- LOGIN
- MemberShip
- 2026-05-02 22:49:22
- Policy
- The method of revaluation of wages that Godex has changed
- by Lee, Tak-Sun Jun 05, 2023 05:36am
- It is noteworthy whether HIRA will act as a variable by changing the evaluation method starting with the re-evaluation of benefit adequacy this year. The improved evaluation method is to increase the objectivity of the social demand part, which is the third evaluation item. Accordingly, in the case of drugs whose clinical usefulness is unclear but whose cost-effectiveness has been recognized, the evaluation of social needs is expected to be evaluated in more detail. The Ministry of Health and Welfare announced on the 30th of last month that the Health Insurance Policy Deliberation Committee deliberated on ways to partially improve the 2024 benefit adequacy re-evaluation target and benefit adequacy re-evaluation method. The proposal to improve the re-evaluation of benefit adequacy reported to this committee was prepared at a meeting held in December of last year to reinforce the method of evaluating social needs. The beginning was because of the Godex capsule. Although the clinical usefulness of Godex Capsule was unclear in the re-evaluation of reimbursement adequacy last year, the cost-effectiveness was recognized due to the drug price cut, and the benefit was finally maintained. In response, the members of the relevant committee raised an issue, and after re-submitting the agenda, the deliberation was finalized. At the same time, it was requested to strengthen the evaluation method such as social demand. The HIRA prepared and reported an improvement plan by reflecting only the part of the evaluation method requested by the committee in the 'review of drug benefit adequacy rationalization plan', which ended in March. In the improvement plan, the terms of the clinical usefulness item were changed to specify the purpose of the evaluation contents, and the quality level was considered in addition to the literature ratio that recognized the effect when evaluating clinical effectiveness. Accordingly, it was decided to judge that it is clinically useful only when it is evaluated above 'some' in terms of medical recommendation and clinical effectiveness. In addition, the social demand item is greatly improved. It will be specified in three evaluation items, including medical factors, and will be evaluated in a score method by determining details. Afterward, the scores were summed up and the final evaluation was decided by a committee composed of 11 members. In the meantime, social demand items have not been evaluated with points, but have been assessed by reviewing society's opinion collection, civic and patient group opinion submissions, financial impact through calculation of financial impact, medical importance, age, and patient's economic burden. In order to maintain benefits, it is possible if it is judged to be primarily clinically helpful or if the clinical usefulness is unclear, but cost-effective and social demand is high. In the revised plan, the conditions for maintaining reimbursement for drugs with unclear clinical usefulness are expected to become much more stringent. In particular, as this improvement plan is decided to be applied for re-evaluation this year, it is expected to affect drugs whose clinical usefulness is unclear. A total of six ingredients are in the process of being re-evaluated this year. Rebamipide, Limaprost Alfadex, Loxoprofen Sodium, Levosulpiride, Epinastine, and Sodium Hyaluronate eye drops will likely have mixed results for pharmaceutical companies.
- Policy
- Vemlidy IMDs compete with lower price
- by Lee, Tak-Sun May 31, 2023 05:37am
- Gilead The latecomers of the hepatitis B treatment Vemlidy that entered the market this year are focusing on their price competitiveness to occupy more shares of the market. Companies that had already reduced their price upon entry are also seemingly holding in check their competitors' prices. According to industry sources on the 29th, Daewoong Pharmaceutical reduced its insurance price ceiling for Vemliver Tab. (tenofovir ala fenamide hemitartar hydrochloride) from KRW 2,828 to KRW 2,473 from June. This is only 3 months after the drug was listed for reimbursement in March. Daewoong Pharmaceutical's voluntary price cuts are interpreted as being conscious of its competitors. Daewoong Pharmaceutical's product had been the most expensive among Vemlidy's latecomers in the market. Currently, all Vemlidy latecomers in the market are incrementally modified drugs that were developed with different salt formations. Dong-A ST, Daewoong Pharmaceutical, and Chong Kun Dang are competing in the market. With Donga ST being the first to be listed in February, Daewoong and Chong Kun Dang also launched their respective IMDs in March. The price of such salt-modified drug is allowed to be set at 90% of the original Vemlidy's highest price, but all three companies reduced the price to be lower than that at the time of initial listing. Dong-A ST’s Vemlia Tab was set at a 70% level at KRW 2,474, Chong Kun Dang’s at 69% at KRW 2,439, and Daewoong’s at 80% at KRW 2,828. All three products received generic marketing exclusivity. Not only were the drugs able to receive reimbursement a month faster than their competitors, but were allowed to sell their products without competition from other identical products for 9 months. The lower price set by the companies are therefore analyzed to be a measure the companies are making to preoccupy the market. Daewoong’s voluntary price cut made its drug KRW 1 cheaper than Donga ST’s Vemlia Tab. Vemlidy is on the rise in the hepatitis B treatment market. Gilead improved the tolerance and nephrotoxicity of its existing HBV treatment Viread and developed Vemlidy. The drug posted outpatient prescriptions of KRW 47.1 billion (UBIST) in Korea last year. Due to increased usage, Vemlidy's drug price was reduced 2 years in a row under the Price-Volume Agreement system. Last year, the price was cut from KRW 3,754 to KRW 3,535 after negotiations as a PVA ‘Type A’ drug, and this year, the price was cut from KRW 3,535 to KRW 3,370 through ‘Type B’ negotiations. This shows how rapidly Vemlidy's prescription volume has been increasing. Targeting this increasing market, the latecomers are aggressively marketing their drugs by lowering the insurance ceiling price. In addition, there is an analysis that the preoccupation of the market with such lower prices will create an entry barrier for other latecomers. In addition to the 3 pharmaceutical companies, Jeil Pharmaceutical, Korea Hutex Pharma, Samil Pharm, Dongkook Pharmaceutical, and Samjin Pharm have received approval for their salt-modified versions of Vemlidy.
- Policy
- Lowest price of dapagliflozin set at 30% of original price
- by Lee, Tak-Sun May 30, 2023 05:33am
- Original In 2 months after the entry of generics in the market, the price difference between the original diabetes treatment Forxiga Tab (Dapagliflozin propanediol hydrate) and its generics has become one-third. This is because in April last year when many Forxiga generics were listed at once to exceed 20, the insurance price ceiling dropped significantly as the listed drugs were applied a stepped pricing model. According to the industry sources on the 26th, the upper insurance price limit was set at KRW 241 for Ahngook New Pharm's ‘Newpharm Dapagliflozin Tab. 10mg’ that was listed on June 1st. Its price had been set at 85% of the 38.69% of the highest price, as more than 20 identical drugs have already entered the market. When more than 20 identical drugs are listed, the price is set at 85% of the lowest price of identical drugs,or 38.69% of the upper limit of the same product, whichever is lower. Based on the calculation standards, the price has fallen to one-third of the original Forxiga’s 10mg highest price (KRW 734). In the case of Forxiga generics, 20 generics immediately entered the market, therefore any additional listings will continue to discount the price. Diabetes treatments such as Forxiga are so cheap, such differences in drug price do not have a significant impact on prescriptions. However, in the case of long-term prescriptions, the patient's burden of cost does increase. For example, Forxiga’s KRW 734 for 90 days amounts to KRW 66,060, whereas the cheapest generic’s KRW 241 for 90 days amounts to KRW 21,690. At 30% of the co-payment rate, the patients will have to bear at most KRW 19,818 for the original and KRW 6,507 for the lowest generic, bringing the price difference borne to KRW 13,311. In other words, the analysis is that generics can be a sufficient alternative to original drugs if the patient wants a cheaper option. However, in Korea, where doctors have the right to prescribe, not many doctors consider the economic feasibility during prescriptions. Therefore, the industry emphasizes that the government should focus on policies that encourage the use of cheaper generics, rather than concentrating only on devising formulas that can reduce drug prices. Meanwhile, the price of the original Forxiga was supposed to be forcibly reduced to KRW 514 following the introduction of generics, but the previous price of KRW 734 won has been maintained as AstraZeneca Korea, the seller, applied and was allowed a stay of execution for the pricing discount from the court.
- Policy
- "Nurturing biopharmaceuticals as the second semiconductor"
- by Kang, Shin-Kook May 25, 2023 11:05pm
- Deputy Prime Minister and Minister of Strategy and Finance Choo Kyung-ho announced that he would soon announce a plan to foster the bio-industry, saying he would foster the second semiconductor for biopharmaceuticals. On the 24th, Deputy Prime Minister Choo visited the Aprogen plant in Osong to inspect the biopharmaceutical production site and said this in a meeting with industry officials. Deputy Prime Minister Choo explained in his remarks at the meeting that followed the on-site inspection of the production facilities that "the bio-industry is emerging as a high-tech industry that promotes the creation of new technologies and industries as it is converged with digital." He said, "It is becoming more important from the perspective of supply chain and economic security. We plan to foster the bio-industry as the second semiconductor." He explained, "The growth of the bio industry, such as the development of innovative new drugs based on AI, depends on how to utilize the vast amount of health and medical data accumulated in NHIS and private hospitals." “We will push for drastic regulatory improvements to nurture data-based digital healthcare startups,” he said. Deputy Prime Minister Chu emphasized, "To leap forward as a global biopharmaceutical production hub, the government plans to actively support private companies' bio investment, such as bio-manufacturing innovation R&D." The participants of the meeting highly appreciated the government's will to foster the bio-industry and requested the government's continued interest and support for the technology-intensive bio-industry, which requires enormous funds and time from product development to commercialization. In response, Deputy Prime Minister Chu said that he plans to announce a plan to foster the bio-industry in the near future, referring to the discussions at today's meeting.
- Policy
- Welireg·Camzyos are approved in Korea
- by Lee, Hye-Kyung May 25, 2023 05:46am
- Welireg, a treatment for von Hippel-Lindau and VHL disease characterized by multiple tumors, and Camzyos, which is used to improve motor function and symptoms in patients with NYHA class II-III obstructive hypertrophic cardiomyopathy, received domestic product approval. It announced on the 24th that it had approved Welireg of MSD Korea and Camzyos of BMS Korea. First of all, Welireg is a drug that inhibits HIF-2α related to cell proliferation, angiogenesis, and tumor growth. It can reduce the risk of surgical excision of the tumor. Hippel-Lindau disease is a rare disease in which incurable multiple tumors occur in the kidney, central nervous system, and pancreas due to mutations in tumor suppressor genes. This drug is used for the treatment of renal cell carcinoma, central nervous system hemangioblastoma, and pancreatic neuroendocrine tumor that does not require immediate surgery in adult patients with von Hippel-Lindau disease, providing new treatment opportunities for patients with these rare diseases. Camzyos is the first treatment for symptomatic obstructive hypertrophic cardiomyopathy in Korea and is expected to provide a new treatment opportunity for patients who have previously used only symptomatic therapy to relieve symptoms. Camzyos has been approved for 4 doses (2.5mg, 5mg, 10mg, 15mg) and relieves excessive contraction of the heart muscle by inhibiting cardiac myosin, and is effective in improving the motor function and symptoms of the patient. The Ministry of Food and Drug Safety said, "We will continue to do our best to expand treatment opportunities for patients with rare and intractable diseases by promptly supplying treatments whose safety and effectiveness have been sufficiently confirmed based on regulatory scientific expertise."
- Policy
- Domestic medical device market share ↑50%
- by Lee, Hye-Kyung May 24, 2023 08:26pm
- The share of domestically produced medical devices exceeded 50%. It is believed that the reason is that the production of medical devices such as in vitro diagnostic devices increased as public health medical products were approved for emergency use after Corona 19. Chae Gyu-han, head of the Medical Device Safety Bureau of the Ministry of Food and Drug Safety, said at a press briefing on the 23rd, “We are analyzing the production status of the medical device industry last year, and the market share of domestic manufactured products exceeded 50%.” It means that we can concentrate on producing essential medical devices for public health when an outbreak occurs and create a system that can stably supply them.” Therefore, the Ministry of Food and Drug Safety has expressed its will to improve the system so that companies with competence do not enter the market due to a lack of licensing experience. Manager Chae said, “The core of Regulatory Reform 2.0, which will be announced soon, is to ensure that the medical device industry develops in line with changes in the policy environment.” " said. Although the domestic medical device industry has developed due to the specificity of COVID-19, it is said that the content of Regulatory Innovation 2.0 will be to prepare an evaluation system to create international-level medical products with prepared technology and experience and to lead the medical device market. While Aimmed's 'Soames' and Welt's 'PillowRx' were recently approved as the 1st and 2nd digital treatment devices (DTx), it also indicated that it would prepare for the development of AI big data-based digital medical products in the future. Manager Chae said, "Deputy Director Oh Yoo-gyeong is also very interested in AI-based medical product development. The Power of the People Rep. Jong-heon Baek and Young-seok Seo of the Democratic Party each proposed the 'Digital Medical Products Act as part of this concern. The bill defines digital medical devices, digital convergence medicines, and digital medical and health support devices as digital medical products prepares an evaluation system and evaluates actual use, introduces an excellent management system certification system, and provides preferential treatment for health insurance benefits. there is. Manager Chae said, "It is expected that the bill will be discussed in the legislative subcommittee, and the management system has been established in line with the era of digital transformation so that digital technology can be applied to medical devices and medicines and used for health care." We will make it legislative,” he said. In line with the enactment of the bill, the Ministry of Food and Drug Safety is also preparing guidelines for the development of digital treatment devices. Manager Chae said, "Even before the legislation, we will prepare guidelines necessary for clinical trials and the development of digital treatment devices." In the field of innovative medical devices and innovative diagnostic devices, he emphasized investment support for selection and concentration. Manager Chae said, “There were concerns about the growth of overall medical devices and the growth of specialized fields, and as a result of conversations with the industry, there were many opinions that selection and concentration were needed.” “I think innovative medical devices and diagnostic devices have competitiveness. We plan to develop areas that can be developed and promote measures such as intensive support.” "Regulatory Innovation 2.0 and the promotion of the Digital Medical Products Act are one of the important projects this year," said Joo Seon-tae, head of the Medical Device Policy Department, who was present at the briefing of the Director of the Medical Device Safety Bureau. did. Seong Hong-mo, head of medical device management, said that she is promoting a project to prepare braille and sign language videos for medical devices to improve information access for the disabled. He said, "The recently revised bill contains information related to information accessibility for the disabled, and it will be a recommendation, not an obligation." He added, "If the sub-law is enacted, it will be conducted by meeting with disabled groups, investigating products that require braille or sign language videos, and recommending them to companies."
- Policy
- Domestic DM combi drugs selected for market competitiveness
- by Lee, Tak-Sun May 23, 2023 05:50am
- LG Chem and Dong-A ST are attracting attention by listing their DPP4i+SGLT2i combination as benefits, lowering the amount added. It is interpreted as being conscious of price competitiveness. According to the industry on the 22nd, Dong-A ST Sugadapa set the upper limit lower than the formula based on addition. This drug is an improved new drug combination, so it was able to receive a drug price that combines 59.5% of the individual ingredients. If there is no addition, you will receive the sum of 53.5% of the individual ingredients. For example, 434 won, 59.5% of 730 won for Evogliptin 5mg and 393 won, 59.5% of 734 won for Dapagliflozin 10mg, was the amount Sugadapa could receive. Dong-A ST decided to list it at 799 won, lower than the formula. As a result, it became the product with the lowest upper limit than the SGTL2!+DPP4i complex, which was released first last month. For the same reason, LG Chem's Zemidapa was also listed lower than the added amount. Zemidapa was able to receive the sum of 68% of the individual ingredients as an innovative new drug complex. The sum of 509 won, or 68% of 749 won for Gemigliptin 50mg and 499 won, or 68% of 734 won for Dapagliflozin 10mg, was able to receive 1,008 won. Zemidapa was also listed at a lower price of 940 won. However, even though the price of Zemidapa has been lowered, the upper limit is higher than that of competing drugs. After one year, the price of the two drugs will drop to the sum of 53.55% of the individual ingredients. Sugadapa is 784 won, and Zemidapa is 794 won. In any case, this is because a lower addition than the existing formula was applied. The low pricing of the two pharmaceutical companies is attracting attention in that they usually try to get a higher upper limit. This can be interpreted as an intention to induce the market to settle down by lowering the price, as metformin + SGLT2i + DPP4i has been covered by reimbursement since April, and the SGLT2i + DPP4i combination was released for the first time in May. Moreover, it seems to be aware of the competition as not only the two domestic products but also four other complexes were released.
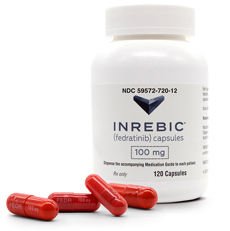
- Policy
- Myelofibrosis tx BMS Inrebic to be covered from June
- by Lee, Tak-Sun May 22, 2023 05:42am
- BMS Korea's myelofibrosis drug Inrebic will be covered from June. This drug is expected as a second-line treatment option for myelofibrosis patients, of which there are about 1,700 in Korea. According to the industry on the 19th, Inrebic will be listed as a salary at 39,520 won per capsule for the maximum amount from June 1st. Inrevic Capsule is indicated for the treatment of splenomegaly or symptoms associated with the following diseases in adult patients previously treated with Ruxolitinib (Brand Name: Jakavi). The following diseases are ▲primary myelofibrosis, ▲myelofibrosis after polycythemia vera, ▲and myelofibrosis after essential thrombocythemia. In February, it passed the HIRA Cancer Disease Review Committee and the Pharmaceutical Reimbursement Evaluation Committee, and after negotiations with the NHIS, it was listed as reimbursement in June. Inrebic is the first medicine to treat myelofibrosis in 10 years. Myelofibrosis is a rare hematological cancer in which normal hematopoietic function is impaired along with the excessive fibrotic proliferation of the bone marrow. Patients experience symptoms that affect their quality of life, including an enlarged spleen, fatigue, itchiness, weight loss, night sweats, fever, and bone pain. Until now, patients with myelofibrosis had no alternative when treatment with the JAK inhibitor Jakavi failed, but with the release of Inrebic, a second treatment option was created. Jakavi is a blockbuster drug with global sales of $388 million last year.
- Policy
- Vemlidy’s price 4.7% ↓...Vemliver’s voluntarily 12.6% ↓
- by Kim, Jung-Ju May 22, 2023 05:42am
- Gilead’s adult chronic hepatitis B treatment, ‘Vemlidi Tab (tenofovir ala fenamide) will be subject to Price-Volume Agreement price cuts and be sold at a 4.7% lower price starting next month. Daewoong Pharmaceutical opted to reduce the price of its latecomer Vemliver Tab by 12.6%. According to industry sources on the 21st, the Ministry of Health and Welfare (MOHW) ‘Amendment to the drug reimbursement list and reimbursement ceiling price table’ contains the abovementioned changes that will become effective as of the 1st of next month. ◆Price cut for PVA drugs = The PVA price cut will be applied to two products next month, and the two drugs underwent PVA negotiations with the National Health Insurance Service, one as a Price-volume agreement type ‘A’ and the other as a Price-volume agreement type ‘B’ drug. First, the drug that completed negotiations as a PVA Type A drug was Abbvie’s Rinvoq (upadacitinib), and its price will be reduced by 0.9%. Among new drugs listed as negotiable after the introduction of the drug price negotiation system, the government applies PVA A type to drugs whose claims amount in the same product category exceeds the expected claims amount by over 30%. By same product category, drugs that have the same company name, route of administration, ingredient, and formulation are classified as drugs of the same product category.’ The drug subject to ‘Type B’ price reduction was Gilead’s Vemlidy Tab, and its price was cut by 4.7%. The government applies PVA Type B and reduces the price of drugs ▲whose price ceiling had been already adjusted according to Type A, ▲ whose claims amount increased by over 60% from the previous year without undergoing Type A negotiations, or 4 years after receiving Type A negotiations, or ▲ has increased by over 10% but the increased amount exceeds KRW 5 billion and does not fall under PVA Type A. ◆Products that submitted voluntary price cuts= Five products in total decided to make voluntary price cuts. When a pharmaceutical company submits a request to lower its drug price to an amount lower than the ceiling price set for the product, the government adjusts the insurance drug price of the product to the requested amount. One voluntary price cut for a latecomer drug was made in line with the PVA price cut of Vemlidy. Daewoong Pharmaceutical opted to make a double-digit reduction in the price of its latecomer Vemliver Tab, by 12.6%. BMS Korea decided to make a price cut in the 1% range for each of its dosage strengths of Baraclude (entecavir). Janssen Korea’ made a voluntary price cut of 0.5% each for its Tremfya Prefilled Syringe (guselkumab) and Tremfya Autoinjection Inj.
- Policy
- Revival of omega-3 fatty acids...4g high-dose recommended
- by Choi, sun May 18, 2023 05:45am
- The Korean Society of Lipid and Atherosclerosis (KSoLA) disclosed the full version of its 5th edition of the Korean Guidelines for the Management of Dyslipidemia, in which the use of omega 3 was subdivided into the use of 'high dose and refined ingredients'. Although there has been controversy over its efficacy, the new guideline puts weight on the fact that the benefits of its use still outweigh its non-use. Annual Scientific Conference of the KSC with Affiliated Cardiac Societies On the 22nd, the Annual Scientific Conference of the KSC with Affiliated Cardiac Societies that was organized by 8 academic societies including the Korean Society of Cardiology, the Korean Heart Rhythm Society, the Korean Society of Heart Failure, and KSoLA, announced the ‘2023 KSoLA Guideline Update,’ ‘Evidence of Guidelines,’ and ‘Limitations in the Evidence’ were announced. A simplified version of the revised guidelines was announced last year, but this year, the full version was released to support its details and rationale. The new changes made in the 5th version were ▲the Method of diagnosis and standards, ▲Treatment standards. First, in the new diagnosis method and standard, the KSoLA showed the result that the non-fasting triglycerides level showed a high correlation with the risk of cardiovascular disease. In the standards, patients with coronary artery disease were recommended to lower their LDL cholesterol target level from less than 70 mg/dL to less than 55 mg/dL and lower it by more than 50% from baseline. In the case of patients with diabetes, the target goal is less than 70 mg/dL for patients with a disease duration of 10 years or more, one or more additional risk factors, or target organ damage, depending on the risk level. In addition, diabetic patients with 3 or more target organ damage or major cardiovascular disease risk factors can selectively consider reducing LDL cholesterol to less than 55 mg/dL. Furthermore, as exercise therapy, the recommendation grade for the use of wearables for fitness was newly presented as IIa, and additionally methods for using wearable equipment and a strategy for promoting physical activity were suggested. In the full version, society took a somewhat reserved position on the use of Omega 3, which was plagued by controversy over its cardiovascular protective effect. Professor Ye-Seul Yang (Endoctrioinology, SNUH), who presented on the 'New Changes on the KSoLA Guidelines,’ said, “The 5th edition of the revised guidelines segmented and reinforced the treatment criteria and treatment targets for dyslipidemia. The new guideline recommended different treatment standards by subdividing diabetes by risk group." Yang said, “Individualized guidelines were set according to specific groups such as those with stroke, chronic kidney disease, the elderly, adolescents, and familial hypercholesterolemia. In terms of drug therapy, the recommended grade was raised to emphasize that statins are a first-line treatment drug, and introduced icosapent ethyl(IPE) and the role of fibrate and omega-3 fatty acids in managing triglyceride.” As for whether omega 3 actually shows a protective effect against cardiovascular disease, large-scale studies have come to different conclusions. The most recent study that proved its efficacy observed an effect when only the IPE component was isolated from the omega-3 and used in high dosages. The KSoLA newly included the use of IPE in the recommendation for patients with atherosclerotic cardiovascular disease or diabetes who still have hypertriglyceridemia even after LDL cholesterol is controlled below the target level with statins. In the treatment of hypertriglyceridemia, "Use of fibric acid derivatives or omega-3 fatty acids that mainly lower triglycerides first are recommended as a priority (IIa, A), and even after achieving LDL cholesterol below the target level through therapeutic lifestyle improvement and statin drug treatment, if the triglyceride is 200 mg/dL or higher or the non-HDL cholesterol level is higher than the target value, drug treatments to lower the triglyceride can be considered (IIa, B)." KSoLA said, "In the recent REDUCE-IT study, 4g of IPE was administered per day in patients at high risk of atherosclerotic cardiovascular disease or diabetes, which reduced the incidence of cardiovascular disease by 26% compared to placebo. If hypertriglyceridemia persists in high-risk patients at a 200 mg/dL or higher level even after lifestyle improvement and statin administration, additional IPE (4g per day) can be administered to prevent cardiovascular disease (IIb, B)." However, KSoLA added a provision on account of the controversy in place. KSoLA added, “The effect of omega-3 fatty acids on reducing the risk of cardiovascular disease is still in controversy because of the varying results shown depending on formulation or dosage. In the recent REDUCE-IT study that was conducted in patients at high risk of cardiovascular disease whose triglyceride levels were higher than 135-499 mg/dL, the use of IPE, a high-purity EPA, twice a day significantly reduced death from cardiovascular disease and the occurrence of ischemic disease." “In the European practice guidelines that were revised in 2019, control of triglyceride using IPE was recommended for patients at high risk of cardiovascular disease, however, IPE is not being sold in the market yet. Also, in another recent study, the STRENGTH study, the combination of EPA and DHA did not show any benefit in cardiovascular disease, but rather increased the risk of atrial fibrillation compared to the control group." Also, the guidelines emphasized the need to use a high-dose high-purity ingredient for the combined use of statin and Omega 3. KSoLA said, “Combination therapy can be used to reduce LDL cholesterol and triglyceride at the same time. "The combination of 4 g of Omega 3 per day with simvastatin significantly decreased triglyceride and slightly increased HDL cholesterol."