- LOGIN
- MemberShip
- 2026-04-23 17:19:13
- Policy
- 8 items, including Infanrix, will be discontinued next year
- by Jung, Heung-Jun Dec 29, 2025 08:55am
- Product photo of GSK's Infanrix-IPV+Hib Inj, Yuhan Corporation's Bambec Tab, and Hanall Biopharma's Cortisolu InjectionEight items, including GSK's hexavalent combination medicine (included in the National Immunization Program (NIP)), will be discontinued for supply starting next year. On the 2nd of next month, the list of items subject to supply suspension, along with their scheduled dates, will be shared on the drug utilization review (DUR) system.The Health Insurance Review and Assessment Service (HIRA) the list of items reported to be suspended for next year's production, imports, and supply shared with relevant organizations and associations.According to the relevant association on December 26, eight items from seven companies, including Eisai Korea, GSK, and Hanall Biopharma, will be discontinued for supply next year.The included items are ▲Hanall Biopharma's Cortisolu Injection (hydrocortisone sodium succinate) ▲Daewoo Pharm's Tobeson Eye Drops (tobramycin, dexamethasone) ▲Tai Guk Pharm's Hyrosone Cream (hydrocortisone) and Gammarin D Liquid ▲Yuhan Corporation's Bambec Tab 10mg (bambuterol hydrochloride) ▲Genuone Sciences' Pencivir Cream (penciclovir) ▲GSK's Infanrix-IPV+Hib Inj ▲Eisai Korea's Plegridy Pen Inj Starter Pack (peginterferon beta-1a, recombinant), among others.Infanrix-IPV+Hib is a hexavalent combination vaccine supplied for the NIP for infants. It is a vaccine that simultaneously prevents meningitis, pertussis, and polio.The relevant vaccination market has been split between Sanofi's Pentaxim and Infanrix. Following the discontinuation of Infanrix on June 30 of next year, demand is expected to shift to Pentaxim from the second half of the year.The list of pharmaceuticals subject to discontinued supply (January 2026): ▲Hanall Biopharma's Cortisolu Injection (hydrocortisone sodium succinate) ▲Daewoo Pharm's Tobeson Eye Drops (tobramycin, dexamethasone) ▲Tai Guk Pharm's Hyrosone Cream (hydrocortisone) and Gammarin D Liquid ▲Yuhan Corporation's Bambec Tab 10mg (bambuterol hydrochloride) ▲Genuone Sciences' Pencivir Cream (penciclovir) ▲GSK's Infanrix-IPV+Hib Inj ▲Eisai Korea's Plegridy Pen Inj Starter Pack (peginterferon beta-1a, recombinant), among others.Eisai Korea's Plegridy Pen Inj is a new drug for relapsing multiple sclerosis that received domestic approval in 2016 and was listed for reimbursement in 2017. As a recombinant treatment for a rare disease, the decision to discontinue supply was made approximately 10 years after its introduction. Supply will be discontinued starting June 30 of next year, the same time as Infanrix.Hanall Biopharma's Cortisoru Inj will be discontinued starting July 1 of next year. As a corticosteroid, it is expected to be replaced by more than 20 other drugs available in the same therapeutic class.With AstraZeneca’s decision to withdraw Bambec Tab. from the market 31 years after its domestic approval, Yuhan Corporation will also cease sales and supply after December of next year.Bambec Tab is a long-acting beta-adrenoceptor agonist (LABA) used to treat asthma, chronic bronchitis with bronchospasm, emphysema, and other lung diseases. Since combination therapies have been increasingly used for asthma treatment recently, the decline in market competitiveness is considered the reason for the withdrawal.The HIRA plans to provide information on the eight items scheduled for discontinuation next year to physicians and pharmacists through the DUR system on the 2nd of next month.
- Policy
- Imjudo, Imfinzi, Xpovio in reimb discussions with NHIS
- by Jung, Heung-Jun Dec 26, 2025 08:40am
- Imjudo (tremelimumab) and Imfinzi (durvalumab) are in price negotiations with Korea’s National Health Insurance Service (NHIS), bringing the drugs closer to reimbursement listing.Once they clear the final hurdle with the NHIS, Imjudo will be newly reimbursed for use in combination with Imfinzi in hepatocellular carcinoma, while Imfinzi is expected to secure a reimbursement expansion for use in combination with gemcitabine and cisplatin in biliary tract cancer.According to industry sources on the 26th, the NHIS is conducting price negotiations for three new drugs and two coverage expansion items.These drugs all passed the Drug Reimbursement Evaluation Committee in November. AstraZeneca Korea is negotiating the reimbursement of both Imjudo and Imfinzi simultaneously.If negotiations conclude as expected, Imfinzi will secure reimbursement for two combination regimens at once: combination therapy with Imjudo and the GemCis (gemcitabine + cisplatin) regimen.AstraZeneca is expected to expand Imfinzi’s use beyond its previous focus on lung cancer into liver cancer and biliary tract cancer.AstraZeneca is expected to expand Imfinzi prescriptions, previously focused on lung cancer, to include liver and bile duct cancers.Additionally, new drugs under negotiation include Janssen Korea's Balversa Tab 3, 4, 5mg (erdafitinib) and Takeda Korea's Takhzyro Prefilled Syringe 300mg (lanadelumab).Balversa Tab, a new targeted therapy for bladder cancer, received reimbursement adequacy approval for treating adult patients with locally advanced or metastatic urothelial carcinoma. Takhzyro Prefilled Syringe received reimbursement adequacy approval for preventing attacks of hereditary angioedema in adults and adolescents. Both products are now awaiting final price agreements.Xpovio 20 mg Tab (selinexor) from Antengene, which received reimbursement expansion approval alongside Imfinzi in November, has also entered price negotiations. Xpovio was approved for reimbursement expansion for use in combination with bortezomib and dexamethasone in adult patients with multiple myeloma who have received at least one prior therapy.However, unlike the other products, Xpovio’s approval came with a conditional clause stipulating that reimbursement expansion is deemed appropriate only if the company accepts a price at or below the evaluated price. If the company is unable to accept the proposed price, there remains a possibility that negotiations could break down.
- Policy
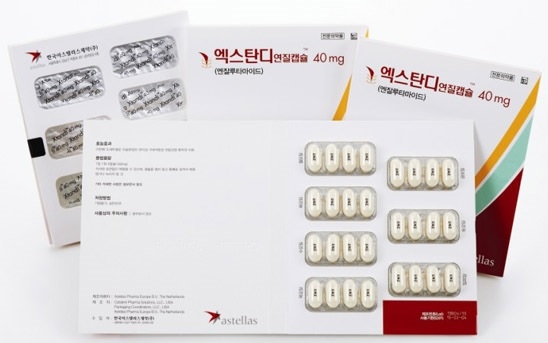
- Second generic for prostate cancer drug Xtandi approved
- by Lee, Tak-Sun Dec 26, 2025 08:40am
- Pharmaceutical companies are rapidly entering the generic market for the prostate cancer treatment ‘Xtandi (enzalutamide)’, which has a market worth approximately KRW 50 billion.Following Alvogen Korea's initial product approval last January, Daewon Pharmaceutical has also successfully obtained approval for its generic drug. These products are expected to establish patent strategies targeting launch after the substance patent expires next June.The Ministry of Food and Drug Safety approved Daewon Pharmaceutical's ‘Enzadex Soft Cap 40mg’ on the 23rd. This product is the second generic drug containing the active ingredient enzalutamide.The first generic was Alvogen Korea's ‘Anamide Soft Cap 40mg’, approved last January. A common feature of both Alvogen Korea and Daewon Pharmaceutical's products is that they import the finished drug from Taiwan.Enzadex Soft Cap is indicated for ▲ treatment of asymptomatic or mildly symptomatic metastatic castration-resistant prostate cancer (mCRPC), ▲ treatment of patients with metastatic castration-resistant prostate cancer (mCRPC) previously treated with docetaxel, ▲ treatment of patients with high-risk non-metastatic castration-resistant prostate cancer (mHSPC), ▲combination therapy with androgen deprivation therapy (ADT) for the treatment of patients with hormone-sensitive metastatic prostate cancer (mHSPC), ▲treatment of patients with high-risk hormone-sensitive non-metastatic prostate cancer (nmHSPC) who have experienced biochemical recurrence (BCR). These indications are identical to those of the original Xtandi Soft Cap 40mg.Xtandi’s formulation patent is set to expire on June 27 next year. However, a formulation patent valid until September 11, 2033, remains a key barrier to generic entry. As a result, multiple generic manufacturers have initiated legal actions to challenge or circumvent the formulation patent.Starting with Alvogen Korea in August, GL Pharma, Hanmi Pharmaceutical, Chong Kun Dang, JW Pharmaceutical, and Kuhnil Pharmaceutical have filed passive scope confirmation trials aimed at clearing the formulation patent hurdle.Industry analysts suggest that if the formulation patent is successfully overcome, generics could enter the market as early as June next year.Astellas' Xtandi competes in the first-line prostate cancer treatment market against drugs like Erleada (apalutamide, Janssen) and Zytiga (abiraterone, Janssen).Notably, since November 2023, the patient coinsurance rate has decreased from 30% to 5%, leading to increased usage. According to the pharmaceutical market research institution IQVIA, Xtandi's sales surged from KRW 23 billion in 2019 to KRW 43.2 billion in 2023.As the market expands, domestic pharmaceutical companies are also eagerly entering the prostate cancer treatment market. Hanmi Pharmaceutical has already moved aggressively by launching Abiteron Tab 500 mg, the first generic version of Zytiga, to secure early market share.With Xtandi’s patent expiry approaching, many generic manufacturers are now closely monitoring the opportunity. Generic companies are simultaneously advancing patent litigation strategies and regulatory approvals, aiming for early entry in June next year.Meanwhile, Astellas has preemptively obtained approval for Xtandi Tab, a new formulation last year, to counter the wave of emerging generic drugs. This appears to be a strategy to dilute the market impact of generic drugs by introducing a new formulation product.
- Policy
- PNH drug 'Piasky Inj' for adults·children wins nod
- by Lee, Tak-Sun Dec 26, 2025 08:39am
- The Ministry of Food and Drug Safety (MFDS), led by Minister Yu-Kyoung Oh, announced that it granted approval of 'Piasky (crovalimab)', a new orphan drug from Roche used for the treatment of paroxysmal nocturnal hemoglobinuria (PNH) in adults and pediatric patients aged 12 and older who weigh 40 kg or more, on December 24.PNH is an acquired hematopoietic stem cell disorder characterized by hemolytic phenomena (hemoglobin escapes from red blood cells) that causes release of dark-colored urine during the night.'Piasky Inj' is an orphan drug that inhibits complement-mediated intravascular hemolysis in patients with PNH. Complement proteins assist the immune response by attacking pathogens such as bacteria and viruses.As a complement protein C5 inhibitor, this drug prevents the binding of C5 to abnormal red blood cells produced by hematopoietic stem cell disorders, thereby protecting the cells from lysis.Piasky is the first treatment approved for pediatric use in South Korea, and it is expected to significantly broaden the therapeutic landscape for both adult and adolescent patients (12 years and older).The MFDS stated that it will make every effort to ensure that new treatments for rare diseases are rapidly made available based on its regulatory science expertise, thereby expanding clinical opportunities for patients.
- Policy
- Expanded reimb for Keytruda·Dupixent
- by Jung, Heung-Jun Dec 24, 2025 08:08am
- As the review of expanded reimbursement for Keytruda (pembrolizumab) and Dupixent (dupilumab) has passed the Health Insurance Policy Review Committee (hereafter referred to as the committee) today (DEC 23), the out-of-pocket cost will drop by up to 95% starting next year.The committee also finalized a restructuring of the compensation system for clinical laboratory testing CDMOs, the formulation of an adjustment system for relative value units, and the launch of an 'innovative pilot project for community-based primary care', set for the second half of next year.The Ministry of Health and Welfare (MOHW) approved expanding health insurance coverage for Keytruda and Dupixent during the committee. While Keytruda was previously covered for four cancer types, including non-small cell lung cancer (NSCLC), coverage will now extend to 17 therapies across nine additional cancer types.Specifically, the expanded coverage includes head and neck cancer, gastric cancer, esophageal cancer, endometrial cancer, small bowel cancer, biliary tract cancer, colorectal cancer, triple-negative breast cancer (TNBC), and cervical cancer.For patients meeting the eligibility criteria for these expanded indications, the annual out-of-pocket cost per patient is expected to drop significantly, from approximately KRW 73.02 million to KRW 3.65 million (with a 5% co-payment for monotherapy).Dupixent, which was previously covered for chronic severe atopic dermatitis, will also be covered for severe Type 2 inflammatory asthma starting in January. The annual cost for severe asthma patients is expected to decrease from KRW 15.88 million to approximately KRW 4.76 million (based on a 30% co-payment).The committee also concluded this year's reimbursement re-evaluation for eight therapeutic ingredients. The reimbursement for spherical adsorptive carbon and artemisia herb extract will be maintained due to price reductions.2025 Reevaluation Results for Pharmaceutical Reimbursement Appropriateness: Reimbursement maintained for 1. olopatadine hydrochloride 2. Clematidis Radix-Trichosanthes Root-Prunella Spike 3. Bepotastine…Drug price reduced and reimbursement maintained for 7. spherical adsorptive carbon and artemisia herb extract (upon voluntary requests for drug price reduction by pharmaceutical companies).The oral formulation of L-aspartate-L-ornithine will remain covered, though its reimbursement criteria will be restricted to hepatic encephalopathy. For three other ingredients, including sulglicotide, the evaluation has been deferred on the condition that a portion of the reimbursement will be recouped if clinical trials fail to prove efficacy.Abolition of clinical lab management fees... new institution-specific fees establishedThe compensation structure for CDMO clinical laboratory testing will be reformed. The consignment fee, which overlaps with existing testing fees, will be abolished, and new fees specific to the CDMO institutions will be established. Furthermore, the billing and payment system will be improved to prevent diagnostic fee discounting.The CDMO fee criteria will be determined based on ▲the current management fee ▲the respective roles of the institutions ▲the financial impact during the regular RVU adjustment process. The KRW 240 billion saved from the abolition of the management fee will be reallocated to increase reimbursement for under-compensated areas, such as consultation fees.Examples of Lab Fee Adjustments: Examples of over-compensated areas, and actual adjustments will depend on finalized cost data. Under-compensated areas within clinical lab testing will not face fee cuts but will be prioritized for balance adjustments.The MOHW plans to revise relevant CDMO regulations in the first half of next year and implement the changes in alignment with the regular relative value unit adjustment cycle. Certification standards for clinical laboratory testing will also be updated.Regular adjustment of the relative value unit (previously updated every 5-7 years)To rationalize the fee compensation system, the government will formulate a regular adjustment model for the relative value unit. Previously, relative value unit restructuring occurred every 5 to 7 years, which critics argued failed to reflect rapid changes in medical practice.The government will review under-compensated and over-compensated services based on medical cost analysis and adjust them to balanced fees.In particular, funds from adjusting for over-compensation in areas such as clinical laboratory tests and imaging (CT and MRI) will be redistributed to under-compensated basic medical services, such as consultation and hospitalization fees at clinics and hospitals. Funds will also be directed toward strengthening compensation for essential healthcare, including surgeries and care that are severe·emergency and pediatrics·deliveries.Medical cost analysis results for relative adjustment...Low margins for drug administration and dispensingThe committee discussed the 2023 medical cost analysis results calculated by the Medical Cost Analysis Committee. These results will serve as the foundation for the 2026 regular RVU adjustments. The '2023 Fiscal Year Cost Analysis Report', which for the first time includes cost-to-revenue ratios by healthcare institution type and specific fee items, is scheduled for publication in the first quarter of next year.The analysis was expanded to include tertiary hospitals and clinics in addition to general hospitals. The report calculated the cost-to-revenue of reimbursements following the establishment of standardized medical cost calculation guidelines.Key findings regarding the cost-to-revenue ratio for covered services (based on tertiary hospitals) showed that clinical laboratory fees (192%), special radiologic imaging fees (169%), and radiation therapy fees (274%) had relatively high profit margins compared to costs.Conversely, drug administration and dispensing fees (11%), basic physical therapy (33%), and basic consultation fees (63%) generated significantly lower revenue than costs.'Innovative Pilot Project for Community-based Primary Care' to launch in JulyThe pilot project will launch next year, initially targeting patients aged 50 and older who require integrated management, with plans for gradual expansion.Under this project, registered patients can receive personalized preventive care, disease and medication management, and lifestyle coaching at their designated clinics, linked to their health check-up results. When necessary, patients can be referred to appropriate medical institutions or receive home-based primary care.Clinics that complete the required training are eligible to participate. Institutions capable of providing multi-professional, multidisciplinary team support can join as hub institutions.The government will introduce the 'Primary Care Functional Enhancement Integrated Fee', which compensates for 'patient registration and continuous management efforts' rather than traditional fee-for-service. The MOHW also plans to pilot multidisciplinary team-based service support and performance-based rewards.The pilot program is scheduled to run for three years, from July 2025 to 2028, with plans to expand its scope to additional regions and institutions starting in 2029.
- Policy
- Gov't to enforce non-face-to-face care Medical Service Act
- by Lee, Jeong-Hwan Dec 24, 2025 08:07am
- Following a Cabinet meeting today (December 23), the South Korean government is set to announce an amendment to the Medical Service Act that formally institutionalizes non-face-to-face care. The Ministry of Health and Welfare (MOHW), led by Minister Jeong Eun Kyeong, is the primary department overseeing this transition.Currently operating as a pilot program, non-face-to-face care will officially take effect one year from the date of announcement, in accordance with the supplementary provisions of the Act. This means that December 24, 2026, will mark the official date when non-face-to-face care becomes a permanent, legalized system in South Korea.This legislative reform comes 15 years after the first amendment proposal was submitted during the 18th National Assembly in 2010.The core principle of the Act is that non-face-to-face care will be centered on returning patients and primary care clinics.First-time patients are also eligible for non-face-to-face care, but with specific caveats. Based on MOHW standards, they can only apply for services within their residential area and will be subject to restrictions on the types of drugs prescribed and the total number of prescription days allowed.For the first time, a provision for the delivery of pharmaceuticals outside of pharmacies will be legalized. This applies to specific groups, including residents of islands or remote areas, long-term care recipients, registered persons with disabilities, patients confirmed with Class 1 or 2 infectious diseases, and patients with rare diseases.Prescribing narcotics or psychotropic substances via non-face-to-face care is strictly prohibited. Additionally, the use of electronic prescriptions will be officially institutionalized specifically for non-face-to-face care services.The National Assembly maintained that while the principle of in-person treatment remains paramount, this legislation provides the legal grounds to supplement care via non-face-to-face care. It also establishes the Minister of Health and Welfare’s authority to manage and supervise non-face-to-face care services via online platforms.Those intending to provide or operate a non-face-to-face care platform must report to the MOHW. Furthermore, if the number of platform subscribers exceeds a certain threshold, the operator must apply for official certification from the MOHW Minister.The MOHW Minister reserves the power to revoke certification if a platform obtains it through fraudulent means or fails to meet the required certification standards.The platforms are prohibited from interfering with a physician’s professional medical judgment, encouraging the misuse or abuse of medical services or pharmaceuticals, brokering, inducing, or instigating collusion in violation of the Pharmaceutical Affairs Act.Furthermore, the platforms are prohibited from demanding or receiving financial incentives, goods, or other economic benefits from medical professionals or pharmacies in exchange for patient or prescription referrals.Prohibited provisions also include recommending or inducing patients to choose specific medical institutions, pharmacies, or medical devices.Both doctors and pharmacists are now mandatorily required to use and verify the Drug Utilization Review (DUR) system when prescribing or dispensing narcotics and psychotropic substances during both in-person and non-face-to-face care consultations. Failure to comply will result in a fine of up to KRW 1 million. This provision was institutionalized as a safeguard during the legalization of non-face-to-face care.Meanwhile, an amendment to the Pharmaceutical Affairs Act aimed at prohibiting non-face-to-face care platforms from concurrently operating as pharmaceutical wholesalers (which was expected to pass alongside the telemedicine bill) remains pending in the National Assembly. The timeline for its final approval remains uncertain.
- Policy
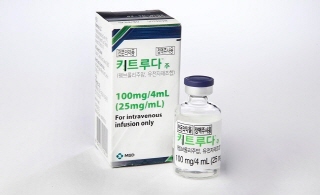
- The implications of Keytruda’s reimb expansion
- by Jung, Heung-Jun Dec 24, 2025 08:06am
- MSD Korea’s immune checkpoint inhibitor Keytruda (pembrolizumab) is expected to complete the process of expanding its reimbursed indications at the Health Insurance Policy Deliberation Committee (HIPDC) meeting scheduled for today (the 23rd).Following approval by the committee, Keytruda’s reimbursement will be significantly expanded from January next year, increasing from the current 7 indications across 4 cancer types to 18 indications spanning 13 cancer types.This marks the culmination of a process that began with the reimbursement application in 2023—nearly three years in the making. Beyond the numerical addition of reimbursement for 9 additional cancer types and 11 new indications at once, the decision signifies enhanced treatment accessibility for previously underserved cancer types.It also stands as a significant precedent, having found a breakthrough for expanding reimbursement indications under the existing RSA framework before the government’s formal introduction of its indication-specific drug pricing system.According to industry sources on the 23rd, the National Health Insurance Service's Health Insurance Review and Assessment Service (HIRA) will vote on expanding Keytruda's reimbursement indications at its meeting today at 2 PM.Starting next January, Keytruda will be reimbursed for gastric cancer, esophageal cancer, endometrial cancer, colorectal cancer, squamous cell carcinoma, cervical cancer, breast cancer, small cell lung cancer, and bile duct cancer. Previously, reimbursement was applied to 7 indications across 4 cancer types: non-small cell lung cancer, Hodgkin lymphoma, melanoma, and urothelial carcinoma.While coverage was previously concentrated on non-small cell lung cancer, the expansion will now allow insurance coverage for a broader range of patients, including those with women’s cancers such as endometrial, breast, and cervical cancer.Furthermore, for several cancer types, including endometrial cancer, small cell lung cancer, and bile duct cancer, reimbursement will be extended to patients with MSI-H (microsatellite instability-high) tumors, significantly improving access for patient groups that had previously been excluded from treatment options.A milestone in multi-indication coverage ahead of indication-based pricingKeytruda, which received a multi-indication approval, has long been at the center of discussions surrounding the potential introduction of an indication-based pricing (IBP) system in Korea.While the government recently announced plans to introduce IBP through a drug pricing system reform, concrete measures have yet to be finalized. In this context, Keytruda's case can be seen as an example where the Health Insurance Review and Assessment Service (HIRA), the National Health Insurance Service (NHIS), and the pharmaceutical company found a broad solution for expanding coverage within the current Reimbursement Strategy Agreement (RSA) framework.With several other multi-indication therapies awaiting reimbursement review, this case is expected to serve as a reference model for future reimbursement decisions, regardless of whether IBP is formally adopted.Keytruda currently holds approvals for 18 cancer types and 35 indications, so further attempts to expand reimbursement coverage are anticipated going forward.Of course, this expansion of Keytruda's indications does not eliminate the need for IBP. How the government's drug pricing system will accommodate blockbuster drugs with multiple indications remains an unresolved policy challenge.However, the significance of Keytruda's recent coverage expansion lies in its active use of RSA to achieve results effectively approaching an indication-based pricing system. The case is meaningful not only for enhancing patient access to treatment but also for clarifying the extent to which existing reimbursement mechanisms can be leveraged to address complex multi-indication therapies.
- Policy
- 3 supply shortage drugs set to receive a price premium
- by Jung, Heung-Jun Dec 23, 2025 08:00am
- Hanmi Pharm's Vildagle Tab and Kyongbo Pharm's Vilda TabWhile drug prices for approximately 4,000 items will be reduced next January, including those affected by the Actual Transaction Price adjustment, prices for certain products at risk of supply shortage will be increased.These adjustments apply to drugs for which the government is either extending price premiums due to low profitability or raising prices to offset production costs for 'discontinuation-prevention drugs'.According to industry sources on December 22, the price premium period for two salt-modified vildagliptin products, scheduled to expire in January, will be extended for another year.Hanmi Pharmaceutical's Vildagle Tab 50mg (vildagliptin hydrochloride) and Kyongbo Pharm's Vilda Tab 50mg (vildagliptin nitrate), both DPP-4 inhibitor diabetes treatments, were initially set to lose their four-year price premiums next month.However, in line with the Drug Reimbursement Evaluation Committee (DREC)'s opinion on stable supply, the premium period will be extended by 1 year.Currently, Vildagle Tab receives a premium of KRW 300 from KRW 240, and Vilda Tab receives KRW 314 from KRW 240, a premium of 25%-30% at the ceiling price.These two products entered the market in 2022 through salt modifications, before the expiration of the Novartis patent on its original drug, Galvus (vildagliptin).While prices typically drop following patent expiration and the entry of generics, these salt-modified products successfully defended their pricing. By continuously raising their price premiums, they are now priced above the KRW 240 ceiling price of the original Galvus.Additionally, the ceiling price for Bukwang Pharmaceutical's Antiroid Tab (propylthiouracil), an antithyroid agent designated as a 'discontinuation-prevention drug', will increase by approximately 11% next month, from KRW 34 to KRW 38.Price compensation for 'discontinuation-prevention drug' is granted to medicines that are essential for clinical treatment but are at risk of production stoppage due to low profitability.As an ingredient designated as a 'National Essential Drug', it has faced concerns about supply instability amid rising raw material costs, given its low drug pricing. The 11% price hike for Antiroid Tab will take effect on the 1st of next month.Meanwhile, the products undergoing price reductions next month total around 4,000 items, including 3,940 drugs whose prices are being adjusted following the government's Actual Transaction Price investigation.
- Policy
- Celltrion has added 'Omlyclo' Pen Inj…rival Xolair
- by Lee, Tak-Sun Dec 23, 2025 07:59am
- Celltrion's 'Omlyclo'Celltrion has started chasing the original product in the Korean market through the expanded formulation of 'Omlyclo (omalizumab)', used to treat allergic asthma and chronic spontaneous urticaria.The original drug is Novartis' Xolair, an injectable generating KRW 20 billion in the domestic market (KRW 21.1 billion according to the 2023 IQVIA). Omlyclo is a biosimilar to Xolair, and it was added to the reimbursement list in September of last year.The Ministry of Food and Drug Safety (MFDS) approved Omlyclo Pen Inj in September. This product is a pen formulation, a different formulation from Omlyclo PFS Inj approved in June of last year.The difference between a pre-filled pen and a pre-filled syringe is whether the needle tip is exposed. A pre-filled syringe is injected with a needle exposed, while a pre-filled pen may reduce fear due to its hidden needle tip. Both formulations can be self-injected.The original drug Xolair has no pen formulation approved in South Korea. Only the conventional injectable and pre-filled syringe formulations are available.Considering these factors, Celltrion may be one step ahead in the future competition against Xolair.When it comes to prices, Celltrion has a competitive edge. Xolair is priced at KRW 216,755 per 150mg. In contrast, Omlyclo costs KRW 173,404, approximately KRW 40,000 less.However, Omlyclo 300mg of higher strength has not yet been added to the reimbursement list, thereby constituting the current weakness.Product sales overseas are also at full scale. In September, it was launched in Europe. In Europe, Omlyclo is reportedly the sole biosimilar to omalizumab.Omlyclo obtained U.S. FDA approval in March, thereby beginning to target the North American market on a full-scale basis.However, due to the patent expiration of Xolair in November, other biosimilars, such as one from Teva Pharmaceutical, are about to enter the market. Celltrion taking the market lead in the early race is expected to drive sustained sales.Xolair's global sales amounted to approximately KRW 5 trillion as of 2023.
- Policy
- Moderna’s RSV vaccine gains expedited approval
- by Lee, Tak-Sun Dec 19, 2025 09:08am
- The Ministry of Food and Drug Safety (MFDS) announced on the 18th that it has approved mRESVIA Prefilled Syringe (respiratory syncytial virus [RSV] mRNA vaccine, Moderna Korea) as the first biologic drug to be authorized under the newly implemented ‘New Drug Marketing Authorization and Review Procedures’ implemented this year.The revised New Drug Marketing Authorization and Review Procedures, introduced earlier this year, include expedited review measures such as the formation of dedicated product review teams and prioritizing GMP reviews. These measures were established as follow-up actions to the increase in new drug approval review fees.For the approval of mRESVIA, the MFDS formed a dedicated review team consisting of 18 members, including specialists in new drug evaluation, conducted priority GMP reviews, and held individualized face-to-face meetings before and after the marketing authorization application. Through close communication with the applicant, the MFDS was able to complete the approval process in an expedited manner.The newly approved imported drug, mRESVIA Prefilled Syringe, is indicated for the prevention of lower respiratory tract disease (LRTD) caused by RSV in adults aged 60 years and older, as well as in high-risk individuals aged 18 to under 60 years. It is the first RSV preventive vaccine in Korea approved using an mRNA platform.Respiratory syncytial virus (RSV) is a virus that causes acute respiratory infections with symptoms similar to those of the common cold.Meanwhile, the first small-molecule drug approved under the new fast-track review system was Xcopri Tab (cenobamate) developed by Dong-A ST, which received marketing authorization in November.The MFDS stated that it plans to further enhance the new drug review system through in-depth preliminary assessments, item-by-item parallel reviews, and stage-specific tailored meetings during the new drug approval process. These efforts aim to facilitate rapid market entry of innovative medicines, provide patients with quicker access to treatment options, and support the growth of the biopharmaceutical industry.