- LOGIN
- MemberShip
- 2026-04-23 17:19:13
- Policy
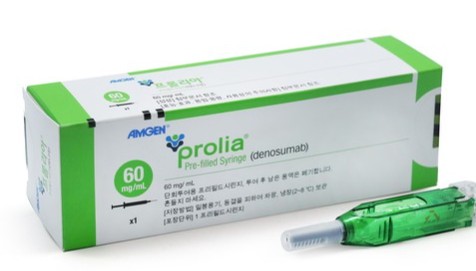
- HK Inno.N enters Prolia biosimilar market
- by Lee, Tak-Sun Feb 06, 2026 06:43am
- HK Inno.N has joined the biosimilar market for the osteoporosis treatment Prolia (denosumab).It is the third company to obtain regulatory approval for a Prolia biosimilar, following Celltrion and Samsung Bioepis. Competition in the approximately KRW 170 billion market is expected to intensify.Korea’s Ministry of Food and Drug Safety approved HK Inno.N’s Izambia Prefilled Syringe (denosumab) on the 4th.Izambia is a biosimilar with the same efficacy and effects as the original drug, Prolia Prefilled Syringe, which contains the same active ingredient. It is indicated for: ▲ Treatment of osteoporosis in postmenopausal women ▲ Treatment to increase bone mineral density in men with osteoporosis ▲ Treatment of glucocorticoid-induced osteoporosis ▲ Treatment of bone loss in non-metastatic prostate cancer patients receiving androgen deprivation therapy ▲ Treatment of bone loss in female breast cancer patients receiving adjuvant aromatase inhibitor therapy.Izambia was licensed from Spain’s mAbxience. In January 2023, HK inno.N signed an exclusive domestic licensing agreement with mAbxience for denosumab biosimilars. Under the agreement, HK inno.N secured exclusive supply rights for two denosumab biosimilars, as well as domestic regulatory approval and commercialization rights.Denosumab is an antibody therapy that targets the ‘RANKL’ protein, which is critical for the survival and function of osteoclasts, thereby exerting potent inhibition of bone resorption. The original drugs are Amgen's osteoporosis treatment ‘Prolia’ and ‘Xgeva’, used for preventing skeletal-related events in patients with bone metastases and treating giant cell tumor of bone. Both products were launched in 2016.On the same day, HK inno.N also received approval for Denbrayce Inj, a biosimilar to Xgeva.Amgen’s ProliaProlia, which recorded annual sales of approximately KRW 174.9 billion in 2024 (IQVIA data), began facing biosimilar competition after its substance patent expired in March last year. Celltrion’s Stoboclo was the first biosimilar to launch, followed by Samsung Bioepis’ Obodence last year.Stoboclo is co-marketed by Daewoong Pharmaceutical, while Obodence is co-marketed by Hanmi Pharmaceutical. Given that the original Prolia is co-marketed in Korea by Chong Kun Dang and Amgen, the denosumab market has effectively become a battleground for major domestic pharmaceutical companies.With HK inno.N now entering the market, competition is expected to further intensify. The newly approved denosumab biosimilars from HK inno.N are expected to be launched within the first half of the year, following completion of reimbursement pricing procedures.
- Policy
- Will the NA pass the 'mandatory INN prescription' bill?
- by Lee, Jeong-Hwan Feb 04, 2026 06:51am
- While the simplified post-notification system for substitution drugs through the government's electronic network was implemented on February 2, the pharmaceutical industry is increasingly focusing on the "limited International Nonproprietary Names (INN) prescription" bills currently pending in the National Assembly.Members of the ruling party on the Health and Welfare Committee have expressed a consensus on the need to accelerate the review of these bills, noting that the Lee Jae Myung administration has chosen mandatory INN prescription for national essential medicines as a national task to resolve supply instabilities.As of February 3, three bills related to limited INN prescription, introduced by Rep. Kim Yoon and Rep. Jang Jong-tae of the Democratic Party, are pending in the National Assembly.Rep. Kim Yoon's Pharmaceutical Affairs Act Bill proposes a legal definition for supply-unstable drugs and allows the Minister of Health and Welfare to recommend the use of INN for national essential and supply-unstable drugs.This method is considered less coercive as it does not mandate health professionals to use INN prescription during patient care and prescription.Rep. Jang Jong-tae's Medical Service Act & Pharmaceutical Affairs Act Bill requires the MOHW to designate supply-unstable drugs after deliberation by a Supply Management Committee. It mandates physicians to use INN instead of brand names when prescribing these designated drugs.Notably, Rep. Jang's proposal is considered highly coercive because it includes a penalty of up to one year in prison or a fine of up to KRW 10 million for physicians who violate the INN mandate.Some ruling party members on the Health and Welfare Committee state that if a legislative subcommittee is held this month, these bills should be officially adopted for review. They argue there is no reason to delay, especially given President Lee's recent criticisms regarding the slow pace of legislation.Rep. Kim's bill has been pending for over a year since its introduction in December 2024.The pharmaceutical industry is showing significant attention as the MOHW and the Health Insurance Review and Assessment Service (HIRA) began the implementation of the simplified post-notification system for substitution drugs, formulations, and dosages on February 2.An official from the Health and Welfare Committee stated, "The ruling and opposition party mangement groups are currently coordinating the schedule for the February legislative subcommittee," and added, "If the schedule is confirmed, there is a high possibility that the INN prescription bills will be placed on the agenda, as they are drawing significant attention from both the proposed offices and the committee members."
- Policy
- Merck’s mirdametinib drug receives GIFT designation
- by Lee, Tak-Sun Feb 04, 2026 06:51am
- Gomekli (mirdametinib) was approved by the US FDA in February last yearMerck’s neurofibromatosis treatment candidate mirdametinib has been designated for the Ministry of Food and Drug Safety’s (MFDS) expedited review support program, GIFT (Global Innovative products on Fast Track).As a result, the drug’s entry into the Korean market is expected to accelerate.According to the MFDS, mirdametinib was designated as a GIFT product on the 20th of last month.GIFT is an expedited review program launched in September 2022 to support rapid product development and provide patients with faster access to new treatment options.The program targets innovative medicines, including those for life-threatening diseases, rare diseases with no existing treatment alternatives, and new drugs developed by certified innovative pharmaceutical companies.The MFDS designates GIFT products based on a comprehensive evaluation of factors such as innovative therapeutic effects, contribution to addressing public health needs, and the developer’s efforts.Once designated, the review period is reduced by at least 25% (from 120 working days to 90 working days).The program also applies rolling reviews, under which submitted data are reviewed on an ongoing basis, and provides close communication between regulators and developers through product briefings and supplementary meetings. In addition, regulatory consulting and other support measures are offered to facilitate rapid commercialization.Mirdametinib is Korea’s 64th GIFT product. The proposed indication submitted to the MFDS is for the treatment of ‘pediatric and adult patients aged 2 years and older with neurofibromatosis type 1 (NF1) who have symptomatic, inoperable plexiform neurofibromas.’Mirdametinib is a selective inhibitor of mitogen-activated protein kinase kinases 1 and 2 (MEK1/2). It exerts antitumor activity by blocking MEK activity, thereby inhibiting the phosphorylation of Extracellular-regulated kinase 1 and 2 (ERK1 and ERK2) within the Mitogen-activated protein (MAP) pathway.It received Fast Track designation from the US FDA on February 11 last year and was approved by the EMA on July 17 last year. In Korea, it was designated as an orphan drug on December 11 last year.Mirdametinib was originally developed by the U.S. biotech company SpringWorks Therapeutics. In April 2023, SpringWorks was acquired by Germany-based Merck for USD 3.9 billion, giving Merck global commercial rights to the drug.Once launched, mirdametinib is expected to compete with the existing neurofibromatosis therapy Koselugo (selumetinib, AstraZeneca).Koselugo, which is also indicated for neurofibromatosis type 1, has been reimbursed by Korea’s national health insurance for patients aged 3 years and older since January 2024.Neurofibromatosis is a type of neurocutaneous syndrome characterized by abnormalities affecting both the skin and the central nervous system. Among its various subtypes, neurofibromatosis type 1 is the most common. Depending on the location of the neurofibromas, patients may exhibit brain tumor symptoms, and spinal involvement can lead to scoliosis. As of 2024, there are 6,490 known patients in South Korea.With recent increases in cases of reduced reimbursement for Koselugo, calls for improving reimbursement criteria are growing among neurofibromatosis patients and medical professionals.
- Policy
- Novartis withdraws all statin agents from the Korean mkt
- by Lee, Tak-Sun Feb 03, 2026 10:32pm
- Product photo of LescolNovartis is withdrawing all its statin agents used to treat hyperlipidemia from the Korean pharmaceutical market.Analysis suggests that the product's competitiveness has weakened due to other statin agents.According to the Ministry of Food and Drug Safety (MFDS) on the 2nd, Novartis Korea reported discontinuing supply of 'Lescol XL Tab (fluvastatin sodium),' the company's only statin-containing drug.Novartis stated, "In accordance with our global strategy and supply planning, we are discontinuing the supply of Lescol XL extended-release Tablet."The supply discontinuation date is set for July 31. Novartis noted, "Following the discontinuation of supply, please consider substituting with other statin-based products of the same mechanism of action (e.g., rosuvastatin, atorvastatin, simvastatin, pravastatin, etc.)."Lescol XL Extended-Release Tablet is currently the only statin product from Novartis that remains approved.Novartis entered the statin market in Korea in 1994, after obtaining MFDS approval for Lescol capsule. However, the product failed to achieve high performance as it lost out in competition with other statin products.Consequently, five of the six approved fluvastatin items were removed from the approval list in 2022 and 2023 due to withdrawals.Lescol XL was the only remaining product. However, following the latest report of supply discontinuation, fluvastatin will be completely withdrawn from the Korean market. There are currently no separate generic items available.Lescol XL Tab is indicated for ▲reduction of risk for cardiovascular disease, ▲hyperlipidemia ▲an adjunct to diet to reduce elevated total cholesterol, LDL-cholesterol, apo-B protein, and triglyceride levels and to increase HDL-cholesterol in pediatric and adolescent patients (boys: aged 9 to 16; girls: aged 10 to 16 and post-menarche) with heterozygous familial hypercholesterolemia who do not respond adequately to dietary therapy.Based on 2024 UBIST data, outpatient prescription amount totaled KRW 2.8 billion, a 23.7% decrease from the previous year. The sales volume is considered small when compared to Viatris' Lipitor (atorvastatin calcium trihydrate), a statin agent, which recorded KRW 188.6 billion in prescription sales during the same period.The underperformance is considered the most significant factor in this withdrawal from the Korean market.
- Policy
- Pharma, preparing for the future…generic launch in 2030s
- by Lee, Tak-Sun Feb 03, 2026 06:24am
- Product photos of Tagrisso (left) and K-CAB (right).Are they already preparing for the future? The Korean pharmaceutical industry is preparing for generics that would be launched five years later. They are also fiercely competing to secure priority marketing authorization by filing patent applications and seeking approvals.According to the Ministry of Food and Drug Safety (MFDS), 'Otinib tablet (Chong Kun Dang),' a generic version of the non-small cell lung cancer treatment Tagrisso (osimertinib), was approved on the 27th and also secured the priority marketing authorization. When assigned a priority marketing authorization, the drug receives an exclusive sales period of 9 months, during which the sale of generics containing the same active ingredient is banned.However, the effective date of the priority marketing authorization is not approaching soon. It is set to begin on December 28, 2023, and terminate on September 27, 2034. Approximately 8 years remain until the product launch. This is because the Tagrisso patent is set to expire on the specified date.Generic versions of K-CAB (tegoprazan), a P-CAB for gastroesophageal reflux disease (GERD), are increasingly receiving approvals. Yet, the priority marketing authorization is scheduled to take effect after August 26, 2031.Until now, pharmaceutical companies that received K-CAB generic approvals include GC Cross, Withus Pharm, Genuonesciences, Korea Drug, Ildong Pharmaceutical, KyungDong Pharm, and Hutecs Korea.As K-CAB is a mega-blockbuster item generating over KRW 200 billion in annual sales, numerous companies are competing for approvals to gain pre-occupancy in the generic market.Generics to Daiichi Sankyo's neuropathic pain treatment Taleaje (mirogabalin besilate) can be launched on or after 2031.Five pharmaceutical companies have applied for approval of their products containing mirogabalin besilate. Several companies have started patent challenges, excluding substance patents. Huons, Daewoong Pharm, Dong-A ST, JW Pharmaceutical, KyungDong Pharmaceutical, and Samjin Pharm.Five pharmaceutical companies that successfully challenged patents would be leading candidates to obtain priority marketing authorization. Despite this advantage, more than 5 years remain until the launch of generics, as the patent on the active ingredient is set to expire on June 4, 2031.Taleaje is still a non-reimbursed drug, and its sales cannot be estimated yet. Despite this circumstance, Korean generic companies have anticipated the bright future of the Taleaje market and have entered the race to secure a generic position.
- Policy
- Korea’s Innovative Pharmaceutical Company certification overhaul
- by Lee, Jeong-Hwan Feb 03, 2026 06:24am
- Director Kang-seop LimAttention is focused on how much the domestic pharmaceutical industry's request for rationalization of the ‘one-strike-out’ rebate regulation will be reflected in the reform plan for the Korea Innovative Pharmaceutical Company certification system, for which a legislative notice is being prepared by the Ministry of Health and Welfare within February.The MOHW has gathered extensive industry feedback on the current rule mandating the immediate revocation of innovative certification for companies found guilty of illegal rebates. Based on this input, the ministry plans to partially modernize regulations that could excessively hinder innovation in the pharmaceutical industry.However, regarding the previously discussed proposal to shift from immediate revocation to a points-based penalty system for rebate violations, the MOHW said it remains cautious and has not yet reached a decision.On the 1st, Director Kang-seop Lim of the Pharmaceutical and Bio-Industry Division met with the Ministry of Health and Welfare's press corps and explained, “We will announce the legislative notice for the enforcement decrees, enforcement rules, and notices related to innovative pharmaceutical companies by February at the latest.”The overhaul of the Korea Innovative Pharmaceutical Company certification system has emerged as a key issue for the industry, as it will operate in line with the drug pricing reform scheduled for implementation this year.Under the proposed drug pricing reform, price premiums would be applied depending on whether a company is certified as an innovative pharmaceutical company and the proportion of R&D investment in new drugs within such companies.Regarding the industry’s primary demand of converting the immediate revocation of innovative certification for rebate violations to a points-based system, Lim said, “This is still under discussion, and no decision has been made.”However, Lim explained that the ministry is preparing the reform plan in full consideration of the industry's persistent demand that the current penalty, where innovative certification is immediately revoked upon confirmation of a rebate incident, is excessively harsh for pharmaceutical companies dedicated to innovation, including new drug development.As the importance of the certification system has grown significantly with the upcoming drug pricing system reform, Lim explained that the ministry is particularly examining the feasibility of introducing several of the improvement measures requested by pharmaceutical companies.Overall, the MOHW is expected to pursue an administrative approach that maintains a certain level of penalties for unfair pharmaceutical trade resulting from illegal rebates, while improving regulations that excessively revoke or withdraw innovative certification based on overly stringent standards.According to industry sources, discussions are underway on rationalizing elements such as the period of application for penalties imposed on companies involved in rebate violations within the legislative and administrative notice.Lim said, “Pharmaceutical companies have repeatedly requested that the criteria for revoking innovative certification due to rebate violations be made more reasonable than they are now. We are internally reviewing measures that can both partially accommodate industry demands while continuing to regulate illegal rebates.”He added, “The decision on whether to adopt a points-based system is still under discussion. Currently, a rebate violation immediately renders a company ineligible as an innovative pharmaceutical company, leading to automatic revocation of certification. We are considering various options to improve this framework in a more rational way. By early February, we must simultaneously announce draft amendments to the enforcement decree, enforcement rules, and public notices. We will prepare a revised innovative certification scheme that enhances predictability for pharmaceutical companies.
- Policy
- Expanding indications…TAVI reimb still limited
- by Hwang, byoung woo Feb 02, 2026 02:17pm
- While TAVI (Transcatheter Aortic Valve Implantation) is becoming the standard for aortic valve replacement globally, in Korea, it remains tied to reimbursement criteria of '80 years old and inoperable.'While technology is advancing, the system remains in place.In the global market, TAVI has surpassed surgical aortic valve replacement (SAVR). According to U.S. Medicare claims data, since the number of TAVI procedures overtook SAVR starting in 2016, the gap has continued to widen.The biggest problem is the reimbursement criteria, which are out of step with global trends. The U.S. (65 and older) and Europe (70 and older) already recommend TAVI as standard treatment, significantly lowering age and risk thresholds. In contrast, Korea still divides reimbursement benefits (5% patient co-payment) based on age 80.For this reason, although TAVI procedures increased rapidly after the 5% co-payment reimbursement was introduced in May 2022, most of the increase was among elderly patients aged 80 and older. The patient group in their 70s appears relatively unchanged.Currently, for a patient in their 70s to receive TAVI, they must either bear costs totaling tens of millions of won, even with selective reimbursement, or receive an "inoperable" determination from two thoracic surgeons.The fact that the procedure fee for TAVI, which requires a high level of concentration from medical staff and cooperation from a multidisciplinary team, is only about one-third that of a general stent insertion (PCI) is also a factor hindering market growth.Presentation materials from the Insurance Committee session of the Korean Society of Interventional Cardiology (KSIC) 2026 Conference in Winter, reorganized by DailyPharm.Currently, the TAVI reimbursed fee is around KRW 540,000. This is significantly lower than PCI (about KRW 1.5 million) or pediatric pulmonary valve implantation (about KRW 2 million). In contrast, the U.S. reflects complexity through a co-surgeon extra-charge structure.Particularly, during a TAVI procedure, an 'integrated heart medical team,' a so-called Heart Team consisting of thoracic surgery personnel, anesthesiology, and radiology, is mandatory.Experts point out that, given labor costs, such a Heart Team has a structure in which the hospital loses money the more procedures it performs. Under the current 'low fee structure,' it inevitably reduces the incentive for medical institutions to actively expand the procedure. Ultimately, advances in technology and the limitations of the system are leading to longer wait times for patients and reduced access to treatment.However, another hurdle, namely the leadership between medical departments, is also mentioned as a limitation to the expansion of reimbursement. Under current law, to decide whether to perform a TAVI procedure, 'unanimous agreement' from an integrated medical team, including two thoracic surgeons, is required.In reality, the procedure is fundamentally impossible in small-to-medium-sized hospitals where only one thoracic surgeon is stationed. For these reasons, concerns are raised that it may serve as a threshold for entering reimbursement rather than as a consultative body to discuss treatment methods.The government stated it would accelerate system improvements by conducting public opinion surveys in the first half of 2026.정부는 2026년 상반기 의견수렴을 통해 제도 개선에 속도를 내겠다고 밝혔다On the one hand, given the need for long-term survival rate research and valve durability, some view the expansion of TAVI reimbursement for low-risk groups as premature.Conversely, it is argued that consideration is needed to select the best treatment for the patient rather than simply applying it uniformly by age.Because of this, the current discussion on expanding TAVI reimbursement can be seen as a conflict between exercising a veto to protect the number of surgeries rather than for the patient's benefit, and concerns about the abuse of procedures whose long-term safety has not been verified.The government signaled visible institutional changes within the first half of 2026. Yoo Jung-min, director of the Division of Health Insurance Benefits at the Ministry of Health and Welfare (MOHW), who spoke at the Insurance Committee session of the recently held the Korean Society of Interventional Cardiology (KSIC) 2026 Conference in Winter, emphasized three major principles: ▲reflecting international trends ▲respecting medical judgment ▲ expanding patient treatment choice.The policy is to restructure the system so that the integrated medical team can discuss "the best treatment" rather than "inoperability."However, industry experts agree that "simple modification of wording is not enough." The opinion is that the distorted growth of the TAVI market can only be corrected if complex factors such as ▲a rational lowering of the age criteria (to age 75, etc.) ▲the realization of fees matching the difficulty of the procedure ▲the incentivization of formal Heart Team operations are considered in the reimbursement discussion.Yoo stated, "We will speed up discussions on system improvement by broadly collecting opinions from various expert groups, such as related academic societies and patient organizations, during the first half of this year," and added, "The Ministry will strive to bring changes that can be made across the overall TAVI reimbursement criteria."
- Policy
- ‘INN prescribing can hinder high-quality drug development’
- by Lee, Jeong-Hwan Jan 30, 2026 11:00am
- Rep. Jia Han (middle) hosted an NA forum on ingredient-name prescribing for drugs with unstable supply, together with the Seoul Medical Association.Amid diverse causes of drug supply instability, such as API shortages, export restrictions at the national and global level, quality issues leading to GMP violation sanctions, and pharmaceutical companies exiting markets due to deteriorating profitability, the medical community has argued that the “limited international nonproprietary name (INN) prescribing” policy adopted as a national agenda by President Jae-myung Lee may be ineffective or even be counterproductive.Critics further pointed out that because pharmacies cannot stock every medication, and as combination drugs for conditions such as hypertension and diabetes cannot be simply substituted, INN prescribing could lead to a “pharmacy hopping” situation in which patients must visit multiple pharmacies to find one stocking the specific drug listed on their prescription.The medical community also reiterated that even drugs with proven bioequivalence are not identical medications.Under INN prescribing, patients may receive generics from different manufacturers depending on which pharmacy they visit or on each pharmacy’s inventory situation, but equivalence among generics is not fully guaranteed.These views were presented on the 29th at a National Assembly forum titled “INN Prescribing for Drugs with Unstable Supply,” hosted by Rep. Ji-ah Han of the People Power Party and organized by the Seoul Medical Association. The presentation was delivered by Chung-gi Kim, Policy Director of the Korean Medical Association.Director Kim explained that drug supply instability arises from multiple causes.He explained that supply disruption arises from a multifaceted combination of structural causes, such as reduced global supply, manufacturing/quality issues, or pharmaceutical companies voluntarily withdrawing product approvals due to drug price/market conflicts, as well as logistical causes stemming from regional/temporal imbalances in distribution.Kim argued that attempting to resolve such multifactorial supply instability through INN prescribing is unreasonable.He cited several limitations of INN prescribing, including the potential to trigger “pharmacy hopping,” the fact that even bioequivalent generics cannot be regarded as identical drugs, and the possibility of discouraging qualitative advancement and formulation innovation in pharmaceuticals.Specifically, he diagnosed that INN prescribing is ineffective because it operates on the premise that a generic substitute exists when production or supply is restricted due to API shortages or quality issues like GMP violations.In cases where pharmaceutical companies voluntarily exit the market due to the low profitability of low-priced essential medicines, INN prescribing could further intensify price competition and accelerate market withdrawal by manufacturers.Even in situations of temporary supply instability caused by logistics imbalances, where certain pharmacies or time periods experience stockouts, INN prescribing offers little benefit, and issues could instead be addressed through direct supply management by medical institutions or adjustments in physicians’ prescribing practices.Director Kim specifically pointed out that, on average, a single pharmacy stocks over 2,000 to 3,000 types of drugs, highlighting the physical limitations of resolving supply instability through INN prescribing.Considering the inherent mismatch where pharmacies cannot stock every drug, and the fact that combination drugs for conditions like hypertension and diabetes, with their varied ingredients and dosages, cannot be simply substituted, INN prescribing risks creating a “pharmacy hopping” problem. This forces patients to search for a pharmacy that stocks all the drugs listed on their prescription.Kim further argued that when INN prescribing leads to substitution of originator drugs with generics, or substitution among generics, the ‘trap of bioequivalence’ may place patients in a situation where they cannot take the exact medicine prescribed by their physician.Furthermore, he argued that INN prescribing could act as a mechanism that discourages pharmaceutical companies from advancing the quality of drugs or pursuing formulation innovations.If ingredient-name prescribing is implemented, manufacturers may be discouraged from investing in high-quality medicines, such as reducing tablet size for patients with dysphagia, improving moisture resistance and stability for better storage and efficacy, masking unpleasant tastes, or optimizing drug release profiles.Kim stated, “Drug supply instability is a matter of national security. The current shortage is a structural phenomenon that has become chronic. This instability stems from dependence on global supply chains, low drug pricing and bidding structures, and weak quality and production incentives.”He elaborated, “The structure that maintains unsustainable prices causes pharmaceutical companies to abandon manufacturing and exit the market, leading to repeated shortages and the collapse of treatment continuity. The solution lies in securing resilience through demand management, including predictive early warning systems, appropriate drug price compensation, supply-chain diversification, and the introduction of alternative treatments.”Kim concluded, “We must shift the policy paradigm from individual drug-centered approaches to treatment-continuity-centered policies, and shift the focus from administrative convenience to clinical impact and patient safety. What is needed is not fragmented or temporary INN prescribing, but an integrated, cross-ministerial governance r framework.”
- Policy
- Gvn’t ‘Reviewing various measures to address drug shortages’
- by Lee, Jeong-Hwan Jan 30, 2026 11:00am
- The Ministry of Health and Welfare (MOHW) has clarified that it is not relying solely on International Nonproprietary Name (INN) prescribing as a solution to addressing Korea’s drug shortages.The ministry stated that its administrative goal is to identify the underlying causes and develop appropriate, stage-specific policy tools to address drug shortages in a targeted manner, in consideration of the varying causes of Korea’s unstable drug supply.It also presented a plan to establish a social governance framework to resolve the unstable supply of medicines. This will involve operating a discussion table where doctors, pharmacists, the government, pharmaceutical companies, and patients all participate, aligned with the implementation of the revised Pharmaceutical Affairs Act.On the 29th, Jun-hyuk Kang, Director of the Division of Pharmaceutical Policy at MOHW, stated at a National Assembly policy forum on INN prescribing for supply-unstable medicines that “the government does not believe that drug supply instability can be resolved solely through INN prescribing.”Kang emphasized that shortages arise from a wide range of factors, including manufacturing, distribution, prescribing, and dispensing, and that the government will identify the root causes and develop appropriate measures to address them.Kang further noted that while past government policies focused on establishing systems to foster the pharmaceutical industry, future efforts will involve administrative support for the industry from a national health security perspective to resolve the issue of unstable drug supply.Kang said, “While there have been efforts to foster the industry in the past, there has never been support for active pharmaceutical ingredients from a public health security perspective. We have pointed out issues such as import dependency and shortage monitoring, and we are strengthening measures on that part as well, including requiring pharmaceutical companies to report supply disruptions.”He added, “Regarding drug pricing, while we've emphasized the specificity of drugs until now, the drug pricing system announced late last year will set prices reflecting contributions to alleviating supply instability. The Ministry of Health and Welfare is examining diverse causes of supply instability and considering phased policy measures accordingly.”Regarding governance mechanisms for addressing drug shortages, Kang stated that although such a structure did not previously exist, it will be established with the enforcement of the amended Pharmaceutical Affairs Act.“There is currently a consultative body under the Pharmaceutical Affairs Act, but it is structured for government-only participation in maintaining the healthcare system. However, the revised Act, which takes effect late this year, provides for a new governance framework. It establishes criteria for essential medicines and enables responses to supply-unstable drugs.”He added, “We have created a structure where representatives recommended by medical associations, pharmacist associations, and patient groups can join and discuss issues related to drugs with unstable supply within this governance framework. Regarding drug usage, we are building a substitution dispensing information system. This will streamline the substitution dispensing process within the legally defined standards, improving convenience for both physicians and pharmacists.He added, “INN prescribing is not being discussed solely for cost savings. The government is considering INN prescribing only for essential medicines, from the standpoint of ensuring treatment continuity for patient life and health. The government is not viewing INN prescribing as the sole solution to supply instability.”
- Policy
- "No drugs available for pediatric obesity…low dose phentermine combi as an option"
- by Lee, Jeong-Hwan Jan 29, 2026 08:17am
- On January 2, Rep. Seo Mi-hwa of the Democratic Party and the Korean Society for the Study of Obesity (KSSO) co-hosted a policy forum at the National Assembly Hall. Rep. Park Jie-won, a running candidate for the next National Assembly Speaker, attended to give a congratulating remarks.The medical community demanded the establishment of a regulatory track to allow limited prescribing of the psychotropic appetite suppressant phentermine·topiramate combination (Qsymia) in cases where the purpose of treating pediatric diseases, such as high-risk groups for adult-onset diseases, is clear.They argued that while obesity drugs such as GLP-1 analogs that can be used for pediatric patients aged 12 and older exist, they present challenges such as the difficulty of the injectable formulation and high medication costs; therefore, treatment options must be expanded by extending the indication for the low-dose phentermine and topiramate combination to those aged 12 and older.The Ministry of Food and Drug Safety (MFDS) understood the demands of prescribing clinicians and academia. Yet, it stated that the social controversy surrounding 'medical narcotic addiction' cannot be completely ignored.The MFDS explained that if a prescribing physician explains that the administration was for the purpose of treating a disease, the administration of narcotic appetite suppressants is possible even for pediatric patients.At the policy forum for expanding opportunities for pediatric obesity treatment medications, held on the 27th and hosted by the KSSO and Rep. Seo Mi-hwa of the Democratic Party, healthcare experts and the regulatory authority, the Ministry of Food and Drug Safety, met to exchange views on expanding the prescription of the phentermine/topiramate combination.Rep. Park Jie-won of the Democratic Party, the oldest active member of the National Assembly and a leading candidate for the next National Assembly Speaker, also attended the forum to support expanding obesity treatment options and to deliver congratulatory remarks.Rep. Seo Mi-hwa said, "Rep. Park Jie-won is the person who led me into politics," and added, "Along with Rep. Park, I will carefully review the opinions of medical sites and experts who believe that expanding treatment options for severe pediatric obesity is necessary.""Time to consider prescription options for pediatric patients aged 12 and older, even with restricted prescription conditions"Professor Hea Young Cho of the CHA University College of Pharmacy, who serves as the President of the Korean Society of Pharmaceutical Sciences and Technology (KSPST), appealed for the great necessity of creating a regulatory track to allow limited administration to pediatric patients aged 12 and older, highlighting Qsymia's low dependence and its clinically proven weight loss efficacy.In particular, Professor Cho noted that while the approval for phentermine monotherapy is set at age 16 and older, the reality that the low-dose phentermine·topiramate combination is more tightly regulated at age 18 and older has some irrational aspects.Professor Cho explained, "Phentermine monotherapy can be prescribed from age 16, yet the topiramate combination therapy is instead mandated for use in patients aged 18 and older," and added, "I wondered why this was the case. The prescription regulation for the combination is higher despite its lower phentermine content."Professor Cho further added, "Topiramate sustains weight loss effects by increasing satiety or stabilizing eating habits. Psychotropic components were developed as combination drugs to produce synergistic effects while lowering the dosage," and added, "Regarding dependence, the U.S. FDA manages the phentermine combination as Schedule IV, which is classified as having the lowest dependence."Professor Cho emphasized, "Because the weight loss effect of the low-dose phentermine combination has been scientifically proven, a relaxed prescription track must be created so that if necessary for pediatric treatment, the drug can be used while tracking and managing results through clinical monitoring," and added, "One way is to use a flexible prescription policy, such as lowering the prescription age for the phentermine combination by imposing limited prescription conditions and requiring sufficient clinical cases."Professor Hea Young Cho , Professor Park Jung-Hwan , Director Lee Jae Hyuk of the Korea Regulatory Affairs Professionals Society (from left)Professor Park Jung-Hwan of the Department of Endocrinology and Metabolism at Hanyang University Hospital, who serves as a director on the Committee of External Affairs and Policy of the KSSO, also urged the MFDS, the regulatory authority for the phentermine combination, not to take all management responsibility but instead grant obligations to prescribing doctors or selling pharmaceutical companies to operate the prescription age range flexibly.Professor Park said, "The use of low-dose phentermine combinations must be activated for pediatric patients. There is a case of a patient where we prescribed semaglutide up to stage 5, costing millions of won. However, the patient did not lose even 1kg of body weight," and added, "There are cases where phentermine·topiramate works for such patients."Professor Park stated, "Although semaglutide has been approved for use in pediatric patients, the reasons for obesity are diverse, so it would be good to relax the prescription age standards to accumulate and observe the necessary data and safety for the country," and "Rather than posing prescription restrictions, wouldn't it be more appropriate for the regulatory authority to open it up limitedly and present guidelines?"Director Lee Jae Hyuk of the Korea Regulatory Affairs Professionals Society also suggested that we should contemplate expanding medical options in the selection of medications for pediatric patients.The suggestion is that although the question of how to manage habitual and addictive drugs is a long-standing proposition for humanity, there is a great necessity to allow their use for medical or academic purposes.Lee said, "Even if production or distribution is strictly controlled, use should not be restricted as much as possible if medical necessity is recognized," and "Regardless of how the issue of abuse is managed as a separate focus, it is not a solution for abuse countermeasures to simply prohibit use unconditionally. A prescription environment where it can be used reasonably must be established.""No issues with pediatric patients using the phentermine combination if the treatment purpose is clear"Jeong Hyeon-cheol, Director of the Narcotics Policy Division at the MFDS, who participated as a panelist, stated that even now, there is no problem with prescribing the phentermine combination for pediatric patients where the necessity for administration is clear, such as those in high-risk groups for adult-onset diseases.This suggests that the MFDS has a sufficient range of understanding regarding the physician's right to prescribe for treatment purposes, and that simply expanding the administration indication for pediatric patients is difficult to view as the only solution.Jeong emphasized, "Regarding the facts, pediatric patients for whom use is essential due to the urgency of disease treatment risks can essentially be seen as not being restricted by prescription regulations," and "However, an inconvenient situation where a doctor has to provide a justification after prescribing may occur. This is because we operate a pre-notice system for the abuse of narcotics."Jeong highlighted, "For example, we request 4,500 doctors identified over a year as having exceeded the action standards for narcotic abuse to explain their reasons for prescription; afterward, through a committee of experts composed of doctors and pharmacists, we verify if the justification is valid, and the number decreases to units of hundreds or tens," and "The MFDS cannot restrict the physician's right to prescribe. It may be inconvenient, but the MFDS is allowing the prescription of phentermine combinations for pediatric patients."Jeong added, "To explain the questions the general public might have regarding the phentermine combination, the MFDS has approval standards, and the insurance authorities have reimbursement standards. The standards inevitably differ for each," and "After listening to the healthcare professionals, there is a need to allow its use for pediatric patients with metabolic syndrome, but the fundamental administrative goal is to prevent the abuse of psychotropic narcotics. If clinical data is submitted, we will review it."Jeong Hyeon-cheol, Director of the Narcotics Policy Division at the MFDS (left), Professor Jae-Hyuk Lee Professor Jae-Hyuk Lee of the Department of Endocrinology at Myongji Hospital (Director of the General Affairs Committee of the KSSO), who served as the chairperson, expressed concern that the method of 'justification after prescription' has significant irrational aspects and may hinder active treatment.Professor Lee emphasized, "The method of letting it pass if a proper exception is justified is not easy in clinical practice. Doctors try to avoid the task of justifying prescriptions that exceed regulations, which makes treatment difficult from the start," and "If the Ministry writes the prescription indication regulations well, healthcare professionals can accept prescribing according to those regulations."Professor Lee suggested, "If the regulations are excessively broad as they are now, it is difficult to use the drug (for pediatric patients) and post-monitoring regulation becomes complicated," and "It is ideal to manage the administration indications well and strictly regulate those that are exceeded. There are difficulties with the current method of 'do not use for now, but if you did, justify it and if that is correct, we will allow it.' Why not create the prescription regulations well so that doctors can use them properly?"