plus FLOT chemotherapy followed by Imfinzi monotherapy as perioperative treatment, compared with a control arm that received FLOT chemotherapy and surgery alone.Study results confirmed that Imfinzi demonstrated clinical benefits in terms of the primary endpoint, event-free survival (EFS), as well as the secondary endpoints of overall survival (OS) and pathological complete response (pCR) rates.Perioperative Imfinzi reduced the risk of disease progression, recurrence, or death from any cause by 29%, showing a significant improvement in EFS. At 24 months, overall survival was numerically higher in the Imfinzi perioperative treatment group at 75.7%, compared with 70.4% in the control group, although statistical significance was not established. The pCR rate was also approximately 2.7 times higher, at 19.2% in the Imfinzi group versus 7.2% in the control group.The safety profile of perioperative Imfinzi observed in the MATTERHORN study was consistent with the known safety profiles for each individual agent.Professor Keun-wook Lee of the Department of Hematology and Oncology at Seoul National University Bundang Hospital stated, “Gastric cancer is a type of cancer in which recurrence occurs in a substantial number of patients even when treated at a resectable stage. In particular, it has been reported that a significant number of patients with stage II–III gastric cancer experience recurrence despite receiving adjuvant therapy after surgery, rendering a new treatment strategy encompassing both pre- and post-operative settings urgently needed.”He added, “In the MATTERHORN study, clinical benefit in terms of event-free survival and overall survival versus placebo was confirmed, and the pathologic complete response rate was also 2.7 times higher than with standard treatment, making this a clinically significant finding.”Hyun-joo Lee, Executive Director of the Oncology Business Unit at AstraZeneca Korea, said, “Through the MATTERHORN study, we confirmed, for the first time as an immuno-oncology agent, clinical benefit in perioperative treatment for patients with resectable gastric cancer. Since Imfinzi has been approved for the treatment of gastric and gastroesophageal junction adenocarcinoma based on this study, we are deeply honored to be able to provide a meaningful new treatment option to Korean gastric cancer patients, for whom there has been a significant unmet need.”