- LOGIN
- MemberShip
- 2026-05-05 07:23:21
- Policy
- To establish a pharmaceutical bio-innovation committee
- by Lee, Jeong-Hwan Mar 03, 2022 06:00am
- Yoon Suk-youl, presidential candidate for the presidential election, pledged to establish a "pharmaceutical bio-innovation committee" directly under the Prime Minister, while creating an ecosystem that can establish sovereignty in pharmaceutical bio and foster key talents and jobs in the pharmaceutical bio industry. It is said that it will overhaul the COVID-19 response system within 100 days immediately after taking office and implement the national responsibility system for vaccine side effects. The pledge also included strengthening access to medicines for patients by diversifying the rapid benefit registration system for drugs with severe rare diseases such as ultra-high-priced anticancer drugs compared to the previous one. On the 24th, Candidate Yoon Suk-youl announced a policy pledge for the power of the people in the 20th presidential election. Candidate Yoon said he would use the health care and pharmaceutical bio-industry as a new path to establishing health security and creating national wealth. He specified in his pledge the establishment of a "pharmaceutical bio-innovation committee" directly under the Prime Minister in line with the establishment of a pharmaceutical bio-control tower directly under the Blue House demanded by the KRPIA. In addition, the vision is to support national R&D to establish vaccine sovereignty and global vaccine hubs and to establish an environment to expand talent and jobs in the pharmaceutical bio industry. ◆ Reorganization of the Infectious Disease Response System = Yoon said the current government failed to respond to COVID-19. They say that they have failed to establish a proper treatment system while causing unnecessary economic damage and prolonging damage to small business owners and self-employed people by keeping distance without principles. In response, candidate Yoon plans to completely reorganize the COVID-19 response system within 100 days. The pledges include scientific and data-based quarantine measures, reorganization of the manual for responding to new infectious diseases, and conversion of public medical institutions to hospitals dedicated to COVID-19. It also declared that it will open the era of biohealth and become a vaccine and treatment powerhouse. He criticized that six domestic vaccines are currently undergoing clinical trials, but have nothing to do with government support. It is pointed out that the SK Bioscience vaccine, which is in phase 3 of clinical trials, is also being developed with technical cooperation and the CEPI support at the University of Washington in the United States. He promised to expand the overall national R&D, including high-speed vaccine development and manufacturing technology, post-corona vaccines and treatments, essential vaccines, and digital quarantine. The government's responsibility system for essential medical care and the national responsibility system for side effects of COVID-19 vaccination were included in the pledge. Under the public policy fee system, policy fees added compared to usual times are paid to doctors and nurses in emergency situations such as COVID-19, and public policy fees are also paid to severe trauma centers, delivery rooms, and newborn rooms. ◆ National Health Protection = Candidate Yoon also pledged to expand support for disaster medical expenses, ease the burden of national nursing expenses, and introduce a rapid registration system for expensive anticancer drugs and new drugs for severe and rare diseases. Among them, the rapid registration system of expensive anticancer drugs and new drugs for severe and rare diseases is a policy proposal that the KRPIA strongly appealed to when it met with party leader Lee Joon-seok. Candidate Yoon said that it takes about two years to pay health insurance benefits for new drugs with severe rare diseases such as cancer, and patients are paying high prices and administering new drugs. RSA also pointed out that it was applied only to 41 drugs, and 32 of them were concentrated on anticancer drugs. Candidate Yoon promised to shorten the health insurance registration process for anticancer drugs and treatments for severe rare diseases. It also said it will use RSA to reduce drug price negotiations and the burden on patients and insurers. In order to secure insufficient emergency and essential medical care and medical personnel in the region, the government will strengthen the publicity of local national university hospitals and upper-level general hospitals and expand them to cities and provinces without upper-level general hospitals.
- Policy
- Negotiations with NHIS on Actemra have been completed
- by Lee, Tak-Sun Mar 02, 2022 05:54am
- Actemra (Tocilizumab), a treatment for rheumatoid arthritis by JW Pharma, has been officially recognized as a treatment for COVID-19 in Korea. The drug, which has been used in patients with severe COVID-19 for purposes other than permission, will also be covered by health insurance benefits from March. On the 24th, the MOHW announced that Actemra is preparing to revise the regulations so that benefits can be applied as a treatment for COVID-19. Earlier, the NHIS negotiated with JW Pharma, a domestic importer and seller, to apply benefits. Through this, it is possible to apply benefits to severely ill patients with COVID-19. According to the revision of the MOHW, medical care benefits are recognized when administered to COVID-19 patients aged 2 or older beyond the scope of the existing permits of Actemra (excluding SC injections). "If a patient is admitted to an intensive care unit or a hospital room within 48 hours and needs respiratory treatment of HFNC (High Flow Nasal Canula) or higher," alternatively, it corresponds to "the case of administration to patients who rapidly deteriorate to a state in which respiratory treatment above High Flow Nasal Cannula (HFNC) is required even though they have been treated with steroid therapy and low flow oxygen therapy." Full support is provided through health insurance without the patient's copayment. Actemra, unlike in Korea, is already used as a treatment for COVID-19 abroad. The U.S. FDA decided on Actemra as a treatment for COVID-19 in June last year. The European Commission also approved it as a treatment for COVID-19 in December last year. Recently, the WHO also added Actemra to the Pre-qualification Review List for COVID-19 Treatment for reference by middle and low-income countries that have difficulty in independent drug screening. It has been used as an offline in Korea, but the problem is that the patient had to pay the full price of the drug because it was not paid. Other COVID-19 treatments approved in Korea are purchased entirely by the state and have no Pt's payment. Therefore, the medical community also requested the purchase of Actemra to the state in terms of equity. Actemra achieved 20 billion won in domestic sales last year, up 24.9% year-on-year. Currently, Actemra 80mg's price is 134,263 won, Actemra 200mg's price is 306,166 won, and Actemra 400mg's price is 537,060 won, while SC (subcutaneous injection) product is 345,682 won. Until now, patients have paid all the drug prices, but if health insurance benefits are applied, the patient will not be burdened with the drug prices. Actemra is also pushing for a change in permission to expand manufacturing sources limited to Japan due to the recent shortage of supply. It is also known that EUA plans as a treatment for COVID-19 are being considered. Meanwhile, JW Pharma also announced on the 25th that Actemra's benefit range will be expanded for the purpose of treating COVID-19 (COVID-19) in accordance with the revision announced by the MOHW. The company explained that the new revision, which will take effect on March 1, has changed its standards to reflect the status of overseas permits such as FDA EUA, clinical research literature, and related academic opinions. An official from JW Pharma said, "Considering the recent surge in confirmed cases of Omikron mutant virus, we have been closely discussing with health authorities to expand the benefits of Actemra. The EUA is also quickly proceeding with the MFDS." Actemra is an antibody treatment that treats diseases such as rheumatoid arthritis and pediatric idiopathic arthritis by inhibiting the binding of IL-6 and its receptors, a protein that causes inflammation in the body. It has been shown that it effectively lowers the mortality rate of severely ill and critically ill patients with COVID-19 and reduces hospitalization time in global clinical trials, a complication of excessive immune responses. JW Pharma acquired Actemra's domestic development and exclusive sales authority from Chugai under Roche Group in 2009 and has since been selling it since 2013 through phase 3 clinical trials for rheumatoid arthritis patients.
- Policy
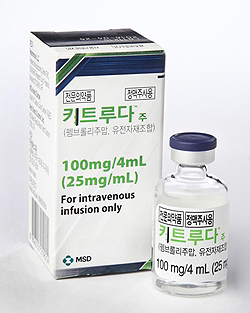
- Keytruda is reimbursed as first-line at ₩2,107,642
- by Kim, Jung-Ju Feb 28, 2022 05:54am
- The insurance price of MDS Korea’s immuno-oncology drug Keytruda(pembrolizumab) inj. that is used for non-small-cell lung cancer and Hodgkin lymphoma will drop 25.6% with its reimbursement extended from the second-line to the first-line starting next month, Astellas Korea’s Xospata 40mg (gilteritinib), as well as Novartis Korea’s Lutathera Inj. that was supplied in Korea as an urgently requested drug will also be individually listed for reimbursement as an expenditure-cap type RSA after reaching an agreement with the National Health Insurance Service. The Ministry of Health and Welfare announced on the afternoon of the 25th that it had deliberated and passed the ‘amendment to the drug benefit list and the maximum ceiling price table (draft)’ that contains the changes above as an agenda at the 5th Health Insurance Policy Deliberation Committee. ◆Keytruda(pembrolizumab) = The immuno-oncology drug Keytruda, which is indicated for the treatment of non-small-cell lung cancer and Hodgkin lymphoma, was listed as second-line treatment in August 2017, and then received an indication extension in February 2018. The company had applied for the reimbursement expansion to the Health Insurance Review and Assessment Service in October 2019 and received deliberation from the Cancer Disease Deliberation Committee on April, August, November 2020, and on May 7th last year. In January of this year, HIRA’s Pharmaceutical Reimbursement Evaluation Committee deliberated and passed Keytruda’s reimbursement expansion agenda. At the time, the PBAC judged that the ICER threshold (economic evaluation result) of Keytruda in NSCLC as monotherapy and as combination therapy in squamous and non-squamous NSCLC was appropriate, and that it was cost-effective as it costs less than its alternative brentuximab vedotin in Hodgkin's lymphoma. Also, the committee considered that the drug was listed for reimbursement in all A7 countries. The company afterward reached a final agreement with the NHIS under the Ministry of Health and Welfare’s negotiation order on the RSA drug price and expected claims amount by mid-February. In the drug pricing negotiations, the company made a refund-type RSA agreement in which the company refunds a specific proportion of the claims amount and a proportion of the amount that exceeds the expected claims cap to the government. Both parties have made an agreement to set the price at ₩2,107,642, a 25.6% discount from the current cap of ₩2,833,278 in consideration of the expanded scope of use, the standard for price cap adjustment, and its fiscal impact on insurance finances. With the refund-type RSA applied, the NHIS expects the actual fiscal spending on the drug to be less than the expected claims amount.. ◆Xospata Tab40mg (gilteritinib fumarate) = Xospata, which was approved for the treatment of acute myeloid leukemia, applied for reimbursement listing in November 2020 after receiving marketing authorization in March 2020. After deliberation by CDDC in February and PBAC in September, HIRA judged that reimbursement was appropriate for the drug. At the time, HIRA considered the fact that the drug is recommended in adult patients with FLT3-positive AML in clinical guidelines and that its cost-effectiveness also meets the pharmacoeconomic evaluation data exemption drug (PE exemption drug) conditions, and is listed in 5 of the 7 A7 countries – the US, UK, Germany, Italy, and Japan. Relevant societies have also judged that the drug improved convenience in intake as an oral formulation and may become a new treatment alternative for the disease. After passing HIRA review, the company completed drug pricing negotiations with the NHIS as a refund-type RSA. In consideration of its price abroad and fiscal impact, the two parties agreed to set the price at ₩214,100 per vial. ◆Lutathera inj.(lutetium Lu 177 dotatate) = Novartis Korea’s Lutathera Inj is a radiolabeled somatostatin analog indicated for the treatment of gastroenteropancreatic neuroendocrine tumors. The drug, which was approved in July 2020, was designated an urgently requested drug when it was first supplied in November 2019 without reimbursement and reimbursed from March 2020 ever since. In November 2020, the company applied for its formal reimbursement listing to HIRA, and the authorities deliberated the case with the CDDC in April 2021 and with DREC in November of the same year. At the time, HIRA considered the fact that the drug was recommended for the treatment of patients whose condition progressed after the current standard-of-care sandostatin lar (octreotide) in clinical guidelines (clinical efficacy aspect) and that the drug met the PE exemption criteria by applying at a price lower than the lowest A7 price(cost-effectiveness aspect), and that the drug is listed in 4 of the 7 A7 countries – the US, France, Japan, and Italy. After passing HIRA review, the company completed drug pricing negotiations with the NHIS as a refund-type RSA at ₩22,104,660 per vial.
- Policy
- BeiGene's Brukinsa has been approved in Korea
- by Lee, Hye-Kyung Feb 28, 2022 05:54am
- Brukininsa 80mg (Zanubrutinib), a BTK inhibitor from Chinese pharmaceutical company BeiGene, has obtained an item license in Korea. It is the second new drug approved by a Chinese pharmaceutical company after Antengene's Xpovio 20mg (Selinexor) in July last year. On the 24th, the MFDS approved Brukinsa, a new drug for treating blood cancer in BeiGene. This drug was recognized for its efficacy and effectiveness▲ in monotherapy in adult patients with mantle cell lymphoma (MCL) who had received more than one treatment before and▲ monotherapy in WM adult patients who had received more than one treatment before. Brukinsa is a Brutons Tyrosine Kinase (BTK) inhibitor that inhibits the survival and spread of malignant B cells by blocking BTK protein, a signaling molecule that affects the survival and development of B cells. Among the drugs licensed in Korea as BTK inhibitors are Janssen's Imbruvica 140mg, AstraZeneca's Calquence 100mg, and Ono's Velexbru 80mg. Brukinsa was quickly approved by the U.S. Food and Drug Administration (FDA) as an MCL treatment in November 2019. At that time, it became famous as the first new drug to be approved by the FDA based on clinical trials in China. It then expanded its indications in September last year when it was approved by the FDA as a treatment for WM patients. Clinical trials are currently underway for CLL, MZL, and FL patients. A new drug approved by a Chinese pharmaceutical company in Korea was first released in July last year. Antengene's Xpovio 20mg (Selinexor) has been approved in Korea for anti-cancer drug which is administered to treat refractory multiple myeloma and refractory giant B-cell lymphoma.
- Policy
- President Moon said, "WHO selects Korea as a vaccine hub"
- by Kang, Shin-Kook Feb 28, 2022 05:54am
- President Moon Jae In evaluated that the government's efforts to become a vaccine hub country have paid off, saying that Korea has been exclusively selected as the "WHO Global Bio Human Resources Development Hub." President Moon posted on SNS on the 24th and said, "This is the result of being internationally recognized for Korea's biopharmaceutical production capacity and excellence in educational infrastructure." He said, "It is the evaluation of the international community that the Korean government is taking the lead in overcoming the World Health crisis by strengthening vaccine cooperation with various countries and organizations, including Hanmi global vaccine partnership." President Moon said, "Korea has become a hub country for education and training and training to increase vaccine capabilities of developing countries. We will actively share our experiences and know-how with the international community, self-sufficiency of vaccines, and resolve vaccine inequality." "It's another opportunity for us. The world's top five vaccine powers and bio-leading countries set by the Korean government are coming to reality, he said. "It will provide young people with opportunities to form world-class education and human networks, and increase the awareness and reliability of our bio companies to help hub overseas exports and vaccine production." President Moon said, "South Korea is successful based on innovation capabilities and human investment," adding, "It is constantly advancing from a country that receives aid, from developing countries to developed countries, from pursuing countries to leading countries." The WHO announced on the 23rd that it has exclusively selected South Korea as "the WHO Global Bio Human Resources Development Hub." WHO Human Resources Development Hub refers to a central institution that provides education and training for vaccine and biopharmaceutical production processes to self-sufficiency vaccines in middle and low-income countries. In response, Minister of Health and Welfare Kwon Deok-cheol said, "The fact that Korea was designated as a hub for fostering WHO manpower is to expect leadership for global health and safety by overcoming COVID-19 and responding to the next pandemic and utilizing domestic capabilities." In response to expectations, we will develop Korea into a global bio-human resource training mecca in the future, he said.
- Policy
- Voluntary price cuts of originals as ‘trade-offs’ an issue
- by Lee, Tak-Sun Feb 25, 2022 05:57am
- ‘Discounting the price of existing drugs to list high-priced new drugs.’ Recently, the pharmaceutical industry has been buzzing with speculations that some companies are voluntarily reducing the price of their original drugs as a fiscal sharing strategy to receive reimbursement. This strategy, which was considered unimaginable before, is raising concern among generic companies, as the price drop of an original drug will threaten the price competitiveness of its generic items. According to the industry on the 24th, prices of 15 MSD Korea’s Januvia products and 2 Astellas Korea’s Vesicare products will be discounted from the 1st of next month. All the pricing decisions were voluntarily made by the company. The price caps of the products will fall from 3.2% to 77% at the most. The industry speculates that the voluntary price cuts are the results of negotiating with the insurance authorities to list their high-priced drugs for reimbursement, as a kind of "trade-off" strategy. In other words, the analysis is that MSD made a “trade-off” to reimburse ‘Keytruda’ as first-line treatment of NSCLC, which will cost at least 170 billion won in insurance finances, and Astellas to reimburse its acute myeloid leukemia treatment ‘Xostapa.’ However, the parties involved are refraining from making official statements regarding the matter. However still, the strategy will ultimately be a ‘win-win’ for both the insurance authorities and pharmaceutical companies, as it would save NHI finances and allow companies to list high-price drugs. But this was not good news for the generic companies. Due to the voluntary price cut of Vesicare, the ceiling price of 75 generics became 16-18 won more expensive than Vesicare. Only 2 generic products will have a lower price than Vesicare. Some hospitals and clinics prescribe products with a lower price cap among the same ingredient drugs in consideration of the patient’s economic status, and generic companies will have lost this price competitiveness due to the voluntary discounts. Moreover, as the well-known originals became cheaper than the less known generics, the generic companies are now more disadvantaged than before. Generic versions of MSD Korea’s Januvia tab. And Janumet tab. may be released upon patent expiry in September 2023. However, the voluntary price cut of the original products has lowered the list price of its generics, increasing the cost burden. Due to such reasons, the industry is seeing that there is a problem in companies voluntarily lowering drug prices of patented drugs for the ‘trade-offs’. A domestic industry official said, “The trade-offs were made in agreement between the insurance authorities and the pharmaceutical companies, but it is unreasonable that this may affect the profitability of other drugs that will be released in the future. The subjects of the trade-offs should be limited to off-patent drugs at the very least.”
- Policy
- JW’s ‘Actemra’ reviewed for reimbursement as COVID-19 Tx
- by Lee, Tak-Sun Feb 25, 2022 05:57am
- The government is reviewing providing nsurance benefits to JW Pharmaceutical’s rheumatoid arthritis treatment ‘Actemra’ (tocilizumab)’ for the treatment of COVID-19. This is because the drug, which is being often used in Korea off-label to treat patients with severe COVID-19, is increasing patient burden due to its non-reimbursed cost unlike other insured COVID-19 treatments like Remdesivir or Regkirona. Also, due to its shortage in supply, the medical community has asked the government to purchase Actemra like other COVID-19 treatments. According to the NHIS webpage on the 23rd, the authorities have conducted negotiations with JW Pharmaceutical on providing health insurance coverage for ‘Actemra.’ The negotiation has been conducted to lower the burden borne by patients by reimbursing ‘Actemra,’ which is currently widely used in patients with severe COVID-19. However, the negotiation results have not been disclosed yet. An NHIS official said, “the NHIS had conducted negotiations for Actemra recently, but did not share details of the negotiation or its results. The industry believes that discussions on providing insurance support would have been conducted for Actemra for equity with other COVID-19 treatments as the drug is being widely used off-label for patients with severe COVID-19 without reimbursement. Off-label prescriptions of Actemra has increased for the treatment of severe cases of COVID-19 last year, to make YoY growth of 24.9% and record ₩20 billion in sales last year Other COVID-19 treatments such as Remdesivir, Regkirona, and Paxlovid have been fully purchased by the Korea Disease Control and Prevention Agency and are provided free for the patients’ use. However, in the case of Actemra, as the drug was not approved as a COVID-19 treatment, the cost needs to be solely borne by the patient. Actemra, which is used for the treatment of rheumatoid arthritis in adults, costs ₩345,682 per shot without reimbursement, bringing an immense burden on the patients. Regarding the matter, the Korean Society of Infectious Diseases and other medical societies have submitted an opinion to the government to ask for the use of Actemra as a treatment for severe COVID-19. The industry believes it is highly likely that the government would temporarily apply insurance benefits to Actemra until the drug is approved as a COVID-19 treatment in Korea. Actemra is already being used with temporary or official approval as a COVID-19 treatment abroad. The US FDA granted Actemra Emergency Use Authorization (EUA) as a treatment for COVID-19. The European Commission also approved the drug as a treatment for COVID-19 in December last year. The WHO had also recently added Actemra to the prequalified COVID-19 treatment list' for reference by low- and middle-income countries that may have difficulty conducting independent reviews. Recently, the community has been suffering a supply shortage of Actemra with its increased use in patients with severe COVID-19. Due to this, the company is working to change its license to expand manufacturers of the drug that is currently limited to Japan. Also, the authorities are known to be reviewing Actemra for emergency use in treating COVID-19.
- Policy
- Pfizer COVID-19 vaccine for children has been approved
- by Lee, Hye-Kyung Feb 25, 2022 05:56am
- Pfizer's COVID-19 vaccine "Cominatiju 0.1mg/mL," which is subject to vaccination for children and adolescents aged 5 to 11, has obtained an item license. The MFDS (Minister Kim Kang-rip) announced on the 23rd that Pfizer Pharmaceutical Korea has approved "Comirati 0.1mg/mL" applied for permission as an imported item. Depending on the design of the clinical trial, different doses were allocated at the age of 11 and 12, and in the case of 5 to 11 years old, the dose was selected in consideration of the results of the immune response obtained from the clinical trial (phase 1). The vaccine is an mRNA vaccine jointly developed by Pfizera of the U.S. and Bioentech of Germany and imported by Pfizer Pharmaceutical of Korea, which will be diluted with 0.9% sodium chloride injection. According to clinical data submitted by pharmaceutical companies, the overall safety information was similar to those aged 16 to 25 (1064 people) when comparing safety and evaluating 3,109 people aged 5 to 11. The most frequent abnormal cases after vaccination were pain at the injection site, fatigue, headache, redness and swelling at the injection site, muscle pain, and chills, and most of the symptoms were mild to moderate. The probability of redness and swelling at the injection site was 26.4% and 20.%, respectively, higher than 10.3% and 11.4% seen at the age of 16-25 years old. The abnormal cases appeared more in the second inoculation than in the first inoculation, but were reported to have disappeared within three days of the outbreak. There were no deaths, myocarditis, pericarditis, or anaphylaxis, and it was confirmed that no serious adverse drug reactions such as drug-related hospitalization or death occurred. COVID-19 immune responses were compared and evaluated at the ages of 5 to 11 (264) and 16 to 25 (253), and the preventive effect was evaluated as the proportion of patients infected with COVID-19 after administering vaccines (1305 patients) and contrast drugs (663 patients) at the ages of 5 to 11. As a result of comparing the immune responses of ages 5 to 11 and 16 to 25 at one month after the completion of the second inoculation, it was found that both the ratio of neutralizing antibodies and the serum response rate were proven effective. As a result of analyzing the preventive effect seven days after the completion of the second inoculation of 1968 people (1305 in the test group and 663 in the control group) who were not infected with COVID-19, the preventive effect of vaccination was 90.7%. Based on this data, experts agreed that the safety and effectiveness confirmed in clinical trial data can be recognized as a result of consultation by the Central Pharmaceutical Review Committee on the 14th. However, although it did not occur in clinical trials as a risk management plan after approval, it was decided to observe the safety of myocarditis and pericarditis as a preventive measure, and to collect and evaluate abnormal cases arising from clinical trials and post-authorization use. Comernaty 0.1mg/mL (for ages 5 to 11) is a vaccine developed and produced separately by Pfizer of the United States for the purpose of preventing COVID-19 aged 5 to 11, and has the same type of additive, usage, and capacity (tojinameran) as previously approved by the Ministry of Food and Drug Safety. The specific vaccination plan will be announced separately by the Korea Centers for Disease Control and Prevention, taking into account the burden of children's diseases (severe rate, etc.), the epidemic of Omicron mutations, and the schedule of introduction of vaccines for children in Korea. In the future, the Ministry of Food and Drug Safety plans to analyze the manufacturing and test results of manufacturers for each manufacturing unit of Comirati 0.1 mg/mL (5-11 years old) and check the quality once again with national shipping approval.
- Policy
- Enactment of the Digital Healthcare Act begins in earnest
- by Lee, Jeong-Hwan Feb 24, 2022 05:59am
- With the National Assembly moving to enact a law specifically for the digital healthcare industry, attention is focused on whether the non-face-to-face treatment and drug delivery platform may gain momentum and make growth. The non-face-to-face and drug delivery platforms have been operating based on the non-face-to-face treatment that was temporarily allowed due to the COVID-19 pandemic and lacks specific legal and institutional grounds or support. If a law is enacted to foster the digital healthcare industry, the law and institutional support for the industry will be improved while the industry may enjoy benefits from the new various special provisions that would be set. NA member Tae-ho Jung of the Democratic Party of Korea recently submitted a bill as representative for the ‘Foster and Support of the Digital Healthcare Industry.’ The law aims to foster and support the digital healthcare industry and stipulate the basic matters necessary for development to strengthen industry competitiveness. The bill defines the Digital Healthcare Industry, subjects for support, and also contains provisions that require a comprehensive plan for foster and support be established every 5 years. Also, the bill will provide the basis for support such as certification, preferential treatment, tax exemptions, and prepurchase approvals for best companies while also providing support for training and employment of professionals and the establishment of a Digital Healthcare Business Association. The healthcare industry has started collecting opinions related to the proposed enactment of the bill. The Korean Hospital Association is collecting opinions from its member countries, and groups related to digital healthcare are expressing their agreement to the enactment of the bill. 7 associations including the Korea Medical Devices Industry Association, Korea Digital Health Industry Association, and the Korea Medical Device Industry Cooperative Association have issued a joint statement welcoming the bill. Digital healthcare is essential for responding to the post-COVID-19, data economy, and non-face-to-face society. In particular, if enacted, the bill will directly influence the management of the non-face-to-face treatment and drug delivery platform. Companies that have jumped into telemedicine and prescription drug delivery service due to COVID-19 have made remarkable growth despite the temporarily approved environment. If the regulations are lifted, industry analysis is that the telemedicine and prescription drug delivery platforms will grow explosively. The enactment of the Digital Healthcare Act is being evaluated as the trigger that will speed up such regulatory reform. In addition, there is a high possibility that dinosaur companies such as Naver and Kakao will enter the non-face-to-face treatment and drug delivery business in addition to IT venture companies. Tae-ho Jung, who submitted the bill said, “The COVID-19 pandemic has rapidly changed our living environment, and the limited amount of medical resources have been greatly affecting public health and the industry in general. It is imperative that we develop the digital healthcare technology to solve chronic problems such as aging and the upsurge of chronic disease patients."
- Policy
- Phase 3 of SK Biopharm's Cenobamate is approved
- by Lee, Hye-Kyung Feb 24, 2022 05:58am
- Clinical trials have been approved at six hospitals, including Seoul National University, for children with partial epilepsy attacks. SK Biopharm will enter phase 3 of clinical trials for pediatric test takers of Xcopri (Cenobamate), a treatment for epilepsy. On the 23rd, the MFDS approved a phase 3 clinical trial plan for Cenobamate (YKP3089) for pediatric testing patients with partial expression attacks submitted by SK Biopharm. This clinical trial will be conducted at six locations, including Yonsei Severance Hospital, Chungbuk National University Hospital, Korea University Guro Hospital, SNU Boramae medical center, Ajou University Hospital, and Seoul National University Hospital. Cenobamate is SK Biopharmate's new drug released in the U.S. in May 2020 after being approved by the U.S. Food and Drug Administration (FDA) in November 2019 for epilepsy partial seizures. Phase 3 clinical trials in the U.S. for adults aged 18 or older with systemic seizure symptoms began in September 2018, and clinical trials in China, Japan, and Korea for adults aged 18 or older were preparing to launch in the U.S., Europe and other Asian regions as patient registration began in March last year. It is known that the registration of phase 3 clinical trials in the United States has recently begun for children and adolescents aged 2 to 17 with partial epilepsy attacks, and Korea will enter phase 3 clinical trials at the same time. SK Biopharm announced on February 8 that its operating profit last year turned into a surplus year-on-year at 95.3 billion won. It recorded a surplus for the first time in five years due to the influx of new drug technology fees, and Cenobamate played a significant role. When Cenobamate transferred its technology to Angelini Pharma (formerly Arvelle Therapeutics), it received $123.22 million worth of technology fees last year. In addition, Cenobamate, which has been released in the U.S. since last year, has generated 78.2 billion won in sales, reflecting its operating profit as it is.