- LOGIN
- MemberShip
- 2026-06-06 10:10:02
- Policy
- Price cuts on existing drugs divided into two phases
- by Jung, Heung-Jun May 18, 2026 09:11am
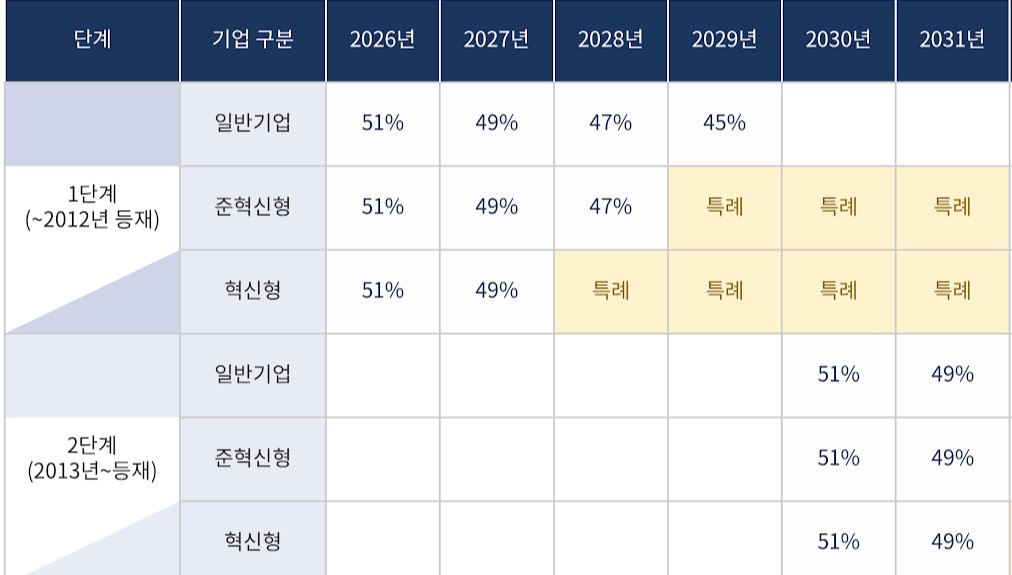
- As the government proceeds with planned pharmaceutical pricing system reforms, follow-up discussions are expected regarding specific classification criteria for price reductions of pre-listed drugs.Key issues include how to distinguish between first-phase and second-phase price reductions for two- and three-drug combination products, as well as how and when later bioequivalence testing should be reflected in drug price reductions.According to industry sources on the 15th, specific criteria for price reductions on pre-listed drugs have not yet been finalized. Discussions on price reductions for pre-listed drugs were not properly addressed during the working-level consultative body meeting between the government and the pharmaceutical industry held in late April.Previously, the Health Insurance Review and Assessment Service (HIRA) decided to divide drugs into two groups based on their listing date in 2012. Price reductions for ingredients listed before 2012 will begin immediately within this year, while reductions for Phase 2 drugs, listed after 2012, will begin in 2030.The timing of implementation differs depending on whether a product is classified into the first or second phase of existing drug price cutsHowever, opinions remain divided regarding the classification of combination products. If the individual ingredients making up a combination drug belong to both Phase 1 and Phase 2 groups, authorities must determine at which timing the combination product itself will face price reductions.The government has stated that if even one ingredient in a combination product was listed before 2012, the combination drug would be classified as a Phase 1 drug.However, there are many issues that require consultation with the industry, such as how to handle cases where even a single ingredient retains a patent or has PMS remaining.Another key issue is how to reduce prices for drugs that failed to meet required standards due to a lack of bioequivalence testing. The reduction rate under the differentiated standards system has been increased from 15% to 20%.For example, if a product price was lowered to 45% and bioequivalence testing was not conducted, it would fall further to 36%. If a rate of 49% is applied, being a product from an innovative pharmaceutical company, it would become 39.2%, while applying a quasi-innovative rate of 47% would drop the price to 37.6%.Since price reductions for pre-listed drugs are implemented over a 10-year period, including a grace period, it is also important to determine how to reflect the results if bioequivalence testing is conducted during that period.In particular, since Phase 2 drugs will only begin facing reductions in 2030, some pharmaceutical companies may attempt to protect reduction rates by conducting bioequivalence studies.Accordingly, industry players are expected to argue that satisfying bioequivalence requirements by the final year converging toward the calculation rate should still qualify as meeting standard requirements.
- Policy
- Calculation rate for generic drug prices set at 45%
- by Lee, Jeong-Hwan May 18, 2026 09:11am
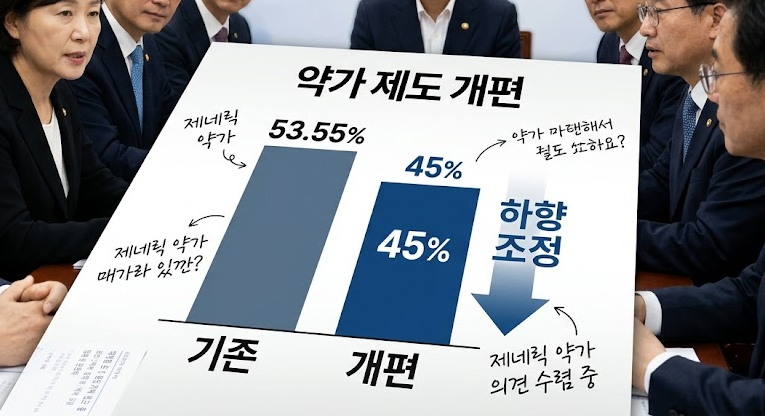
- The government has announced that a drug pricing reform plan, which cuts the drug price calculation rate for currently listed generics from 53.55% to 45%, will take effect on August 1st.The reform plan also includes improvements regarding the management of multiple-listed items, drug price calculations for transfers and acquisitions, support criteria for drugs subjected to market withdrawal, and criteria for semi-innovative pharmaceutical companies.On the 14th, the Ministry of Health and Welfare (MOHW, Minister Jung Eun-kyeong) issued a public notice for the partial amendment to the "Criteria for determination and adjustment of drugs." The ministry plans to finalize the amendment after gathering public feedback by July 13th.The implementation date for the drug pricing reform specified in the amendment notice is August 1st. The government announced that it will gather opinions until July 13th.Generic drug price calculation rate 45%....products failing to meet requirements will be priced below 36%First, the drug price calculation rate for currently listed generics will be reduced from 53.55% to 45%. The calculation rate applied to generics that fail to meet the baseline requirements will also be reduced from 85% to 80%.The baseline requirements for the drug price include whether the company conducted an independent bioequivalence test and whether it used registered drug master file (DMF) ingredients.Generics that meet all baseline requirements will be priced at 45%, those meeting some requirements will be priced at 36%, and products failing to meet any requirements will be priced at 29%.In the case of tiered pricing, the price will be cut once the number of listed items with the same formulation exceeds 13. This is a tighter restriction compared to the current threshold of 20 or more items.If the sum of the newly applied product and the number of currently listed items with the same formulation exceeds 14, the upper limit price will be fixed at 85% of the calculated amount once the price premium period ends.Innovative pharmaceutical companies, semi-innovative pharmaceutical companies, and supply-stabilizing leading pharmaceutical firms will receive preferential drug pricing. Among drugs that meet all baseline requirements, items from innovative pharmaceutical companies will receive a 60% price premium. The premium rate for items from semi-innovative pharmaceutical companies or supply-stabilizing leading pharmaceutical companies will be 50%.Definitions for semi-innovative pharmaceutical companies and supply-stabilizing leading pharmaceutical companies were also established. A supply-stabilizing leading pharmaceutical company is defined as a firm whose ratio of low-profit prevention support drugs, or the ratio of billing amounts among its listed drugs, is 20% or higher.Regarding transfers and acquisitions, the government decided to restrict the succession of existing upper limit prices for items involving a change in a manufacturer's status, excluding inheritance or mergers. Even if a generic item that maintains a high drug price is acquired, the recalculated drug price will be applied from the time of the transfer and acquisition.This regulation is designed to block back-door strategies to evade drug price cuts by purchasing items that maintain high prices.Support for drugs facing production discontinuation will be strengthened. The designation criteria for these drugs are KRW 578 for oral medications, KRW 44 per minimum unit for oral liquids, KRW 3,080 for external preparations, and KRW 5,783 for injections.A new premium clause was also created for pharmaceutical companies that have contributed to supply stabilization. The premium evaluation items include the track record of stable supply fulfillment, national essential medicines, single-listed medicines, low-priced medicines, the use of domestically produced raw ingredients, an annual billing amount of less than KRW 500 million in the previous year, treatments for statutory infectious diseases, and infectious disease crises or urgent supply shortage situations.Price-volume linkage system officiated…implemented on April 1st and October 1st of each yearThe timing of drug price cuts resulting from the price-volume linkage system and the expansion of the scope of use has been unified. The amendment specified that the ex officio adjustment of the upper limit price of drugs will be implemented on April 1st and October 1st of each year, unless there are special circumstances.In addition, a basis was established for pharmaceutical companies to refund the increased expenditure on health insurance incurred during the implementation grace period to the National Health Insurance Service if a drug price adjustment is issued at a time other than the regular implementation date.Meanwhile, the MOHW plans to implement the announced reform plan on August 1st. Regulations related to the regularization of the price-volume linkage will apply starting in January of next year (2027). The first regular drug price adjustment will take effect on April 1st, 2027.
- Policy
- Merck’s two new rare disease drugs receive GIFT designation
- by Lee, Tak-Sun May 15, 2026 02:44pm
- AI-generated imageThe Ministry of Food and Drug Safety (MFDS) is speeding up domestic approval timelines by designating two rare disease treatments from Merck as subjects for Korea’s Global Innovative products on Fast Track (GIFT) program.The MFDS announced that it designated Merck’s desmoid tumor treatment ‘Ogsiveo Tab’ and tenosynovial giant cell tumor (TGCT) treatment ‘Pimicotinib Cap’ as the 70th and 71st GIFT products, respectively. This designation is a measure to support the expedited approval of new drugs from innovative pharmaceutical companies that either have no existing treatment options or have demonstrated improved efficacy and safety.Ogsiveo Tab (nirogacestat hydrobromide), which was designated as a GIFT product on April 21, is a treatment for adult patients with desmoid tumors requiring systemic therapy. The drug suppresses tumor growth by inhibiting gamma secretase (GS) and blocking Notch signaling pathways.The drug has already received approval from the US FDA (November 2023) and the European EMA (August 2025), and was designated as an orphan drug in Korea on February 24, 2026. The MFDS selected it for expedited review based on the lack of existing treatment options.Then, on April 27, “Pimicotinib Cap (pimicotinib hydrochloride monohydrate),” a treatment for tenosynovial giant cell tumor(TGCT), was added to the GIFT list. The treatment works by selectively inhibiting the colony-stimulating factor-1 receptor (CSF-1R) to block disease progression.Pimicotinib is currently under development with FDA Fast Track and Breakthrough Therapy Designation (BTD) status in the United States, but has not yet received full approval in any global market. MFDS acknowledged the drug’s potential efficacy improvements and decided to manage it as a fast-track review product.The two products designated under GIFT will benefit from review periods shortened by approximately 25% compared to standard reviews. In addition, a rolling review of prepared materials and customized consultations with professional reviewers prior to the submission of the marketing authorization application will be provided, which is expected to significantly accelerate the timeline for the drug’s introduction in Korea.An MFDS official stated, “The exact indications and efficacy will be finalized after reviewing the submitted data. We will spare no effort in the review process to ensure that patients with intractable rare diseases can be provided with new treatment opportunities as quickly as possible.”
- Policy
- New drug review timeline cut from 295 to 240 days
- by Lee, Tak-Sun May 14, 2026 09:28am
- As the Ministry of Food and Drug Safety (MFDS) moves to shorten new drug approval review timelines from 295 days to 240 days while strengthening communication with companies. The agency will introduce face-to-face meetings between companies and reviewers prior to the application stage, as well as a “checklist” for companies to self-review their data, aiming to reduce the total approval period by nearly 2 months compared to the previous process.The MFDS announced that it has prepared a revised draft of the “New Drug Product Approval and Review Procedures (Civil Servant Guidelines),” which is currently undergoing a public comment period, and plans to fully implement it starting October 1. This revision, coming approximately one and a half years after the guidelines were established in December 2024, was pursued to maximize the predictability of new drug approvals.From post-submission to pre-submission… introduction of advance face-to-face meetingsThe most significant change is the introduction of ‘pre-NDA meetings.’ Under the original 2024 version, a dedicated team was formed and the review began within 10 days of receiving the application; however, the revised version requires the company and the MFDS to begin discussions 3 months prior to application submission.When a company requests a face-to-face meeting, a dedicated team is formed immediately, and through at least 2 meetings, any deficiencies in the data will be identified in advance. This is expected to serve as a key mechanism to accelerate the entire process by reducing the time spent on ‘requests for additional information,’ which frequently occur during the official review stage.AI-generated graphic imageIntroducing a ‘checklist’ to prevent ‘insufficient data’ at the sourceThe ‘checklist’ system, which requires companies to self-verify the completeness of their submitted data, is another key change in this amendment. Previously, the MFDS would notify companies of required corrections on a case-by-case basis after receiving the data; now, companies must complete a self-inspection using a detailed checklist starting from the face-to-face meeting stage prior to application.Products that undergo this procedure will see a significant reduction in errors or omissions in their documentation, resulting in the “preliminary review” period, conducted immediately after submission, being shortened from the previous 7 days to within 3 days.Approval timeline shortened by 55 days… “295 Days → 240 Days”Through these strengthened communication systems, the Ministry of Food and Drug Safety (MFDS) has set the target approval period for new drugs at 240 days, down from the previous 295 days. This represents a reduction of approximately 55 days compared to the original regulations.In addition, the govenrment has maximized review efficiency by codifying a ‘rolling review’ procedure for frequent exchange of opinions during the review process and by moving the GCP (Good Clinical Practice) site inspection, which previously took place after the first round of supplementary submissions, to the early stages of the review (within 60 to 120 days after submission).An MFDS official stated, “This revision goes beyond simply shortening the timeline; it institutionalizes ‘pre-submission communication’ and ‘self-assessment by companies’ to overcome the limitations identified during the operation of the original version. Once the new procedures take effect this coming October, the speed at which innovative new drugs enter the market, both domestically and internationally, will increase dramatically.”The industry’s response has also been favorable. An official from the Korea Biomedicine Industry Association said, “The introduction of the checklist will help ensure that more comprehensive data is submitted when applying for new drug approval and will likely reduce the need for supplementary data in the future. This is interpreted as a positive gesture where the MFDS and companies join hands to successfully complete the product approval process.”
- Policy
- ‘Fast reimb of Lynparza and Elahere is the solution’
- by Lee, Jeong-Hwan May 13, 2026 09:10am
- There is a growing call to improve the low survival rate of ovarian cancer patients in Korea by rapidly expanding reimbursement for AstraZeneca’s Lynparza (olaparib) and AbbVie’s Elahere (mirvetuximab soravtansine).Expanding reimbursement for ovarian cancer treatments has already been included in the government’s 2026 implementation plan under the Second Comprehensive National Health Insurance Plan, and experts have noted ample rationale for improving access.On the 12th, Professor Yoo Young Lee of Samsung Medical Center presented her view at a policy forum hosted by Representative Joo Young Lee of the Reform Party, under the theme “Improving Treatment Access and Health Insurance Coverage to Enhance Ovarian Cancer Survival.”Professor Lee described ovarian cancer as the most lethal female cancer, noting that while its incidence rate is low, the mortality rate is high once diagnosed. In fact, the incidence rate of ovarian cancer in Korea is approximately 12 cases per 100,000 people, accounting for 2.4% of all female cancers and 1.2% of all cancer cases.However, the number of patients has increased steadily over the past 20 years, rising approximately 2.4-fold from 1,353 in 1999 to 3,263 in 2022. It has been on a steep upward trajectory over the past 5 years, influenced by factors such as an aging population and changes in birth rates.Notably, the 5-year survival rate for ovarian cancer stands at 66.7%, the lowest among major female cancers. Approximately 1,465 deaths occur annually, ranking it eighth in mortality among female cancers.To improve survival rates, Professor Lee emphasized the importance of early diagnosis along with aggressive maintenance therapy.One major reason for the low survival rate is that about 70% of cases are diagnosed at advanced stages (Stage III–IV), when the cancer has already spread extensively within the abdominal cavity.The low survival rate is also influenced by the disease’s specific characteristics: early symptoms are vague, there are no effective early screening methods, and because the cancer is located deep within the abdominal cavity, it metastasizes rapidly after onset.Citing the results of clinical trials (SOLO1, PRIME, PRIMA, PAOLA-1), Professor Lee stated that administering PARP inhibitors to ovarian cancer patients who test positive for HRD (homologous recombination deficiency) through genetic testing can reduce the risk of recurrence by 40–70% and mortality risk by 30–40%.Professor Lee’s point is that survival rates can be significantly improved by combining bevacizumab and Lynparza, but only in HRD patients.In addition, for platinum-resistant ovarian cancer (PROC) patients with frequent relapse, she recommended expanding access to antibody-drug conjugate (ADC) therapies through reimbursement. Elahere is the first treatment in about a decade to demonstrate improvements in overall survival (OS) and progression-free survival (PFS) following bevacizumab.Professor Lee emphasized, “The combination therapy of olaparib, a PARP inhibitor, and bevacizumab extended progression-free survival by nearly 30 months in HRD-positive patients, reducing the risk of disease progression or death by 59%. Olaparib plus bevacizumab is the only combination that has improved overall survival in HRD-positive ovarian cancer.”She added, “The olaparib plus bevacizumab combination is the only Category 1 option recommended in NCCN guidelines and is recognized as standard therapy in major developed countries. However, in Korea, the treatment that has demonstrated overall survival benefits remains non-reimbursed, limiting patient access.”Professor Lee stated, “In the case of platinum-resistant ovarian cancer, Elahere has, for the first time in a long while since bevacizumab, significantly demonstrated an improvement in progression-free survival. As an ADC, it is designed to deliver cytotoxic agents directly to tumor cells, acting like a biologically guided missile.”Professor Lee proposed policy measures such as a pre-reimbursement post-evaluation system, flexible cost-effectiveness assessment, raising or applying flexible ICER thresholds, and incorporating clinical expert opinions more actively throughout the innovative new drug reimbursement process to ensure that unmet clinical needs and clinical necessity in actual treatment settings are faithfully reflected in reimbursement evaluations.She emphasized the need for rapid reimbursement of the bevacizumab + olaparib combination therapy for HRD-positive patients and the ADC drug Elahere for platinum-resistant cases.She concluded, “Ovarian cancer is often diagnosed at Stage III or IV and has frequent recurrence, making it the most lethal gynecologic cancer. Expanding access to innovative therapies through faster reimbursement and broader coverage is urgent. Given that ovarian cancer is a key priority in national coverage policy, prompt expansion of reimbursement access is essential.”
- Policy
- Changes to Price-Volume Agreement (PVA) negotiation guidelines
- by Jung, Heung-Jun May 12, 2026 11:30am
- The scope of drugs eligible for one-time rebate contracts as an alternative to permanent price cuts during Price-Volume Agreement (PVA) negotiations will be expanded.Under the government's policy to address low birth rates, drugs reported increased use due to expanded coverage for infertility procedures will now be eligible for one-time rebate agreements.When medicines from national stockpiles are deployed to respond to legal infectious diseases, they will be included in the list of drugs excluded from PVA negotiations. On the 11th, according to industry sources, the National Health Insurance Service (NHIS) is collecting opinions on the draft amendment to the "Detailed Operating Guidelines for Price-Volume Agreement Negotiations" until this afternoon.The NHIS aims to clarify the targets for one-time rebates, which serve as an alternative to price cuts, and expand their scope by establishing new criteria.As these regulations were originally introduced in response to COVID-19, the targets have been clarified as follows ▲Cases where a production/import request or administrative action from related agencies is confirmed for responding to an infectious disease at the 'Caution' level or higher of the crisis alert ▲Cases where it is confirmed through related agencies that an unavoidable supply disruption or shortage occurred for the sole alternative to the negotiated drug during the year preceding the analysis period due to issues with production facilities or raw material supply.Furthermore, a new clause has been established for cases where the use of drugs under 'infertility procedure reimbursement standards' has increased due to expanded policy support, such as changes to reimbursement criteria.Inclusion in these categories does not automatically mandate a one-time rebate contract. The new regulation also allows for the adjustment of the negotiation reference price for one-time rebate targets.Regarding drugs excluded from negotiations, drugs under a Risk Sharing Agreement (RSA) will be removed from the exclusion list. Drugs used as state-stockpiled materials to respond to legal infectious diseases will be added.For ingredients classified as national stockpiled materials under the Act on the Prevention and Control of Infectious Diseases, any usage of these stockpiled drugs for legal infectious disease response will result in an exclusion from PVA negotiations.In contrast, considering the purpose of the RSA, which is to share the uncertainty of the impact on insurance finances, relevant drugs will no longer be eligible for exclusion from negotiations.The definition of 'voluntary price reduction,' which qualifies a drug for exclusion from negotiations, will also be clarified. Currently, items where the 'reduction rate through voluntary application is greater than the reduction rate according to the negotiation reference price' are excluded from negotiations.However, this will be restricted so that voluntary price reductions resulting from evaluation outcomes, such as reimbursement adequacy re-evaluations or prior price cuts for reimbursement expansion, or those following specific contract terms with the NHIS do not qualify for exclusion.The updated PVA negotiation guidelines containing these details will be implemented following the opinion-gathering period and will apply starting with drugs currently undergoing monitoring or negotiation.
- Policy
- Middle East war crisis accelerate regulatory flexibility
- by Lee, Jeong-Hwan May 12, 2026 11:30am
- Amid the government’s push to address gaps in regional, essential, and public healthcare, the prolonged Middle East conflict is accelerating efforts to ease regulatory barriers through AI and telemedicine.The Ministry of Health and Welfare plans to take the lead in advancing regulatory reforms to fully utilize new medical technologies, such as AI and telemedicine, in order to strengthen regional, essential, and public healthcare and respond to the crisis caused by the war in the Middle East.The Ministry has already begun implementing measures to directly deliver medical supplies, such as syringes, IV sets, and medications, via telemedicine to patients with rare and intractable diseases, who are facing even greater difficulties in securing medical supplies due to the war in the Middle East.On the 9th, the Ministry announced plans to explore regulatory rationalization measures that would allow for the aggressive use of AI and telemedicine as part of efforts to strengthen regional, essential, and public healthcare.Currently, the Ministry is in the final stages of administrative procedures to establish a Regional, Essential, and Public Healthcare Division (hereinafter referred to as the “REPH Division”). Following consultations with the Ministry of the Interior and Safety, a consensus has been reached on the necessity of establishing the division, and efforts to secure the necessary personnel and budget are currently underway.Based on the establishment of the REPH Division and organizational restructuring, the Ministry of Health and Welfare intends to identify additional measures to integrate new medical technologies into regional, essential, and public healthcare.A prime example is the Primary Care Innovation Pilot Project, an initiative where local governments and regional primary care institutions design policies to proactively reform regional, essential, and public healthcare and submit them to the Ministry of Health and Welfare. Upon review, the Ministry provides policy and budgetary support.In particular, as the government is now able to respond to public health needs through dedicated public health officials based on the supplementary budget passed by the National Assembly, it will identify additional cases of regulatory exemptions for AI and telemedicine.An example of such a regulatory exemption is the collaboration between the Ministry of Health and Welfare and the telemedicine platform SolDoc to allow the direct delivery of medical supplies via telemedicine, aimed at minimizing the impact on patients with rare and intractable diseases caused by the fallout from the Middle East war.The Ministry of Health and Welfare has already begun administering the direct delivery of medical supplies to patients with rare diseases using a qualification verification system linked to SolDoc and medical institutions.These supplies, which include syringes, IV sets, suction tips, suction catheters, sterile saline, and disinfectant swabs, are essential for the home treatment of patients with rare diseases. The Ministry plans to gradually expand this in stages to include the delivery of medications as needed.With the passage of the revised Medical Service Act by the National Assembly, telemedicine will be fully institutionalized starting this December. The main provisions of the law include broadly permitting telemedicine for patients with rare diseases and allowing hospitals and higher-level medical institutions to provide telemedicine services under certain restrictions.The Ministry of Health and Welfare plans to expand telemedicine services based on regional, essential, and public healthcare until the Telemedicine Act takes effect.A Ministry official explained, “Since addressing gaps in regional, essential, and public healthcare is a key policy direction for the Ministry, we will develop additional measures to strengthen regional, essential, and public healthcare using AI and telemedicine, alongside the establishment of the new REPH Division.”A medical industry official also noted, “Following efforts to strengthen regional, essential, and public healthcare, and the prolonged Middle East war, the Ministry of Health and Welfare has repeatedly shown signs of seeking to relax healthcare regulations through telemedicine and new AI technologies. We are closely monitoring the situation, as there have been some instances where sufficient consultation with healthcare counterparts has not been conducted under the pretext of regulatory exemptions.”
- Policy
- Review initiated on introducing 'indication-based drug pricing'
- by Jung, Heung-Jun May 11, 2026 09:18am
- The National Health Insurance Service (NHIS) is proceeding with a fiscal impact review for the introduction of an indication-based pricing system. The final decision on whether to implement the system is expected to be determined by the results of this research.In March, the Health Insurance Policy Review Committee (HIPRC) decided to review the feasibility and effectiveness of indication-based pricing. Consequently, the NHIS has initiated follow-up measures by commissioning a research project.According to the NHIS on the 8th, a public bid has been announced for a research project titled “Analysis of Indication-Based Drug Price Evaluation Status and Feasibility Review Study,” which is to be completed by the end of this year. The research is scheduled to run for 5 months following contract signing, with a planned completion date between November and December.The NHIS plans to finalize the research project related to the of indication-based pricing system by November or December. There have been consistent criticisms that the current single-price operation of the national health insurance system has limitations in adequately reflecting the value of innovative new drugs for severe and rare diseases.Through this study, the NHIS will analyze the benefits and effects of improved accessibility under an IBP system, as well as the limitations and costs associated with allocating the health insurance budget. The research findings will serve as a reference for future policy decisions.The contents of the research consists of ▲Literature review of previous domestic and international studies related to IBP ▲Current status of IBP operations in major overseas countries ▲Objective measurement and analysis of patient accessibility benefits and fiscal impacts for multi-indication drugs ▲Opinion surveys and in-depth interviews with the public, academia, the pharmaceutical industry, and the government ▲Comprehensive policy recommendations for system improvement.Specifically, the NHIS plans to evaluate fiscal impacts through drug price simulations for each indication. The study will also cover the types of drugs to be included in the system, reimbursement evaluation, drug price calculation methods, and mechanisms for post-reimbursement expenditure management.Furthermore, the NHIS will examine the overall aspects of the indication-based pricing system, including its pros and cons, operational points of contention, and policy considerations.The NHIS anticipates that "We will derive mid-to-long-term development directions for the 'expansion of scope of use' system based on the categorization of reimbursement expansion for multi-indication drugs."The research results are planned for use in establishing a mid- to long-term roadmap for creating an innovative new drug ecosystem and in informing government policy decisions.The research schedule includes an interim report meeting in September~October and a final report meeting in October~November. The research is scheduled for completion at the end of the year.
- Policy
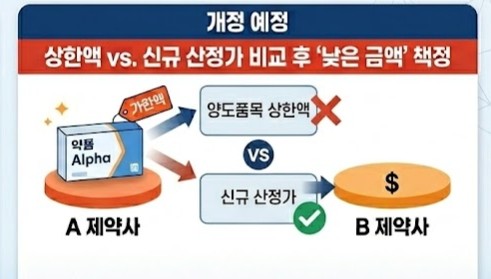
- Inheritance of drug price ceiling to be blocked in transfers
- by Jung, Heung-Jun May 08, 2026 01:15pm
- Changes are expected to the current rules that allow drug price ceilings to be inherited during product transfers. The system is likely to revert to past regulations that apply the lower of the new calculated price and the price at the time of transfer.As a result, acquiring high-priced generics to avoid the impact of drug pricing reform will no longer be viable.According to industry sources on the 6th, a working-level consultative body comprising the Ministry of Health and Welfare and industry representatives recently discussed changes to the transfer regulations that were not brought before the Health Insurance Policy Deliberation Committee in March.The rules governing price ceilings in transfers have changed multiple times. Until 2020, prices were set based on the lower of the final ceiling price of the transferred product and the newly calculated price.However, following the stepwise pricing reform, industry backlash arose as transfers were treated as new listings, leading to significant price drops.In response, the government allowed inheritance of the existing ceiling price starting during transfers in 2021.Now, the government plans to revert to the previous rule, applying the lower of the existing ceiling and the new calculated price.Even if a high-priced generic is acquired, the newly calculated price at the moment of transfer is applied, effectively enforcing a lowest price rule.Pharmaceutical companies that had considered acquiring high-priced generics to avoid pricing reform will lose this option.With the reform, the generic pricing rate has been reduced to 45%, and the reduction rate for products failing to meet the baseline requirements has been strengthened from 85% to 85%. Combined with multi-listing management rules, prices could fall to as low as 30.6% within one year if bioequivalence studies are not conducted.Previously, companies might have chosen to acquire products with higher maintained prices.However, if prices are reset based on the calculation price at the time of transfer, transfers that take existing drug prices into account become meaningless.
- Policy
- Innovative pharmas need permanent pricing incentives
- by Lee, Jeong-Hwan May 08, 2026 01:15pm
- Rep. In-soon Nam of the Democratic Party of Korea has drawn attention by stating that permanent pricing incentives for innovative pharmaceutical companies should be institutionalized in order to advance Korea into a pharmaceutical and biotech powerhouse and create blockbuster new drugs.Rep. Nam criticized the Ministry of Health and Welfare, noting that while the ministry recently formalized a differential drug pricing system between innovative and non-innovative pharmaceutical companies through a resolution by the Health Insurance Policy Deliberation Committee, the inclusion of preferential pricing for innovative pharmaceutical companies only as a temporary provision left some gaps.Regarding legislation to introduce limited international nonproprietary name (INN) prescribing, Rep. Nam also expressed the view that it would be reasonable to either remove penalty provisions for doctors who fail to comply with mandatory INN prescribing regulations or to downgrade and relax these penalties to administrative fines.Her point is that policy and legislation should be pursued with a focus on ensuring that the public can access frequently out-of-stock and essential medicines without barriers, setting aside conflicts between specific professions such as doctors and pharmacists.Having officially announced her candidacy for Deputy Speaker of the second half of the 22nd National Assembly, Rep. Nam In-soon promised that, if elected, she would strive to create a national environment that advances the pharmaceutical and biotech industry and enhances public access to essential medicines.On the 7th, Rep. Nam held a meeting with the press corps and stated, “As the Jae-myung Lee administration has adopted a national agenda to transform Korea into a leader in digital health and the pharma-biotech industry, I will consider various ways to support this goal as Deputy Speaker.”“Pharma-biotech lost priority under Yoon administration… will restore momentum”Rep Nam assessed that while fostering the pharma-biotech industry has always been a priority across administrations, interest declined under the former Yoon, weakening growth momentum.In contrast, she noted that the Lee administration has set a goal of becoming one of the world’s top 5 pharma powerhouses and developing domestic blockbuster drugs, raising expectations for policy reforms across related ministries, including the Ministry of Health and Welfare.She therefore argued for the need to permanently extend preferential drug pricing for innovative pharmaceutical companies with the goal of fostering blockbuster new drugs.Rep Nam stated, “I have consistently advocated for differential pricing between innovative and general pharmaceutical companies, and I welcome its inclusion in the recent reform. However, making the incentive temporary limits its impact on new drug R&D investment.”“The government’s goal is to improve the domestic pharmaceutical industry’s structure using the funds secured through drug price cuts. If we want innovative new drugs and blockbusters to emerge in Korea, preferential treatment for innovative pharmaceutical companies cannot be temporary. Instead of offering temporary preferential treatment to innovative companies, we must remove the ‘temporary’ qualifier to allow for active support.”Rep Nam also noted that the Health and Welfare Minister Eun-Kyeong Jeong has shown a strong commitment to fostering domestic new drug development.She said, “The Lee administration has declared its intention to nurture blockbuster drug companies and has merged the Bio Committee and Bio-Health Innovation Committee. Previously, the Ministry of Health and Welfare did not appear to take a leading role, but after speaking with Minister Jeong Eun-kyung following the integration of the innovation committees, it appears the Ministry intends to take the initiative and carry out its duties. The atmosphere will change going forward.”He noted, “Currently, South Korea lags behind advanced countries in terms of pharmaceutical technology, new drug development, and medical AI, so we must catch up quickly. Since I enacted the Special Act on the Promotion of the Pharmaceutical Industry, preferential measures have been stalled for several years. If elected Deputy Speaker, I will prioritize this area in my legislative work.”“Let’s carry out limited INN prescribing focused on patients, without provisions punishing doctors”Regarding the limited INN prescribing bill currently pending in the National Assembly’s Health and Welfare Committee, Rep Nam expressed the view that even if provisions punishing doctors who violate the mandatory generic prescribing requirement are removed, a legislative environment must be created that ensures patients can access essential medicines without inconvenience.She emphasized that, since limited INN prescribing is a key policy agenda item of the current administration, efforts should focus on addressing resistance from the medical community and ensuring smooth institutional adoption.The current bill mandates INN prescribing for selected drugs designated by the Ministry of Health and Welfare following a government committee review, and stipulates penalties of up to one year in prison or a fine of up to KRW 10 million for violations.Rep Nam clearly stated that these penalty provisions should either be removed entirely or downgraded to administrative fines.She also noted the need to carefully consider concerns raised by the Korean Medical Association regarding the scope of bioequivalence testing and trust in substitution and INN prescribing.Rep Nam said, “The KMA is opposing the measure, raising concerns about the broad scope of bioequivalence testing, and their arguments should be taken seriously. We need further discussion on how to incorporate these concerns into policy. For now, it seems there is a lack of objective data or research on generic substitution and INN prescribing, so we need to accumulate more data.”She added, “Gradual implementation of INN prescribing for out-of-stock and essential medicines could serve as a middle ground between physicians and pharmacists. “Public awareness of generic substitution and INN prescribing has significantly increased during the COVID-19 pandemic, as more people have experienced receiving alternative drugs with the same active ingredient.”Nam emphasized, “We should activate systems such as post-notification of substitution and build more research data to support future legislative discussions. But imposing criminal penalties for INN prescribing violations is inappropriate. A more reasonable approach would be to remove penalties or reduce them to administrative fines, prioritize patient-centered legislation, and consider enforcement measures in stages.”“Warehouse pharmacy regulations need to prevent misuse”The recent amendment to the Pharmaceutical Affairs Act, which strengthens regulations on warehouse-style pharmacy advertising, is also one of Nam’s achievements.She explained that the ultimate goal of the regulation is to prevent excessive consumption and misuse of medicines that could lead to adverse effects.The provision in the Pharmaceutical Affairs Act prohibits expressions that may encourage misuse and delegates authority to the Ministry of Health and Welfare to specify concrete and unique expressions that cannot be used in pharmacy names or promotional slogans through subordinate regulations, prioritizing public safety above all else.Rep Nam said, “We also brought forward the implementation date from six months to three months after the government’s promulgation, and added a supplementary provision allowing public health centers to provide administrative guidance to pharmacists opening new pharmacies before the law takes effect. I was the one who codified the provision for pharmacists to provide medication guidance within the Pharmaceutical Affairs Act. At first, pharmacists weren’t very fond of it, but today, medication guidance has become a crucial system and phrase for ensuring public medication safety.”She continued, “When I look at warehouse-style pharmacies, I'm sorry whether pharmacists can actually provide proper medication guidance to patients. We must prevent the public from perceiving medications as mere commodities. Even after my election as Deputy Speaker, I will engage in policy monitoring to curb the misuse of medications by the public by regulating warehouse-style pharmacies.”