- LOGIN
- MemberShip
- 2026-05-04 12:09:25
- Policy
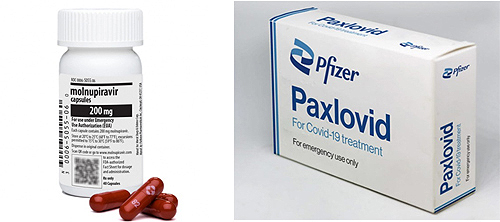
- New guidelines for Lagevrio & Paxlovid have been released
- by Kim, Jung-Ju Jun 02, 2022 05:58am
- (from left) Lagevrio and PaxlovidNew clinical guidelines for Lagevrio and Paxlovid's administration of COVID-19 patients have been released. Lagevrio considers medication if it is difficult to use other treatments among confirmed patients with mild and severe symptoms over the age of 18, and Paxlovid considers patients aged 12 or older who weigh more than 40kg. Eight member societies of the National Evidence-based Healthcare Collaborating Agency (Director Han Kwang-hyeop) and the Korean Academy of Medical Sciences (Chairman Jung Ji-tae) announced additional clinical treatment guidelines for the diagnosis and treatment of COVID-19. This guideline reflects the results of a comprehensive review of papers published around the world with the participation of the National Evidence-based Healthcare Collaborating Agency, a specialized research institute under the MOHW, and the COVID-19 diagnosis and treatment. The eight member societies are the Korean Academy of Medical Sciences, the KSID, the Korean Academy of Tuberculosis and Respiratory Diseases, the KSPID, the Korean Society of Radiology, the KSEM, the Korean Society of Critical Care Medicine, the KSLM, and the KSCM. The National Evidence-based Healthcare Collaborating Agency explained that COVID-19 new mutations are continuously occurring and that the administration of oral treatments has been clinically safe and effective in preparation for the re-pandemic this fall. Looking at the details, first of all, Lagevrio has a risk factor for severity among mild or moderate COVID-19 confirmed patients aged 18 or older, but if it is difficult to use other COVID-19 treatments, medication is considered and recommended to be used within 5 days after symptoms occur. The risk factor refers to immunodeficiency or underlying disease patients known to have a high risk of severe COVID-19 progression or low vaccination effect, and if it is difficult to use other treatments, it refers to cases where monoclonal antibodies, including Paxlovid and Remdesivir, are difficult to use. In the case of Paxlovid, medication is considered for COVID-19 infections aged 12 or older with a mild or moderate weight of 40Kg or more, and it is recommended to use it within 5 days after symptoms occur. This clinical treatment guideline was developed by applying the GRADE methodology. The National Evidence-based Healthcare Collaborating Agency searches and reviews the latest evidence every month, and if it is deemed necessary to revise the recommendations quickly, it is revised and announced every three to four months after discussion by the Committee of Medical Guidelines composed of representatives of the Association. Anyone can download this recommendation from www.neca.re.kr, Research Information >> COVID-19 Living Guideline. Han Kwang-hyeop said, "The number of COVID-19 confirmed cases is gradually decreasing, but COVID-19 new mutations continue to appear, so it is important to prepare through scientific evidence." He said, "In the future, the National Evidence-based Healthcare Collaborating Agency plans to continue to develop clinical care guidelines based on the latest evidence in cooperation with the government and the medical community."
- Policy
- The GMP reinforcement bill passed the plenary session
- by Lee, Jeong-Hwan Jun 02, 2022 05:58am
- Sustainable Support for New Drug Development Pharmaceutical Companies Increasing Punishment for Violations of GMP A bill to extend special cases for innovative pharmaceutical companies and a bill to significantly strengthen the level of GMP regulatory management compared to the previous one passed the plenary session of the National Assembly on the night of the 29th. With the revision of the Special Act on the Promotion and Support of the Pharmaceutical Industry, the deadline for special provisions on the construction of research facilities for innovative pharmaceutical companies and exemption from various levies will be extended to March 30, 2032. It is expected that it will continue to support innovative pharmaceutical companies that lead the development of new drugs in the future and boost investment in the biohealth industry. The level of regulation will increase as the National Assembly passes the revision of the Pharmaceutical Affairs Act, which promotes the improvement of the quality of domestic distributed drugs by strengthening the standards for drug manufacturing and quality management. The standards for drug manufacturing and quality management are strengthened to secure the effectiveness of the system and strengthen the international competitiveness of products, ▲ upgraded the basis for determining compliance with drug manufacturing and quality control standards (GMPs) to the law, ▲ establishment of sanctions standards for serious violations, such as being judged to be suitable for GMP by false or illegal means, ▲ appointment of GMP investigators and grounds for entry, and ▲preparation of the basis for publication of the details of disposition to drug-related business operators whose administrative disposition has been confirmed. Currently, in order to manufacture and sell medicines, the MFDS stipulates that they are suitable for GMP by formulation or manufacturing method, but it was raised to the law to strengthen the management of serious violations related to GMP. Sanctions standards have been established for serious violations related to GMP-related violations, punitive fines, and penalties of up to 5 years or 50 million won. In order to increase the administrative efficiency of GMP investigation and evaluation work, GMP education and training were appointed as manufacturing and quality control investigators for medicines, etc., and provided a basis for entry and investigation. The head of the MFDS has prepared a basis for informing the public of the violation and the details of the disposition to those whose administrative disposition has been confirmed.
- Policy
- 22nd new homegrown drug Acelex adds side effect after PMS
- by Lee, Hye-Kyung May 31, 2022 06:06am
- Crystal Genomics’ ‘Acelex Tab (polmacoxib)’ that was approved as the 22nd homegrown new drug in Korea will be adding ‘strokes, etc’ to its list of adverse events. The Ministry of Food and Drug Safety announced that it had completed the opinion review for the Change in Acelex's Indications based on Reevaluation Results (draft) and will be reflecting the changes in the indication from August 26th. Acelex was approved on February 5th, 2015 as an anti-inflammatory analgesic to relieve the symptoms of osteoarthritis patients. The drug was also designated the 22nd new homegrown drug to receive marketing authorization in Korea. The drug has been supplied to large general hospitals and university hospitals through Dong-A ST since September 2015, and Crystal Genomics also signed a joint marketing agreement for its sale with Daewoong Pharmaceuticals in March 2018. The post-marketing surveillance on Acelex was conducted for 6 years on 3,289 patients for the MFDS reevaluation in Korea. The PMS results showed that the incidence of adverse events regardless of their causality was 7.66% (252 of 2289 patients, 285 cases). Among these adverse events, one serious adverse drug reaction whose causal relationship with the drug could not be ruled out, a stroke, was reported with an incidence rate of 0.03%. The incidence rate of unexpected drug adverse events whose causal relationship with the drug could not be ruled out was 0.43% (14 of 3289 patients, 14 cases), where rare cases of stroke, paresthesia, hypoesthesia, rash, drug rash, skin erosion, tachycardia, conjunctival congestion, gastrointestinal disorders, abdominal distension, decreased appetite, daydreams, and difficulty urinating were reported. The MFDS said “We ask relevant groups to be aware of the change and to disseminate the news to their member and non-member companies. Also, institutions involved in pre-or post-management of pharmaceuticals should use the changed order for the review of civil documents such as marketing authorization of pharmaceuticals as well as for pharmaceutical surveillance monitoring-related tasks such as post-management of labels.
- Policy
- President Visited KDCA to Strengthen Bio-Health Investment
- by Lee, Hye-Kyung May 31, 2022 06:05am
- President Yoon Seok-yeol visited the KDCA in Osong at 2 p.m. today (26th) to emphasize strengthening investment in key infrastructure in the bio-health industry. On this day, President Yoon discussed with experts how to promote scientific quarantine to prepare for the COVID-19 re-pandemic in autumn and winter at the KDCA Emergency Situation Center, and inspected the BioBank of the National Institute of Health. President Yoon said, "We should thoroughly prevent the disease so that the public does not worry. We need to establish a reliable scientific quarantine system for the people as soon as possible, he said. "As we always emphasized the importance of an expert-centered scientific quarantine system based on data, a balanced approach is needed so that people can continue their economic activities without any inconvenience." After arriving at the KDCA Emergency Center, President Yoon listened to a briefing on the response to the COVID-19 crisis and held an expert video conference attended by Baek Kyung-ran, KSID Chairman Kim Nam-joong, Gachon University Professor Chung Jae-hoon, and IVI Deputy Secretary-General Song Man-ki. At the meeting, President Yoon listened to experts' opinions after receiving a report from Commissioner Baek on the progress of upgrading the quarantine system. After the expert meeting, President Yoon moved to BioBank of the National Institute of Health. The progress of the national bio big data construction project was checked by looking around the storage where blood, cells, and DNA were stored. President Yoon said, "BioBank is a core infrastructure for the bio-health industry, and the government will reduce the burden of research and development costs on companies by increasing investment in core infrastructure in the bio-industry." He said, "The KDCA hopes to push ahead with big data construction projects so that genetic information can be used as a valuable resource for the development of biohealth industry technology, such as vaccine development, and actively improve related systems and regulations." Meanwhile, BioBank is also playing a key role in in the development of in vitro diagnostics and treatments by distributing human resources for infectious diseases held in emergency situations such as COVID-19. With the aim of starting the project in 2024 to promote precision medical care through the establishment and opening of health and medical big data, the MOHW, the Ministry of Science and ICT, the Ministry of Trade, Industry and Energy, and the Korea Centers for Disease Control. The KDCA expects to boost the growth of the bio-health industry ▲ through new technologies and bio-big data, such as analyzing and designing drug structures combining bio-big data and artificial intelligence technology, ▲in the past, as the data-based health care paradigm shifts from empirical and intuitive medical care, the development of customized disease prevention, diagnosis, and treatment technologies for each patient is activated, and ▲ improving treatment efficiency through new drug-responsive drugs tailored to patients than current standard, universal, and empirical treatments.
- Policy
- Will Dukarb Plus next month rise as Boryung’s savior?
- by Lee, Tak-Sun May 30, 2022 05:42am
- Picture from the Dukarb Plus launch symposium that was held on the 25th at Shilla Hotel Seoul A product that uses the antihypertensive ingredient fimasartan (Kanarb) that Boryung Pharmaceutical developed will be released next month. The product, Dukarb Plus, is a fixed 3-drug combination of fimasartan, amlodipine, and hydrochlorothiazide. Dukarb Plus will be listed for insurance benefit as of June 1st. The release of Dukarb Plus has been receiving attention due to the imminent expiration of the substance patent for Kanarb. Boryung Pharmaceutical is hoping Dukarb Plus will act as a savior against the flood of generics set to enter with Kanarb’s patent expiry. 3 doses of Dukarb Plus that were approved on March 31st are set to receive insurance benefits only 3 months since their approval. Dukarb Plus tab 30/5/12.5mg will be listed at ₩668, Dukarb Plus tab 60/5/12.5mg at ₩809, and Dukarb Plus tab 60/10/12/5mg at ₩880. The drug is used to treat patients with essential hypertension whose blood pressure is not adequately controlled with fimasartan+amlodipine combination therapy. By their indication, the drug’s use is expected to be limited to those who do not respond to Kanarb (fimasartan potassium) or Dukarb (fimasartan potassium+ amlodipine) In other words, with Dukarb Plus, the company may be able to retain the patients that use Kanarb or Dukarb. This is important as Boryung’s Kanarb and Dukarb are soon to be challenged by the entry of their generics as their substance patent will expire next year. Generic companies are busy developing generic versions of the two drugs in line with the substance patent expiry of Kanarb and Dukarb due on February 1st, 2023. Although a composition patent that lasts until August 8th, 2031 remains for Dukarb, generic companies are making efforts to invalidate or avoid the patent and launch latecomers after its substance patent expiry on February 1st, 2023. One relief on Boryung’s part is that the trial filed to avoid the composition patent filed by the generic companies had been turned down in March. However, the possibility remains for their entry as the generic companies immediately filed an appeal to the Patent Court of Korea. Market sharing is inevitable if generics pour into the market that had been monopolized by Kanarb and Dukarb. As the price of the originals is discounted upon the generics’ entry, this is expected to directly affect the sales of originals. Therefore, the performance of Dukarb Plus is ever more important to make up for the lost performance of the originals. If Dukarb Plus settles in the market, it may also be used as a later-line drug of Kanarb and Dukarb generics. One factor in favor of Dukarb Plus is that the share of three-drug combination therapies is increasing in the antihypertensive drug market. The market size has continued to increase since the first three-drug combination Sevikar HCT (olmesartan medoxomil+ amlodipine+hydrochlorothiazide, Daiichi Sankyo Korea) was released to exceed ₩100 billion last year. In other words, market size had increased fourfold in the past 5 years. In addition to Sevikar HCT, blockbuster drugs such as Amosartan Plus (Losartan+ amlodipine+chlorthalidone, Hanmi Pharmaceutical) and True Set (telmisartan + amlodipine+ chlorthalidone) were born in the increased market. Being the first three-drug combination that contains the ARB class fimasartan ingredient, the industry expects Dukarb Plus to follow the road to success taken by Sevikar HCT and Amosartan Plus. Based on UBIST last year, outpatient prescriptions of Sevikar HCT reached ₩34.8 billion, Amosartan Plus ₩28.2 billion, and Ture Set ₩15 billion. In the same period, Boryung’s fimasartan monotherapy Kanarb recorded ₩51.9 billion, and two-drug combo Dukarb ₩41.1 billion.
- Policy
- Chong Kun Dang faces Hanmi due to Rivaroxaban 2.5mg
- by Lee, Tak-Sun May 29, 2022 05:14pm
- Riroxia 10mg, which was released five months before the expiration of the original material patent by Chong Kun Dang, dominated the generic market. Next month, capsule-type 2.5mg products will be listed, and it is expected to face off with Hanmi over the generic marketChong Kun Dang anticoagulant Rivaroxaban 2.5mg will be listed as the second generic drug after Hanmi. Since Chong Kun Dang has shown extraordinary affection for anti-coagulation oral new drugs, it is expected that it will also focus its efforts on low dose Rivaroxaban products. According to the industry on the 22nd, Riroxia capsules 2.5mg will be listed next month. The original Rivaroxaban product is Bayer's Xarelto. However, 2.5mg product could not be released because it still has a patent for its product and a patent for its use, and Hanmi Pharmaceutical and SK Chemical acquired generic for exclusivity. There is also a difference in adaptation of the low dose (2.5 mg) and other doses. If 10, 15, and 20 mg are also used for treatment such as treatment of cardiac venous thrombosis and pulmonary embolism, 2.5 mg prodcut is used for the prevention of cardiovascular disease like aspirin. Studies show that using Xarelto 2.5 mg with Aspirin relatively reduced the risk of death from stroke, myocardial infarction, and cardiovascular disease in adult patients with coronary artery disease or symptoms than Aspirin alone. Accordingly, Bayer, which owns Xarelto, also paid more attention to generic blocking than other doses, such as registering 2.5 mg patent and use patent separately in Korea. Hanmi Pharmaceutical and SK Chemical succeeded in invalidating and avoiding 2.5mg of patents, respectively, and through this, they obtained generic for exclusivity. When the material patent was terminated in October last year, other pharmaceutical companies were not able to release 2.5mg products, but Hanmi, which acquired generic for exclusivity, was able to release the product. Currently, Original Bayer and Hanmi Pharmaceutical are the only pharmaceutical companies that have registered Rivaroxaban 2.5mg products. Chong Kun Dang could have been released in October last year if it had solved the original patent. Chong Kun Dang products are capsule formulations and are different from those of Korea. It seems that the registration was belatedly promoted because the patent for use was still in existence at the time. Currently, the Korean Supreme Court has ruled that the patent is invalid due to the request from Hanmi, and the final registration is about to be invalidated. The patent for use also expires on the 7th of next month. Although Chong Kun Dang belatedly releases 2.5mg products, they are faster than other post-refining drugs. This is because Rivaroxaban 2.5mg tablets can be released only on July 4 when the generic for exclusivity period between Hanmi and SK ends. Chong Kun Dang is working harder than any other pharmaceutical company in Korea on anticoagulant oral medicine generics. Rivaroxaban's other products, excluding 2.5 mg, also reduced some of the indications related to prolongation of life and pushed ahead with the launch five months before the expiration of the material patent. Chong Kun Dang requested a trial to confirm the scope of rights to avoid material patents. The request for the trial was dismissed in July last year, and Chong Kun Dang appealed to the Patent Court. However, Chong Kun Dang explained that Bayer's application for an injunction against patent infringement requested by the Seoul Central District Court has been rejected based on the judgment that it does not infringe material patents due to reduced indications. Chong Kun Dang ranks first in the Rivaroxaban market. Chong Kun Dang is conducting marketing under the plan to expand the original anticoagulant drugs to local clinics as they remain only in prescriptions for hospitals. Riroxia 2.5mg, which is listed at this time, is also expected to launch a large-scale marketing by Chong Kun Dang. It is expected that they will make desperate efforts to fight against Hanmi, which dominated the late drug market. The drug price was also set at 700 won, the same as 2.5 mg of Riroxban in Korea and Hanmi. As it is a showdown between the Chong Kun Dang, which has the strongest sales power in Clinics, and Hanmi, fierce competition is expected.
- Policy
- Xarelto's generic 15 items have been revoked
- by Lee, Hye-Kyung May 25, 2022 05:47am
- The MFDS canceled the item license of Rivaroxaban-based products, which were sold before the expiration date of the registered patent right as of the 27th. The cancelled product is Rixa 10mg, 15mg, 20mg, Wirelto 10mg, 15mg, 20mg, Jabaroxa 10mg, 15mg, 20mg Xarel-Q 10mg, 15mg, 20mg Xarelivan 10mg, 15mg, 20mg. The composition patent of the original Bayer Xarelto is until November 2024, and the material patent ended in October last year. However, it was confirmed that five pharmaceutical companies distributed generics to wholesalers before Xarelto's patent expired on October 3 last year, and the cancellation of the item license was decided this time. The reason was that generic products were distributed to wholesalers before the expiration of Xarelto's patent (October 3, 2021). According to Article 76(1)5-8 of the Pharmaceutical Affairs Act, "If a person who applied for an item permit or permission to change a registered patent right to sell it after the expiration of the period sells the drug" is subject to cancellation and suspension. The items that were decided to cancel the item license were reportedly distributed to some wholesalers first for distribution purposes after October 13 last year. Xarelto's outpatient prescription (UBIST) performance reached 59.6 billion won last year.
- Policy
- GSK meningococcal vaccine Bexero PFS was approved in Korea
- by Lee, Hye-Kyung May 25, 2022 05:47am
- The MFDS recently approved the item permission of Bexero PFS applied by GlaxoSmithKline (GSK). Bexero PFS is used to prevent invasive meningitis disease by Neisseria meningitidis B group at least 2 months of age. The safety and effectiveness of this vaccine in infants under 8 weeks of age have not been established. This vaccine injects deeply into the muscles. It is recommended to inoculate infants under 12 months of age to the outside of the thigh and to the upper arm deltoid area for those older than that. If inoculated at the same time as other vaccines, the injection site is different. Bexero PFS should not be intravenously, subcutaneous, or intradermally injected, nor should it be mixed in the same syringe as other vaccines. Bexero PFS is the only vaccine approved in Europe in 2016 to prevent type B meningitis and is a drug obtained by GSK through asset exchange with Novartis in 2015. The vaccination purpose of this vaccine is to prevent invasive meningococcal disease (IMD) by promoting antibody production that recognizes the vaccine antigens NHBA, NadA, fHbp, and PorAP1.4 (immune dominant antigen present in OMV components). In approximately 1000 meningococcal B group isolates collected in 5 European countries from 2007 to 2008, between 73 and 87% of meningococcal B group isolates, depending on the country of origin, showed MATS antigen profiles prevented by this vaccine. Phase 3 clinical trials showed that the vaccine was inoculated twice every two months for complement deficiencies (40 patients) aged 2 to 17, asymptomatic or spleen impaired patients (107), and healthy subjects (85) of the same age, with 87% and 97% for fHbp antigens, 95% and 100% for PorA P1.4 antigens, and 73% and 94% for NHBA. In the UK in September 2015, Bexsero PFS was introduced as NIP with a schedule of two additional inoculations (two months of age and four months of age) to infants.
- Policy
- Atopic Dupixent Pen will be available in Korea
- by Lee, Hye-Kyung May 24, 2022 05:54am
- Dupixent, an atopic treatment that is expected to improve convenience when self-injecting at home with easy operation, will be available in Korea. The MFDS recently approved 200mg and 300mg of Dupixent PF Pen applied by Sanofi Aventis. Dupixent PF Pen formulation, which was approved by the U.S. FDA in June 2020, secured adaptations to atopic dermatitis and asthma among the existing Dupixent PFS-approved indications. Existing Dupixent was approved in Korea as a treatment for atopic dermatitis that was ineffective with existing treatment in January 2018, and additional indications were approved as a severe or intractable bronchial asthma treatment that cannot control asthma symptoms with existing treatment in March 2019. Dupixent PF Pentype has the advantage of being able to inject drugs with simple operation. It consists of a visual and auditory feedback device that shows visual indications and sounds when the administration function is executed, as well as a pre-filled pen with hidden need or syringe with need shield. Existing PF pen is used for self-administration after being administered at hospitals and clinics or trained by medical personnel on how to administer it.
- Policy
- Fexclu priced at 90% of the weighted average of K-CAB+PPIs
- by Lee, Tak-Sun May 20, 2022 06:12am
- The price of ‘Fexclu Tab (fexuprazan), the potassium-competitive acid blocker (P-CAB) class gastroesophageal reflux disease (GERD) treatment developed by Daewoong Pharmaceutical was set at 90% of the weighted average price of the same class drug ‘K-CAB (tegoprazan, HK Inno.N) and PPI (proton-pump inhibitors) drugs. On the 12th, the Drug Reimbursement Evaluation Committee of the Health Insurance Review and Assessment Service had deemed that reimbursement for the drug was adequate if the company accepts a price below the appraised amount. The appraised amount was set at 90% of the price of ‘K-CAB+PPI’ after classifying the alternative of Fexuclu as 'K-Cap + PPI’ in the calculation formula. According to industry sources on the 19th, HIRA delivered its appraised amount to Daewoong Pharmaceutical. Daewoong Pharmaceutical, which had been hoping to be appraised using only K-CAB’s price, received the results with some disappointment. The price of K-CAB had been set at a more expensive level than others, being applied the 'preferential treatment for new drugs that entered the global market’ system that does not exist anymore. The current price ceiling for K-CAB is set at ₩1,300, but as most PPIs are now off-patent and have received significant price discounts, the weighted average price that includes PPIs as alternatives will widen the price gap between Fexclu and K-Cab. The current ceiling price of major PPIs are Nexium 40mg (esomeprazole) ₩1,015, Lanston LFDT tab 30mg (lansoprazole) ₩829, Noltec tab 10mg (ilaprazole) ₩1,131. If a drug’s price is set low in Korea, other countries that reference Korea’s price would also set a lower price for the drug, ultimately creating an unfavorable environment for overseas expansion of drugs. This is why the domestic pharmaceutical industry had requested a preferential clause be added for new homegrown drugs. An industry official pointed out, "In some cases, domestic prices are what impedes overseas penetration of the drugs, and the current drug price calculation standards do not properly reflect the efforts invested by the companies in developing new drugs." However, it is expected that Daewoong Pharmaceutical will accept the given price and proceed to conduct pricing negotiations with the NHIS, as the company has been conducting pre-marketing activities with the aim to release the drug in July, and the low possibility that the decision will be overturned even if the company raises an objection to HIRA’s appraised price. Therefore, Fexclu is expected to engage in a one-on-one competition with K-CAB in Korea with its lower price. K-CAB had raised ₩109.6 billion in outpatient prescriptions (source:UBSIT) last year.